Modified vs. Unmodified siRNA: A Comprehensive Guide to Efficacy, Design, and Therapeutic Application
This article provides a systematic comparison of unmodified and chemically modified small interfering RNAs (siRNAs) for researchers and drug development professionals.
Modified vs. Unmodified siRNA: A Comprehensive Guide to Efficacy, Design, and Therapeutic Application
Abstract
This article provides a systematic comparison of unmodified and chemically modified small interfering RNAs (siRNAs) for researchers and drug development professionals. It explores the fundamental limitations of unmodified siRNAs, including rapid nuclease degradation and immunogenicity, and details how strategic chemical modifications overcome these hurdles to enhance stability, specificity, and therapeutic efficacy. The content covers practical design methodologies, troubleshooting for experimental optimization, and advanced computational tools for validation. By synthesizing insights from recent high-throughput studies and clinical advancements, this guide serves as a strategic resource for informed siRNA design and application in both basic research and clinical therapeutics.
The Naked Truth: Understanding Unmodified siRNA Limitations and the Rationale for Chemical Enhancement
Small interfering RNA (siRNA) represents a revolutionary class of therapeutics with the potential to silence virtually any disease-causing gene in a sequence-specific manner. [1] By harnessing the conserved RNA interference (RNAi) pathway, exogenously administered siRNA duplexes, typically 21-23 nucleotides in length, can guide the degradation of complementary messenger RNA (mRNA), thereby preventing the synthesis of target proteins. [2] [1] This mechanism offers a precise approach for treating a wide range of diseases, from genetic disorders to viral infections and cancer. [3] [2] [4]
However, the journey of siRNA from a laboratory tool to a clinically viable drug is fraught with challenges, primarily when the siRNA is used in its unmodified form. [1] [5] Two of the most significant inherent barriers are their extreme susceptibility to degradation by nucleases in biological fluids and their propensity to activate the innate immune system. [2] [1] [5] These limitations drastically reduce the half-life of unmodified siRNAs, impair their ability to reach target cells in sufficient quantities, and can trigger undesirable inflammatory responses, compromising both efficacy and safety. [2] [5] This guide objectively compares the performance of unmodified siRNAs against their chemically modified counterparts, providing experimental data and methodologies that underpin this critical comparison in therapeutic development.
Quantitative Comparison: Unmodified vs. Chemically Modified siRNAs
The following tables summarize key experimental data comparing the properties of unmodified and chemically modified siRNAs, highlighting the performance gaps and the efficacy of various modification strategies.
Table 1: Comparative Stability and Pharmacokinetic Profiles
| Parameter | Unmodified siRNA | Chemically Modified siRNA | Experimental Measurement Method | Key Findings |
|---|---|---|---|---|
| Serum Half-Life | 6 minutes to ~1 hour [5] | Significantly extended (e.g., days for PS-modified oligonucleotides) [1] | Incubation in serum followed by gel electrophoresis or HPLC to intact siRNA; Pharmacokinetic (PK) studies after intravenous administration. [1] | Modifications like Phosphorothioate (PS) linkage confer high resistance to nuclease degradation. [1] |
| Nuclease Resistance | Highly susceptible to degradation by endo- and exonucleases. [2] | Greatly enhanced; specific patterns of 2'-OMe/2'-F can stabilize for intracellular depot. [6] [2] | QuantiGene assay or RT-qPCR to measure intact mRNA levels after siRNA serum pre-incubation. [6] | Full chemical modification is required for stability in the harsh endosome environment post-cell uptake. [6] |
| Renal Clearance | Rapid clearance due to low molecular weight and hydrophilicity. [5] | Reduced clearance; PS modifications increase protein binding (e.g., to albumin), prolonging circulation. [1] | PK studies monitoring siRNA levels in blood and urine over time. [1] | Appropriate protein binding prolongs half-life in circulation, which can be as long as a few days. [1] |
Table 2: Immunostimulation Profile and Silencing Efficacy
| Parameter | Unmodified siRNA | Chemically Modified siRNA | Experimental Measurement Method | Key Findings |
|---|---|---|---|---|
| Immune Activation (TLR7/8) | High, via GU-rich motifs (e.g., 5'-UGU-3', 5'-GUCCUUCAA-3') [3] | Greatly reduced; e.g., 2'-O-Me modification acts as a TLR7 antagonist. [3] [2] | ELISA to measure cytokine production (IFN-α, IL-6, TNF-α) in human PBMCs or plasma after siRNA treatment. [3] [7] | Substituting U or G residues with A can decrease pro-inflammatory cytokine production. [3] [7] |
| Immune Activation (RIG-I) | High, triggered by blunt-ended dsRNA and 5'-triphosphate groups. [3] | Suppressed; e.g., 2'-O-Me modification eliminates RIG-I mediated triggering. [3] | Reporter assay for interferon-stimulated response elements (ISRE); RT-qPCR for IFN-β mRNA. [3] | Incorporation of 2'-O-Me into both strands can eliminate immune stimulation without impairing RNAi. [3] |
| Gene Silencing Potency (in vivo) | Low to moderate, variable due to degradation | High and sustained; optimized modification patterns preserve or enhance RISC loading and activity. [6] [8] | QuantiGene assay or RT-qPCR to measure target mRNA levels in tissues; Western blot for protein knockdown. [6] | siRNA modification pattern (e.g., level of 2'-O-methyl content) significantly impacts efficacy. [6] |
| Off-Target Effects | Higher risk due to RISC loading of sense strand and tolerance to mismatches. [1] | Reduced; modifications like 2'-O-Me at position 2 of antisense strand decrease off-target silencing. [3] [1] | Microarray or RNA-Seq transcriptome analysis of cells treated with siRNA. [3] | Asymmetric modification (heavy on passenger strand) biases RISC loading towards the guide strand. [2] |
Experimental Protocols for Evaluating siRNA Challenges
To generate the comparative data cited in this guide, researchers employ standardized experimental protocols. Below are detailed methodologies for key assays.
Protocol 1: Assessing Nuclease Stability
Objective: To determine the resistance of siRNA to degradation by nucleases present in serum. [1] [5]
- Sample Preparation: Dilute the siRNA (unmodified and modified variants) in a solution containing 10-50% fetal bovine serum (FBS) or human serum. Use a nuclease-free buffer as a control.
- Incubation: Incubate the reaction mixtures at 37°C. Remove aliquots at predetermined time points (e.g., 0, 15, 30, 60, 120, 240 minutes).
- Reaction Termination: Halt the nuclease activity at each time point by adding a denaturing gel loading buffer or by heat-inactivating the serum.
- Analysis:
- Gel Electrophoresis: Separate the intact siRNA and its degradation fragments using denaturing or native polyacrylamide gel electrophoresis (PAGE). Stain the gel with a nucleic acid stain (e.g., SYBR Gold) and visualize using a gel documentation system. The intensity of the full-length siRNA band is quantified over time.
- HPLC Analysis: Use ion-pair reversed-phase high-performance liquid chromatography (HPLC) to separate and quantify intact siRNA. The area under the peak corresponding to the full-length product is measured.
Data Interpretation: A slower rate of degradation, evidenced by the persistence of the full-length siRNA band or peak over time, indicates superior nuclease stability. Chemically modified siRNAs typically show a significantly longer half-life under these conditions. [1]
Protocol 2: Evaluating Immune Activation
Objective: To quantify the immunostimulatory potential of siRNA through cytokine release. [3] [7]
- Cell Culture: Isolate human peripheral blood mononuclear cells (PBMCs) from fresh blood or use a relevant cell line (e.g., plasmacytoid dendritic cell line or HEK293 cells engineered to express TLR7/8).
- Transfection/Treatment: Seed cells in culture plates. Treat with siRNA (unmodified and modified) using a transfection reagent or simply add the siRNA directly to the culture medium. Use known TLR ligands (e.g., imiquimod for TLR7) as positive controls and untreated cells as negative controls.
- Incubation: Incubate cells for 6-24 hours at 37°C and 5% CO₂.
- Sample Collection: Collect cell culture supernatants after incubation.
- Cytokine Measurement: Use enzyme-linked immunosorbent assays (ELISAs) specific for key cytokines such as interferon-alpha (IFN-α), tumor necrosis factor-alpha (TNF-α), and interleukin-6 (IL-6) to quantify their concentrations in the supernatants, following the manufacturer's protocol.
Data Interpretation: High levels of cytokine production in response to an siRNA sample indicate strong immune activation. Effective chemical modifications (e.g., 2'-O-Me, 2'-F) will show a marked reduction in cytokine levels compared to unmodified siRNA. [3] [7]
Protocol 3: Measuring Gene Silencing Efficiency (QuantiGene Assay)
Objective: To directly quantify the knockdown of target mRNA in cells, which reflects the functional efficacy of the siRNA. [6]
- Cell Treatment: Plate appropriate target cells and transfert with siRNA using a standard transfection protocol. Include a non-targeting siRNA (scrambled sequence) as a negative control.
- Incubation: Allow gene silencing to proceed for 48-72 hours.
- Cell Lysis: Lyse the cells according to the manufacturer's protocol for the QuantiGene assay.
- Hybridization: The lysate is added to a capture plate where branched DNA (bDNA) probes hybridize to the target mRNA.
- Signal Amplification & Detection: A series of amplifier and label probes hybridize to the complex, resulting in signal amplification. Chemiluminescence is measured using a luminometer. The signal intensity is directly proportional to the amount of target mRNA in the original lysate.
Data Interpretation: Silencing efficiency is calculated as the percentage reduction in luminescent signal relative to the negative control. This method directly measures endogenous mRNA levels and is not affected by siRNA-mediated transcriptional regulation or off-target effects on housekeeping genes used in RT-qPCR. [6]
Visualizing Immune Activation Pathways and Experimental Workflows
siRNA Immune Recognition Pathways
The following diagram illustrates the primary pathways through which unmodified siRNAs activate the innate immune system, a key challenge that modified siRNAs are designed to overcome.
Workflow for siRNA Stability and Efficacy Testing
This diagram outlines a standard experimental workflow for comprehensively evaluating the stability and functional efficacy of siRNA candidates, as described in the protocols.
The Scientist's Toolkit: Essential Reagents for siRNA Characterization
Table 3: Key Research Reagent Solutions for siRNA R&D
| Reagent/Category | Specific Examples | Primary Function in Experimental Context |
|---|---|---|
| Chemical Modification Phosphoramidites | 2'-F, 2'-O-Me, 2'-MOE, LNA, UNA, Phosphorothioate (PS) [6] [2] [1] | Used in solid-phase synthesis to produce chemically modified siRNA strands. Enhance nuclease resistance, reduce immunogenicity, and fine-tune pharmacokinetics. [6] [2] |
| Stability Testing Reagents | Fetal Bovine Serum (FBS) / Human Serum, HPLC columns (e.g., ion-pair RP), PAGE reagents [1] [5] | Provides nucleases for stability challenge. Tools for separating and quantifying intact siRNA and its degradation fragments. |
| Immune Assay Kits | Human IFN-α ELISA Kit, Human TNF-α ELISA Kit, Human IL-6 ELISA Kit [3] [7] | Quantifies cytokine secretion from immune cells (e.g., PBMCs) to objectively measure the immunostimulatory potential of siRNA. |
| Gene Silencing Assay Kits | QuantiGene Plex Assay, RT-qPCR reagents & kits [6] | Directly measures levels of target endogenous mRNA in cell lysates, providing a readout of siRNA functional efficacy. |
| Delivery/Transfection Reagents | Cationic lipids (Lipofectamine), GalNAc conjugates, lipid nanoparticles (LNPs) [2] [5] | Facilitates cellular uptake of siRNA for in vitro and in vivo studies, a critical step for functional testing. |
The empirical data and experimental protocols detailed in this guide unequivocally demonstrate the fundamental limitations of unmodified siRNAs as therapeutic agents. Their rapid degradation in serum and pronounced activation of the innate immune system present formidable barriers to clinical efficacy and safety. [2] [1] [5] The field has conclusively shown that strategic chemical modification—including 2'-sugar modifications, phosphorothioate backbones, and base alterations—is not merely an enhancement but a necessity. [3] [6] [1] These modifications directly address the core vulnerabilities of unmodified siRNA by dramatically improving metabolic stability, suppressing immune recognition, and ultimately enabling effective and durable gene silencing in vivo. The transition from unmodified to chemically engineered siRNA scaffolds, as evidenced by the growing number of approved drugs, marks a critical evolution in realizing the full therapeutic potential of RNA interference.
Small interfering RNA (siRNA) therapeutics represent a revolutionary class of drugs capable of silencing disease-causing genes with high specificity via the RNA interference (RNAi) pathway [5]. Since the first FDA approval in 2018, several siRNA drugs have been successfully commercialized for conditions like hereditary transthyretin-mediated amyloidosis, acute hepatic porphyria, and hypercholesterolemia [2]. However, the translational journey from a promising molecule to a clinically viable drug is fraught with challenges. Unmodified siRNAs are inherently unsuitable as medications due to their extreme susceptibility to rapid degradation by ubiquitous nucleases in biological fluids, a short plasma half-life of less than 10 minutes, and a high propensity to elicit undesired immune responses [5] [9]. These intrinsic properties drastically reduce their therapeutic half-life and prevent them from reaching target cells in sufficient quantities, thus compromising their efficacy and safety profile [2]. This review delineates how strategic chemical modifications are imperative to overcome these clinical barriers, directly comparing the performance of unmodified and chemically modified siRNAs through experimental data.
Comparative Analysis of Modified vs. Unmodified siRNA Performance
Chemical modifications are engineered to address specific limitations of unmodified siRNA without compromising their ability to engage the RNAi machinery. These modifications can be categorized based on the structural component of the siRNA they target: the sugar ribose, the phosphate backbone, the nucleobase, or the terminal groups [2].
The table below summarizes the key performance differentiators between unmodified and chemically modified siRNAs across critical parameters for clinical application.
Table 1: Performance Comparison of Unmodified vs. Chemically Modified siRNAs
| Performance Parameter | Unmodified siRNA | Chemically Modified siRNA | Key Modifications & Experimental Evidence |
|---|---|---|---|
| Serum Stability & Half-life | Rapid degradation in serum; Plasma half-life <10 min [9]. | Greatly enhanced stability. LNA modifications confer superior serum stability [10]. | LNA, 2'-F, 2'-OMe, PS backbone. In mouse serum, LNA-modified siRNAs showed superior stability versus unmodified siRNA [10]. |
| In Vivo Knockdown Efficacy | Ineffective in tumor knockdown without formulation [10]. | Superior efficacy. ~50% target mRNA reduction in tumor xenografts without formulation [10]. | LNA, UNA. In a mouse model, LNA and UNA modified siRNAs achieved significant EGFP knockdown where unmodified siRNA failed [10]. |
| Off-Target Effects | High number of differentially expressed off-target genes [11]. | Up to 90% reduction in off-target genes [11]. | LNA in seed region. Silencer Select siRNAs with LNA reduced off-target genes by 90% in gene expression arrays [11]. |
| Silencing Duration (Dividing Cells) | Silencing lasts 3-7 days in vitro [12]. | Extended duration. >30 days with 5'-(E)-Vinylphosphonate (5'-VP) [12]. | Fully modified backbone + 5'-VP. In dividing cancer and immune cells, 5'-VP modification was critical for long-term silencing [12]. |
| Immune Stimulation | High risk of activating innate immune system via TLRs/RLRs [5]. | Greatly reduced immunogenicity [5]. | 2'-OMe, 2'-F, Pseudouridine. Incorporation of 2'OMe or LNA prevents recognition by Toll-like receptors [10] [2]. |
Detailed Experimental Protocols and Data
Experiment 1: In Vivo Efficacy and Stability in a Tumor Xenograft Model
- Objective: To evaluate the in vivo efficacy, serum stability, and biodistribution of LNA- and UNA-modified siRNAs compared to unmodified siRNA.
- Methodology:
- siRNA Design: Tested sequences targeting EGFP mRNA, including unmodified, LNA-modified, UNA-modified, and a novel LNA-stabilized sisiRNA design [10].
- In Vivo Model: Nude mice bearing human MiaPaca-II tumor xenografts [10].
- Administration: siRNAs were administered "naked" (without formulation) via osmotic minipump at a dose of 0.25 mg/kg/day for one week [10].
- Stability Assay: Incubation of siRNAs in fresh mouse serum with analysis over time [10].
- Biodistribution: Mice were administered tritiated siRNA and sisiRNA; tissue distribution was measured after 30 minutes [10].
- Efficacy Readout: EGFP mRNA levels in tumors were quantified to assess knockdown [10].
- Key Results:
- Stability: LNA-modified siRNAs demonstrated superior stability in mouse serum. UNA-modified siRNA showed partial breakdown but retained a stable core fragment [10].
- Biodistribution: LNA-modified siRNA and sisiRNA showed nearly identical profiles, with primary uptake in kidneys and significant uptake in tumors and liver [10].
- Efficacy: LNA, UNA, and sisiRNA treatments achieved approximately 50% reduction in EGFP mRNA, whereas unmodified siRNA was ineffective [10].
Experiment 2: Profiling and Mitigating Off-Target Effects
- Objective: To quantify and reduce sequence-specific off-target effects mediated by the siRNA seed region.
- Methodology:
- siRNA Design: Comparison of unmodified siRNAs and siRNAs incorporating Locked Nucleic Acid (LNA) modifications. Bioinformatic filtering was applied to remove sequences with seed regions resembling miRNAs [11].
- Cell-based Assay: Transfection into HeLa cells [11].
- Global Gene Expression Analysis: Whole-genome microarrays were used to detect differentially expressed genes (≥2-fold change) compared to mock-transfected controls [11].
- Key Results:
- Off-Target Reduction: Negative control siRNAs with Silencer Select LNA modifications reduced the number of off-target genes by up to 90% compared to their unmodified counterparts [11].
- Phenotypic Reliability: The modifications eliminated off-target apoptotic phenotypes observed with 10 different unmodified siRNAs, leading to more reliable data for genes like PLK and WEE1 [11].
Experiment 3: Extending Silencing Duration in Dividing Cells
- Objective: To investigate factors, particularly chemical modifications, that extend the duration of gene silencing in rapidly dividing cells, which is crucial for immuno-oncology applications.
- Methodology:
- siRNA Design: Utilized a fully modified backbone (2'-F, 2'-OMe, phosphorothioates) with an asymmetric structure. The key variable was the stabilization of the guide strand with a 5'-(E)-vinylphosphonate (5'-VP) moiety [12].
- In Vitro Model: Rapidly dividing cancer and immune cells [12].
- In Vivo Model: Mouse models for in vivo validation [12].
- Dosing Strategy: Tested single versus repeated dosing to saturate intracellular depots [12].
- Efficacy Readout: Duration of target mRNA silencing was monitored over time [12].
- Key Results:
- Saturation Dosing: Repeated dosing to saturate intracellular depots had a minimal impact on prolonging silencing duration in dividing cells [12].
- 5'-VP is Critical: The 5'-VP modification on the guide strand, in combination with a fully modified backbone, was essential for extending silencing duration beyond 30 days, both in vitro and in vivo [12].
- Potency: The 5'-VP modification also contributed to greater potency at lower concentrations [12].
Visualizing the siRNA Pathway and Modification Strategies
The RNAi Mechanism and Clinical Barriers
Chemical Modification Strategies to Overcome Barriers
The Scientist's Toolkit: Essential Reagents and Solutions
Table 2: Key Research Reagents and Technologies for siRNA Development
| Reagent / Technology | Function & Role in siRNA Research | Example & Key Benefit |
|---|---|---|
| Locked Nucleic Acid (LNA) | Ribose modification that "locks" the sugar structure. Enhances binding affinity (Tm), nuclease stability, and reduces off-target effects by promoting guide strand bias [10] [11]. | Silencer Select siRNAs: Incorporate LNA to reduce off-target effects by up to 90% and improve potency [11]. |
| 2'-Fluoro (2'-F) | Sugar modification that dramatically improves resistance to nuclease degradation and enhances siRNA potency while maintaining AGO2 compatibility [13] [2]. | Patisiran & Inclisiran: Utilize 2'-F in their modification patterns to achieve long-lasting gene silencing in patients [2] [9]. |
| Phosphorothioate (PS) | Backbone modification where a sulfur atom replaces a non-bridging oxygen. Increases stability against nucleases and improves pharmacokinetics by promoting binding to serum proteins [2] [12]. | Standard in clinical siRNAs: Used in nearly all advanced therapeutic siRNAs to prolong half-life in serum and tissue [12]. |
| 5'-(E)-Vinylphosphonate (5'-VP) | A 5'-end modification that confers resistance to 5'-exonucleases and enhances AGO2 binding. Critically extends silencing duration in rapidly dividing cells [12]. | Dividing Cell Therapy: Enabled >30 days of silencing in cancer and immune cells, overcoming a major hurdle for immuno-oncology [12]. |
| GalNAc Conjugation | A trivalent N-acetylgalactosamine ligand that enables highly efficient uptake into hepatocytes via the asialoglycoprotein receptor (ASGPR) [2] [9]. | Givosiran, Inclisiran: Enables subcutaneous administration with high potency and long dosing intervals (e.g., twice yearly) [9]. |
| Advanced Bioinformatics | Machine learning algorithms for siRNA design that predict efficacy and minimize off-target effects through seed region filtering and thermodynamic analysis [11]. | Silencer Select Algorithm: Increases predictive accuracy by 28% over previous generations, enabling highly potent siRNAs [11]. |
The collective experimental evidence solidifies the therapeutic imperative for chemical modifications in siRNA-based drugs. Data from in vivo models demonstrates that strategic incorporation of modifications like LNA, UNA, 2'-F, and 5'-VP directly translates to superior clinical attributes: enhanced stability overcoming rapid degradation, significantly reduced off-target effects ensuring cleaner phenotypes, and extended duration of action enabling viable dosing regimens. While unmodified siRNAs remain a valuable research tool, their inherent instabilities and immunogenic potential render them clinically unviable. The ongoing refinement of chemical architectures and modification patterns, as evidenced by the progression of siRNA drugs through clinical trials, continues to expand the frontiers of treatable diseases, solidifying RNAi as a pillar of modern therapeutics.
Small interfering RNAs (siRNAs) are a class of double-stranded oligonucleotides, typically 21-25 nucleotides in length, that harness the natural cellular pathway of RNA interference (RNAi) to silence the expression of disease-causing genes [6]. Since the first FDA approval of an siRNA therapeutic in 2018, these agents have revolutionized the treatment of liver diseases and hold promise for a wide range of conditions [6]. However, unmodified siRNAs face significant challenges that limit their therapeutic application, including rapid degradation by nucleases in serum, activation of the innate immune system, and difficulties in cellular uptake [14] [15]. Chemical modification of the siRNA backbone and sugar moiety is therefore not merely an option but a necessity to impart "drug-like" properties, enhancing stability, specificity, and pharmacokinetic profiles while minimizing unwanted immune responses [14].
The following diagram illustrates the core workflow for developing therapeutic siRNAs, highlighting where chemical modification design fits into the process.
Detailed Analysis of Core Modification Types
2'-O-Methyl (2'-OMe) Modification
The 2'-O-methyl (2'-OMe) modification involves the addition of a methyl group to the 2' oxygen of the ribose sugar. This alteration increases nuclease resistance and reduces the immunostimulatory potential of siRNA, making it one of the most widely used modifications in therapeutic sequences [14] [16].
Key Functional Impacts:
- Stability & Affinity: Replacing the 2'-OH group with a 2'-OMe group significantly enhances the metabolic stability of siRNAs against degradation by endo- and exonucleases. It also increases binding affinity to the target mRNA [17] [16].
- Thermodynamics: 2'-OMe modifications generally favor the C3'-endo (North) sugar pucker, which is characteristic of A-form RNA helices and pre-organizes the oligonucleotide for duplex formation [17].
- Immunogenicity: A key benefit of 2'-OMe modification is its ability to mitigate unwanted immune stimulation. One study demonstrated that while an unmodified siRNA stimulated production of INFα and TNFα in human peripheral blood mononuclear cells, a fully modified counterpart did not [14].
2'-Fluoro (2'-F) Modification
The 2'-deoxy-2'-fluoro (2'-F) modification substitutes the 2'-hydroxyl group on the ribose ring with a fluorine atom. This change confers exceptional thermal stability and nuclease resistance, and is exceptionally well-tolerated by the RNA-induced silencing complex (RISC) [14].
Key Functional Impacts:
- Duplex Stability: The 2'-F modification imparts significant thermal stability to the siRNA duplex. Studies show the melting temperature (Tm) of a 2'-F-modified siRNA can be almost 15°C higher than that of an unmodified duplex [14].
- Mechanism of Stability: Contrary to the common assumption that stability gains are primarily entropic due to conformational pre-organization, research indicates the higher stability of 2'-F-RNA is chiefly due to a favorable enthalpy change [14]. This suggests fluorine may polarize the nucleobase, strengthening Watson-Crick hydrogen bonds and base stacking.
- Hydration Properties: Crystallographic studies reveal that the fluorine atom at the 2'-position does not participate in hydrogen bonds with water molecules in the minor groove. This results in poorer hydration of the 2'-F-RNA duplex compared to unmodified RNA, which has an intricate water structure in the minor groove [14].
Phosphorothioate (PS) Modification
The phosphorothioate (PS) modification involves the substitution of one of the non-bridging oxygen atoms in the phosphate backbone with a sulfur atom. This is a backbone modification, distinct from the 2' sugar modifications, and primarily enhances stability and pharmacokinetics [18] [19].
Key Functional Impacts:
- Nuclease Resistance: The PS linkage confers resistance to nuclease degradation, dramatically increasing the half-life of siRNAs in biological fluids [19].
- Protein Binding: PS modifications enhance hydrophobic protein-binding properties, which can improve tissue distribution and cellular uptake by promoting associations with serum proteins [18] [16].
- Stereochemical Complexity: A significant drawback of PS modifications is that each PS linkage introduces a chemically undefined stereocenter (unless synthesized with stereodefined amidites). This results in a mixture of 2^n unique molecules in the final product, complicating analysis and purification [20].
Phosphorodithioate (PS2) and Combined Modifications
The phosphorodithioate (PS2) modification replaces both non-bridging oxygen atoms in the phosphate backbone with sulfur atoms. This eliminates the chiral center, creating an achiral linkage and simplifying the manufacturing process [20] [19].
Key Functional Impacts of PS2:
- Achiral Advantage: The absence of a stereocenter in PS2 linkages allows for better batch-to-batch quality control, a significant advantage over PS modifications [19].
- Stability and Potency: PS2-modified siRNAs show improved serum stability and gene silencing over PS-modified siRNAs. When combined with 2'-OMe groups in a MePS2 configuration (PS2 with a 2'-OMe on the same nucleotide), siRNAs can show dramatically increased loading into the RISC and enhanced anti-tumour activity, outperforming modifications with 2'-OMe or PS2 alone [19].
Table 1: Comparative Overview of Core siRNA Modifications
| Modification Type | Chemical Change | Primary Benefits | Key Trade-offs/Considerations |
|---|---|---|---|
| 2'-O-Methyl (2'-OMe) | Methyl group on 2' oxygen | • Nuclease resistance• Reduced immunogenicity• High binding affinity | • Can be poorly tolerated on antisense strand in some contexts [14] |
| 2'-Fluoro (2'-F) | Fluorine atom replaces 2' hydroxyl | • High thermal stability (ΔTm up to +15°C) [14]• Excellent nuclease resistance• Well-tolerated by RISC | • Alters hydration profile• Mechanism is enthalpically driven [14] |
| Phosphorothioate (PS) | Sulfur replaces one non-bridging oxygen | • Nuclease resistance• Enhanced protein binding & pharmacokinetics | • Creates chiral center (complex mixture)• Can reduce potency if overused [18] |
| Phosphorodithioate (PS2) | Sulfur replaces both non-bridging oxygens | • Achiral (simplified production & analysis)• Improved stability & potency vs. PS [20] | • Newer modification, less extensively studied |
Comparative Experimental Data and Performance
The efficacy of siRNA modifications is highly dependent on their position within the duplex, the sequence context, and the biological target. Systematic studies provide critical insights into how these modifications perform head-to-head.
Table 2: Experimental Data from Comparative Modification Studies
| Study Focus / siRNA Construct | Key Experimental Findings | Reference / Model |
|---|---|---|
| 2'-F vs. Unmodified | • Tm increase: ~15°C (86.2°C vs. 71.8°C)• Serum half-life: >24 h vs. <4 h (unmodified)• In vivo potency: ~2-fold higher than unmodified siRNA [14] | Factor VII (FVII) mouse model [14] |
| 2'-F/2'-OMe Alternating Duplex | • Tm increase: 20°C vs. unmodified RNA• In vitro potency: >500-fold increase vs. unmodified [17] | Human phosphatase and tensin homolog mRNA target [17] |
| PS2 vs. PS Stabilization | • Single PS2 provided similar or improved stabilization vs. two PS linkages.• Several PS2 siRNAs showed increased in vivo activity and duration [20] | GalNAc-conjugated siRNA in vivo studies [20] |
| MePS2 (2'-OMe+PS2) vs. counterparts | • 6-fold enhancement in EphA2 silencing vs. unmodified.• 4 to 7.5-fold enhancement vs. PS2 or MePS alone.• Increased RISC loading identified as a key mechanism [19] | Ovarian cancer cell lines & mouse models [19] |
| Tolerance on Antisense Strand | • 2'-F was tolerated on antisense strand.• 2'-O-Me and LNA were not tolerated on antisense strand in vivo [14] | Factor VII (FVII) mouse model [14] |
Essential Experimental Protocols for Evaluation
Protocol for Measuring Serum Stability
Objective: To determine the resistance of a modified siRNA to nuclease degradation in biological fluids.
- Incubation: Suspend the siRNA (e.g., 0.75 μL of a 200 μM solution) in 95% fetal bovine serum (FBS) or human serum (e.g., 14.25 μL). Incubate at 37°C.
- Time-Course Sampling: Remove aliquots at various time points (e.g., 10 min, 1 h, 5 h, 24-48 h).
- Protein Removal: At each time point, stop the reaction and remove nucleases/proteins by extraction with TE-saturated phenol, followed by ethanol precipitation. Use glycogen (e.g., 1 μL of 20 mg/mL) to facilitate quantitative RNA recovery.
- Analysis: Resuspend the purified siRNA and analyze integrity by denaturing or native polyacrylamide gel electrophoresis (PAGE). Stain with ethidium bromide and visualize using a phosphorimager. Intact siRNA appears as a single, distinct band, while degradation products form a smear or lower molecular weight bands.
Protocol for Determining Melting Temperature (Tm)
Objective: To quantify the thermal stability of the modified siRNA duplex.
- Sample Preparation: Combine equimolar amounts of sense and antisense strands in a suitable buffer (e.g., 100 mM NaCl, 10 mM sodium phosphate, pH 7.0, 0.1 mM EDTA). A typical final duplex concentration is 4 μM.
- Thermal Denaturation: Use a UV-spectrophotometer equipped with a temperature controller. Heat the solution at a constant rate (e.g., 0.5°C/min) from a low (15°C) to a high temperature (95°C) while monitoring the absorbance at 260 nm.
- Data Analysis: Plot the absorbance versus temperature (melting curve). The Tm is defined as the temperature at the midpoint of the transition between the double-stranded and single-stranded states, typically determined from the first derivative of the melting curve.
Protocol for Assessing In Vivo Silencing Efficacy
Objective: To evaluate the functional gene silencing capability of a modified siRNA in an animal model.
- Formulation & Dosing: Formulate the siRNA with an appropriate delivery system (e.g., LNP01 liposomes for liver-specific delivery, GalNAc for hepatocyte targeting, or DCA for extrahepatic delivery). Administer a single dose to mice via a relevant route (e.g., intravenous or subcutaneous injection). A common dose is 3-20 mg/kg.
- Tissue Collection: At a predetermined endpoint (e.g., 1-week post-injection), euthanize the animals and collect target tissues (e.g., liver, kidney, muscle). Preserve tissues for RNA analysis (e.g., in RNAlater).
- mRNA Quantification: Homogenize tissue samples. Quantify the levels of the target mRNA and a stable housekeeping gene (e.g., Hprt, Ppib) using a sensitive assay such as the QuantiGene 2.0 Assay or quantitative RT-PCR.
- Data Normalization: Normalize the target mRNA level in each treated sample to the average level in the control (e.g., PBS-injected) group to calculate the percentage of silencing.
The following diagram outlines the logical decision-making process for selecting modifications based on desired molecular properties.
The Scientist's Toolkit: Key Research Reagents & Materials
Table 3: Essential Reagents for siRNA Modification Research
| Reagent / Material | Function in R&D | Specific Application Example |
|---|---|---|
| 2'-F/2'-OMe Phosphoramidites | Building blocks for solid-phase synthesis of modified siRNA strands. | Synthesis of nuclease-resistant sense and antisense strands with enhanced affinity [14] [6]. |
| Phosphorothioate & Phosphorodithioate Reagents | Introduce backbone modifications during synthesis for stability. | DDTT (3-[(dimethylaminomethylene)amino]-3H-1,2,4-dithiazole-5-thione) is used as a sulfurizing agent [18]. |
| DCA-functionalized CPG Support | Solid support for synthesizing lipid-conjugated sense strands. | Enables exploration of extrahepatic delivery for DCA-conjugated siRNAs [18]. |
| Controlled Pore Glass (CPG) with Unylinker | Standard solid support for antisense strand synthesis. | General synthesis of oligonucleotides without a 3' conjugate [18]. |
| Pf1 Phage Solution | Induces weak molecular alignment for NMR structure determination. | Used in measuring residual dipolar couplings (RDCs) for 3D structure elucidation of modified siRNA [17]. |
| QuantiGene 2.0 Assay | Directly quantifies target mRNA levels from tissue lysates without RNA purification. | Assessing in vivo silencing efficacy of modified siRNAs; measures endogenous mRNA [18] [6]. |
| Peptide Nucleic Acid (PNA) Probe | Hybridization-based detection and quantification of intact siRNA in tissues. | PNA hybridization assay to determine tissue concentration of siRNA antisense strand post-injection [18]. |
The empirical optimization of 2'-OMe, 2'-F, phosphorothioate, and newer modifications like phosphorodithioate has been fundamental to the clinical success of siRNA therapeutics. The data clearly show that while unmodified siRNAs can be functional in controlled settings [15], their therapeutic application is severely limited without chemical enhancements. The choice of modification is a balancing act, requiring careful consideration of the trade-offs between stability, potency, specificity, and manufacturability.
Future directions in the field point toward rational design to reduce the heavy reliance on extensive screening. The integration of machine learning and molecular modeling is showing great promise in predicting how modification patterns at specific positions (e.g., in the seed region of the guide strand) influence biological activity [8] [21] [16]. Furthermore, as the field pushes siRNA therapeutics beyond the liver, the interplay between chemical modification, siRNA duplex structure (asymmetric vs. blunt), and conjugate identity will become even more critical to unlock robust silencing in extrahepatic tissues [18] [6].
Small interfering RNA (siRNA) therapeutics represent a breakthrough modality for precision medicine, enabling sequence-specific silencing of disease-causing genes through the RNA interference (RNAi) pathway [22] [2]. However, the inherent physicochemical properties of unmodified siRNA molecules create significant pharmacokinetic barriers that have limited their clinical translation. Unmodified siRNAs are highly soluble macromolecules with strong negative charges, making plasma membrane penetration difficult [23]. They are also vulnerable to rapid degradation by nucleases present in plasma, tissues, and cytoplasm [22] [5]. Furthermore, their relatively small size (approximately 13-15 kDa) and hydrophilic nature result in rapid renal clearance, with unmodified siRNAs exhibiting plasma half-lives as short as 5 minutes to 1 hour following systemic administration [22] [5]. These pharmacokinetic limitations necessitate innovative chemical and formulation strategies to transform siRNA from a biological tool into a viable therapeutic modality.
Key Pharmacokinetic Barriers for Unmodified siRNA
Table 1: Primary Pharmacokinetic Challenges of Unmodified siRNA
| Challenge | Impact on Pharmacokinetics | Consequence |
|---|---|---|
| Nuclease Degradation | Rapid cleavage by serum and tissue nucleases | Half-life of minutes in biological fluids |
| Rapid Renal Clearance | Quick elimination via glomerular filtration | Reduced systemic exposure and target engagement |
| RES Uptake | Opsonization and phagocytosis by mononuclear phagocytic system | Accelerated clearance from circulation |
| Poor Cellular Uptake | Inability to cross hydrophobic cell membranes | Limited intracellular delivery to cytoplasm |
| Endosomal Trapping | Entrapment in endosomal compartments after cellular uptake | <1% of internalized siRNA reaches cytosol [22] |
| Immunostimulation | Recognition by Toll-like receptors and other immune sensors | Off-target inflammatory responses and toxicity |
The journey of siRNA from administration to target site encounters multiple extracellular and intracellular barriers. Systemically administered siRNAs face immediate degradation by ubiquitous ribonucleases in biological fluids, drastically reducing their effective half-life [2]. Their inherent negative charge and hydrophilic nature hinder efficient cellular uptake, preventing them from crossing cell membranes and achieving adequate intracellular concentrations [2] [5]. Even after cellular internalization, the majority of siRNA molecules remain trapped in endosomal compartments, where they are either degraded or recycled back to the surface for extrusion from the cell [22]. Less than 1% of internalized siRNA molecules successfully escape the endosomal compartment to reach their cytoplasmic site of action [22].
Strategic Solutions: Chemical Modifications to Enhance Stability
Chemical modifications represent the foremost strategy for addressing the pharmacokinetic limitations of unmodified siRNA. These modifications primarily target three structural components: the ribose sugar, phosphate backbone, and nucleobases.
Table 2: Key Chemical Modifications and Their Pharmacokinetic Impacts
| Modification Type | Specific Modifications | Pharmacokinetic Benefits | Experimental Evidence |
|---|---|---|---|
| Ribose Sugar (2'-position) | 2'-O-methyl (2'-OMe), 2'-fluoro (2'-F), 2'-O-methoxyethyl (2'-MOE) | Enhanced nuclease resistance, reduced immunogenicity, improved binding affinity | 2'-OMe and 2'-F most common (63% of modifications) [24] |
| Phosphate Backbone | Phosphorothioate (PS) | Increased protein binding, prolonged circulation time, enhanced tissue penetration | PS modifications increase resistance to nucleases [22] [2] |
| Nucleobase | 5-methylcytosine, pseudouridine, 2-thiouridine | Reduced immune recognition, maintained or improved binding affinity | 5-methylcytosine lowers immune activation [2] |
| Terminal/Conjugate | Cholesterol, GalNAc, lipids, PEG | Improved cellular uptake, tissue-specific targeting, reduced renal clearance | GalNAc enables hepatocyte-specific delivery [2] [25] |
The strategic placement of chemical modifications throughout the siRNA structure follows carefully designed patterns to balance stability enhancement with preservation of RNAi activity. Position-specific preferences have been identified—for instance, positions 2, 6, 14, and 16 of the antisense strand and positions 7, 9, and 11 of the sense strand prefer less bulky residues like 2'-F over 2'-OMe [26]. Fully modified siRNAs demonstrate remarkable advantages in medicine development by creating an intracellular depot of siRNA that gets slowly released over time into the cytoplasm for loading into RISC, defining the long-term efficacy of siRNA drugs [6].
Advanced Delivery Platforms for Improved Pharmacokinetics
Beyond chemical modifications, advanced delivery systems play a crucial role in enhancing the pharmacokinetic profile of siRNA therapeutics. The dominant delivery platforms can be categorized into viral vectors, lipid-based systems, polymer-based carriers, and direct conjugates.
Lipid-Based Nanoparticles (LNPs): LNPs represent the most clinically advanced platform, combining ionizable lipids, helper lipids, PEGylated lipids, and cholesterol to form stable nanoparticles that protect siRNA from degradation and enhance cellular uptake [2] [5]. The FDA-approved therapy patisiran (ONPATTRO) utilizes LNP technology for treating hereditary transthyretin-mediated amyloidosis, demonstrating significantly improved pharmacokinetics compared to unformulated siRNA [2].
GalNAc Conjugation: Bioconjugation with N-acetylgalactosamine (GalNAc) has emerged as a breakthrough strategy for hepatocyte-specific siRNA delivery. GalNAc conjugates target the asialoglycoprotein receptor (ASGPR), which is abundantly expressed on hepatocyte surfaces [25]. This approach has led to the development of multiple FDA-approved siRNA therapeutics, including givosiran, lumasiran, and inclisiran, which demonstrate extended duration of action allowing for quarterly or biannual dosing [2] [25].
Additional Delivery Strategies: Polymer-based vectors utilizing materials such as polyethylenimine (PEI), poly-L-lysine (PLL), chitosan, and PAMAM dendrimers have been optimized for enhanced cellular uptake and endosomal escape [2]. Antibody-oligonucleotide conjugates represent an emerging approach for targeted delivery to specific cell types by leveraging the selective binding properties of antibodies to target tumor-specific antigens [25].
Experimental Evidence: Quantitative Comparison of PK Parameters
Substantial experimental data demonstrate the profound impact of chemical modifications and delivery systems on siRNA pharmacokinetics.
Table 3: Experimental Comparison of Modified vs. Unmodified siRNA PK Parameters
| siRNA Format | Half-Life | Clearance Rate | Dosing Interval | Key Modifications/Delivery |
|---|---|---|---|---|
| Unmodified siRNA | 5 min - 1 hr [22] [5] | Rapid renal clearance | Multiple doses daily | None |
| Patisiran (LNP-formulated) | Significantly extended | Reduced RES clearance | Every 3 weeks [22] | LNP encapsulation |
| Inclisiran (GalNAc-conjugated) | Extended tissue residence | Hepatic targeting | Twice yearly [26] | GalNAc conjugate, extensive 2'-modifications |
| Givosiran (GalNAc-conjugated) | Prolonged stability | Reduced renal excretion | Monthly | GalNAc, optimized modification pattern [26] |
In vitro studies systematically evaluating modification patterns reveal that specific 2'-F placements at critical positions significantly enhance siRNA efficacy. Research shows that modifying positions 3, 4, and 5 of the antisense strand and position 17 of the sense strand with 2'-F nucleotides improves target mRNA inhibition while maintaining favorable safety profiles [26]. The optimal modification pattern must balance nuclease resistance with maintained RISC loading and activity—over-modification at certain critical positions can interfere with RISC recognition and decrease gene-silencing efficacy [22].
Research Toolkit: Essential Reagents and Methodologies
Table 4: Essential Research Reagents and Methods for siRNA PK Studies
| Reagent/Method | Function/Application | Key Features |
|---|---|---|
| Phosphorothioate Amidites | Backbone modification | Enhances nuclease resistance and protein binding |
| 2'-F/2'-OMe Amidites | Ribose sugar modification | Improves stability and reduces immunogenicity |
| GalNAc Synthesis Reagents | Liver-targeted conjugation | Enables hepatocyte-specific delivery via ASGPR |
| Ionizable Lipids | LNP formulation | Facilitates endosomal escape and cytoplasmic delivery |
| LC-MS/MS Bioanalysis | PK quantification | Sensitive detection of siRNA and metabolites (LLOQ: 1-20 ng/mL) [25] |
| Stem-loop RT-qPCR | Alternative bioanalysis | High sensitivity for siRNA quantification |
Experimental Protocol for siRNA Stability Assessment:
- Incubation Conditions: Incubate modified and unmodified siRNA variants in human serum (50%) or plasma at 37°C
- Time-point Sampling: Collect aliquots at predetermined intervals (0, 5, 15, 30, 60, 120, 240 min)
- Sample Processing: Extract siRNA using solid-phase extraction or protein precipitation
- Analysis Method: Quantify intact siRNA using LC-MS/MS or specialized electrophoresis
- Data Analysis: Calculate half-life using non-compartmental analysis of concentration-time profiles
Efficacy Testing Workflow:
- In Vitro Screening: Transfect modified siRNAs into relevant cell lines (e.g., Hep3B for liver targets)
- mRNA Quantification: Measure target mRNA levels using qPCR 24-48 hours post-transfection
- Protein Analysis: Assess target protein reduction via Western blot or ELISA 72-96 hours post-transfection
- Dose-Response: Establish IC50 values across concentration gradients
- In Vivo Validation: Administer lead candidates to animal models and measure target engagement in tissues
The strategic implementation of chemical modifications and advanced delivery systems has fundamentally transformed the pharmacokinetic profile of siRNA therapeutics, enabling their successful clinical translation. The evolution from unmodified siRNA with half-lives of minutes to modified conjugates with dosing intervals of months represents a remarkable advancement in oligonucleotide therapeutics. Current clinical data demonstrate that optimized siRNA agents like inclisiran achieve sustained target protein reduction for up to six months following a single subcutaneous dose, a feat impossible with unmodified siRNA [26]. The continued refinement of modification patterns and delivery platforms promises to further enhance the pharmacokinetic properties of siRNA therapeutics, potentially expanding their application to additional tissue targets beyond the liver and enabling treatment of a broader range of diseases. As the field progresses, the integration of computational modeling with experimental data will further accelerate the rational design of siRNA therapeutics with optimal pharmacokinetic and pharmacodynamic properties.
Building a Better siRNA: Strategic Modification Patterns and Delivery Systems for Maximum Effect
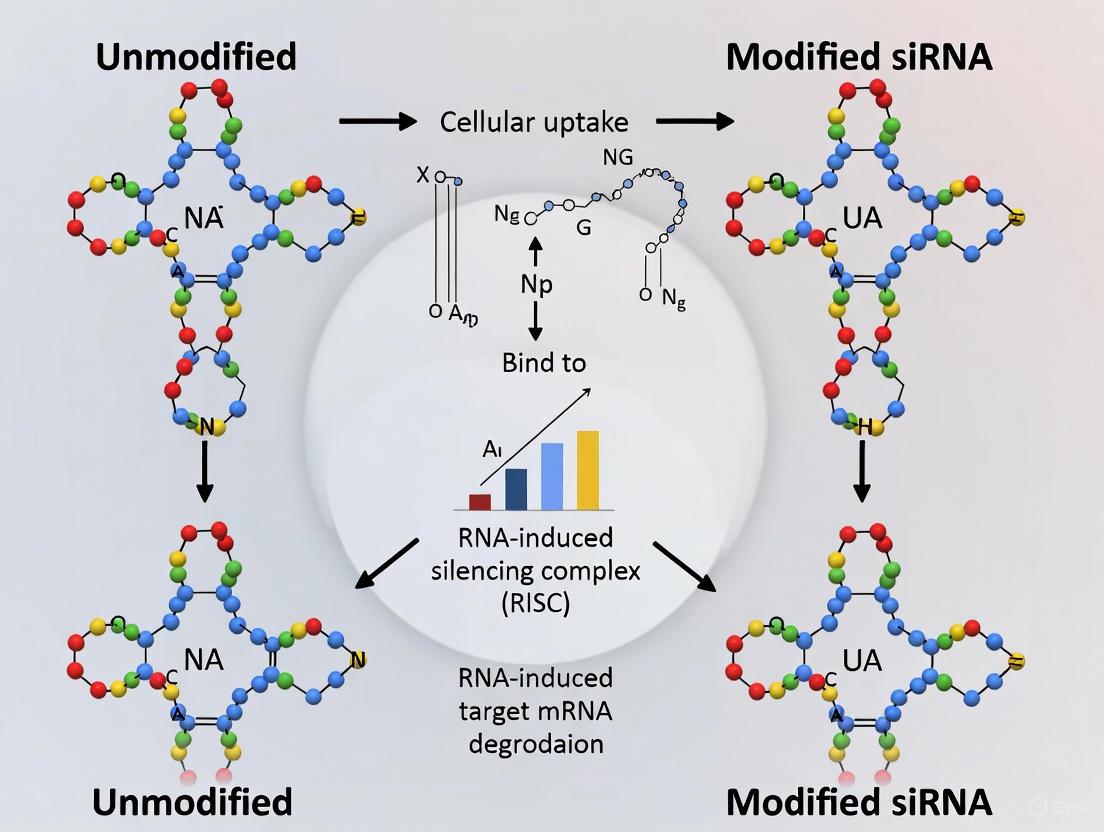
The therapeutic application of small interfering RNA (siRNA) represents a paradigm shift in precision medicine, enabling the targeted silencing of disease-causing genes through mRNA degradation. Since the discovery of RNA interference (RNAi), siRNA has evolved from a research tool to an established drug class, with six FDA-approved therapies currently on the market and hundreds more in development [2] [27]. However, unmodified siRNAs face substantial biochemical challenges that limit their clinical utility, including susceptibility to nuclease degradation, rapid renal clearance, potential immunogenicity, and inefficient cellular uptake [2] [27]. Chemical modifications address these limitations by enhancing nuclease resistance, improving pharmacokinetic profiles, and facilitating delivery, while simultaneously introducing a critical consideration: how these alterations impact the delicate functionality of the RNA-induced silencing complex (RISC).
The RISC machinery, with Argonaute 2 (AGO2) at its catalytic core, is responsible for siRNA-mediated gene silencing. The guide strand of the siRNA duplex must load into RISC, engage with complementary mRNA via Watson-Crick base pairing, and facilitate AGO2-mediated cleavage between positions 10 and 11 relative to the guide strand's 5' end [2]. Chemical modifications that stabilize siRNA against degradation can potentially interfere with these processes by altering duplex thermodynamics, impairing RISC loading, or disrupting the catalytic cleavage mechanism. This guide systematically compares the impact of various chemical modification scaffolds on RISC function, providing researchers with evidence-based insights for designing potent therapeutic siRNAs.
Key Modification Patterns and Their Biochemical Rationale
Common Chemistries and Their Properties
Chemical modifications primarily target three components of the siRNA molecule: the sugar moiety, phosphate backbone, and nucleobases. Each confers distinct biochemical advantages that must be balanced against potential impacts on RISC activity and specificity.
Table 1: Common siRNA Chemical Modifications and Their Primary Effects
| Modification Type | Key Examples | Primary Biochemical Effects | Considerations for RISC Function |
|---|---|---|---|
| Sugar (Ribose) | 2′-O-methyl (2′-OMe), 2′-fluoro (2′-F), 2′-O-methoxyethyl (2′-MOE) | Enhances nuclease resistance, improves binding affinity, reduces immunogenicity | Bulky groups (e.g., 2′-OMe) at critical positions can impair RISC loading and catalytic efficiency |
| Phosphate Backbone | Phosphorothioate (PS) | Increases serum stability, enhances protein binding and tissue distribution | Can slightly reduce silencing potency; strategic placement minimizes impact on RISC |
| Nucleobase | 5-methylcytosine, pseudouridine, 2-thiouridine | Modulates immune recognition, can enhance stability and base-pairing affinity | Generally well-tolerated if base-pairing is preserved; minimal impact on RISC mechanics |
| Conformational Constraint | Locked Nucleic Acid (LNA), Unlocked Nucleic Acid (UNA) | LNA increases binding affinity; UNA introduces flexibility | LNA can over-stabilize duplex; UNA can reduce off-targets by modulating seed region interactions |
The strategic application of these modifications creates "scaffolds" or patterns that determine the overall performance of therapeutic siRNAs. Research indicates that modification patterns are typically regarded as core intellectual property for siRNA therapeutic companies, highlighting their critical importance [26].
Position-Specific Effects on RISC Function
Recent systematic studies have revealed that the positional context of chemical modifications significantly influences their impact on RISC function. A comprehensive analysis of approximately 1,260 differentially modified siRNAs demonstrated that the chemical modification pattern—particularly the level of 2′-O-methyl content—significantly impacts efficacy, while structural features like symmetric versus asymmetric configurations show minimal effects [6]. However, the relationship between modification content and efficacy is not straightforward; specific positions within the siRNA guide strand exhibit distinct sensitivities to modification types.
Table 2: Position-Specific Modification Effects on siRNA Efficacy
| Strand | Position | 2′-F Preference | 2′-OMe Tolerance | Functional Rationale |
|---|---|---|---|---|
| Antisense (Guide) | 2, 6, 14, 16 | Preferred | Tolerated but may reduce activity | Critical for AGO2 binding and catalytic function |
| Antisense (Guide) | 3, 4, 5 | Context-dependent preference | Generally tolerated | Affects guide strand flexibility and target recognition |
| Antisense (Guide) | 9-11 | Not preferred | Well-tolerated | Forms catalytic core; modifications may interfere with cleavage |
| Sense (Passenger) | 7, 9, 11 | Preferred | Tolerated | Affects passenger strand degradation and RISC loading bias |
| Sense (Passenger) | 16, 17 | Position 17 may be preferred | Generally tolerated | Terminal positions less critical for RISC function |
Novel scaffold designs systematically evaluate these positional preferences. For instance, Huang and colleagues designed modification patterns (DV25-29) that strategically placed 2′-F modifications at positions 3, 4, and 5 of the antisense strand and position 17 of the sense strand to enhance efficacy [26]. Their research demonstrated that these optimized scaffolds improved the in vitro activity of a PCSK9-targeting siRNA, ultimately leading to the identification of a clinical candidate (YK1597-DV29PG5) with favorable preclinical profiles.
Experimental Approaches for Evaluating Modification Impact
Methodologies for Assessing RISC Function and Silencing Efficacy
Rigorous evaluation of chemical modification scaffolds requires integrated experimental approaches that measure both biochemical properties and functional outcomes in physiologically relevant systems.
Cell-Based Silencing Assays: Researchers typically employ dual-luciferase reporter systems where the target sequence is inserted into the 3′-UTR of a firefly luciferase gene, enabling precise quantification of silencing efficiency through luminescence measurements [6]. This approach isolates the impact of modifications on RISC-mediated silencing from potential confounding factors related to native mRNA context. For therapeutically relevant assessment, follow-up experiments in native expression systems using techniques like QuantiGene or qPCR are essential, as the native context of the target mRNA—including exon usage, polyadenylation site selection, and ribosomal occupancy—significantly influences siRNA performance [6].
siRNA Library Screening: Large-scale systematic evaluations involve designing siRNA panels targeting multiple therapeutically relevant genes (e.g., APP, BACE1, MAPT, SNCA) with variations in modification patterns and target positions [6]. This approach enables statistical analysis of the relative contributions of modification type, position, and target sequence context to overall silencing efficacy.
Off-Target Effect Assessment: The siRMSD parameter quantifies structural distortion induced by chemical modifications and serves as a predictive metric for off-target effects [28]. Modifications at positions 2–5 that disrupt the A-form RNA duplex on AGO2 prevent stable binding to partially complementary off-target mRNAs, thereby reducing off-target effects, while modifications at positions 6–8 have minimal impact on off-target potential [28].
Experimental Workflow for siRNA Scaffold Evaluation
Key Research Reagents and Solutions
Table 3: Essential Research Reagents for siRNA Modification Studies
| Reagent/Solution | Function in Experimental Protocol | Specific Application Example |
|---|---|---|
| Chemically modified phosphoramidites | Solid-phase oligonucleotide synthesis | 2′-F, 2′-OMe phosphoramidites for incorporating specific modifications [6] |
| GalNAc conjugation reagents | Hepatocyte-targeted delivery | Enables efficient liver delivery for in vivo efficacy studies [26] [2] |
| Phosphorothioate modifiers | Backbone modification | Enhances nuclease resistance and pharmacokinetics [2] |
| Dual-luciferase reporter systems | Reporter-based efficacy screening | Isolates RISC function from native mRNA context effects [6] |
| QuantiGene assay kits | Native mRNA quantification | Measures silencing in physiological expression context [6] |
| Hep3B cell line | In vitro hepatocyte model | Evaluates siRNA efficacy in liver-relevant context [26] |
| Drosophila S2 cells | Insect cell model for structural studies | Assesses impact of length and overhangs on RNAi efficiency [29] |
Comparative Analysis of Modification Scaffolds
Efficacy Across Modification Patterns
Systematic evaluation of modification scaffolds reveals distinct performance characteristics. A comprehensive study comparing different modification patterns across multiple target genes found that while modification pattern significantly impacts efficacy, the optimal scaffold varies depending on the specific target sequence and biological context [6]. For example, the number of effective siRNAs identified per target gene showed substantial variation, highlighting the interplay between modification pattern and target-specific factors.
In a direct comparison of novel modification patterns (DV25-29) applied to a PCSK9-targeting siRNA, researchers observed measurable improvements in in vitro silencing efficacy compared to established modification scaffolds [26]. The optimal pattern (DV29) incorporated specific 2′-F placements at positions 3, 4, and 5 of the antisense strand while omitting 2′-F at position 10 of the sense strand, challenging previous assumptions about modification preferences.
Structural Considerations Beyond Chemical Modifications
While chemical modifications dominate scaffold optimization, physical structure also contributes to siRNA efficacy. Research in Drosophila S2 cells demonstrated that siRNAs with 2-nucleotide overhangs consistently outperform blunt-ended structures, highlighting the importance of terminal geometry for RISC loading [29]. Additionally, siRNA efficacy drastically decreases at lengths below 17 nucleotides but can be restored by extending to 19 base pairs, indicating length thresholds for maintaining RISC compatibility [29].
siRNA Modification Impact on RISC Function
Discussion: Implications for Therapeutic siRNA Design
Integrating Modification Strategies with Target Considerations
The optimal chemical modification scaffold cannot be considered in isolation from target-specific factors. Research demonstrates that siRNA efficacy variability is partially explained by mRNA-specific features including exon usage, polyadenylation site selection, and ribosomal occupancy [6]. Notably, the differences in effective siRNA hit rates between targets are largely mitigated when efficacy is tested in reporter assays rather than native expression contexts, confirming that native mRNA-specific features significantly influence siRNA performance [6].
This interplay between modification pattern and mRNA context necessitates a holistic design approach. While certain modification patterns generally enhance stability and reduce immunogenicity, the optimal positioning of specific modifications (particularly 2′-F versus 2′-OMe) appears sequence-dependent. Consequently, lead identification should incorporate screening of multiple modification scaffolds across therapeutically relevant cellular models rather than relying solely on reporter assays.
Emerging Trends and Future Directions
The field continues to evolve with several emerging trends. First, computational approaches like siDPT leverage transformer-based architectures to predict siRNA efficacy by learning from preference-pair datasets, potentially accelerating the design of optimized modification scaffolds [30]. Second, research continues to elucidate the structural basis of modification effects, with parameters like siRMSD providing quantitative metrics for predicting off-target potential based on structural distortion [28]. Finally, the expansion of siRNA therapeutics beyond hepatocytes to extrahepatic tissues introduces additional design constraints that may necessitate tissue-specific modification optimizations.
As the structural and mechanistic understanding of RISC function deepens, the rational design of chemical modification scaffolds will increasingly incorporate predictive modeling of how specific alterations impact each step of the silencing pathway—from RISC loading and guide strand positioning to target recognition and catalytic cleavage. This progression from empirical optimization to mechanism-based design promises to enhance the efficiency of therapeutic siRNA development and expand the scope of treatable diseases.
Chemical modification scaffolds fundamentally determine the therapeutic potential of siRNA by balancing biochemical stability with RISC functional compatibility. The evidence indicates that modification pattern—particularly the strategic placement of 2′-F and 2′-OMe groups at position-sensitive locations—significantly impacts silencing efficacy, while blunt-ended versus overhang structural configurations show minimal effects. The most effective scaffolds emerge from systematic evaluation that considers both the chemical nature of modifications and their positional context within the siRNA duplex. As the siRNA therapeutic field expands beyond liver targets to address neurological, oncological, and metabolic disorders, the optimization of modification scaffolds for tissue-specific environments and delivery systems will remain a critical frontier in realizing the full potential of RNA interference-based medicine.
In the realm of RNA interference (RNAi) research, the nucleotide sequence of small interfering RNA (siRNA) has long been the primary focus for designing effective gene silencing tools. However, emerging evidence demonstrates that structural characteristics of the siRNA duplex, particularly the distinction between asymmetric and blunt-ended configurations, profoundly influence silencing efficacy, specificity, and therapeutic potential. While sequence selection determines target specificity, structural attributes govern intracellular processing, RISC (RNA-induced silencing complex) loading efficiency, and strand selection bias—all critical determinants of ultimate knockdown efficiency [31] [1].
The investigation into siRNA structure-function relationships represents a crucial dimension in the broader thesis comparing unmodified versus modified siRNA efficiency. As research advances from basic science to therapeutic applications, understanding how structural modifications enhance siRNA performance becomes increasingly vital. This guide provides a comprehensive comparison of asymmetric versus blunt siRNA duplex structures, examining their respective mechanisms, quantitative performance metrics, and appropriate methodological applications for researchers and drug development professionals.
Molecular Mechanisms: Structural Influence on RNAi Pathway
RNAi Pathway and Key Structural Determinants
The RNAi pathway begins with the introduction of double-stranded siRNA into the cytoplasm. The central enzyme Dicer processes these molecules, after which they are loaded into the RISC. The critical structural determinants influencing this process include:
- Terminal overhangs: Natural siRNAs typically have 3' overhangs of 2 nucleotides, which facilitate Dicer recognition and processing [32].
- Thermodynamic stability profile: Asymmetry in the thermodynamic stability across the duplex, particularly lower stability at the 5'-end of the antisense strand, promotes proper strand selection [33] [1].
- Duplex length: Optimal lengths of 20-25 base pairs ensure proper engagement with the RNAi machinery [31] [32].
The following diagram illustrates how different siRNA structures engage with the RNAi pathway:
Strand Selection Mechanics
A pivotal difference between asymmetric and blunt siRNA structures lies in their interaction with the RISC loading complex. The Argonaute 2 (Ago2) protein within RISC exhibits a strong preference for siRNAs with thermodynamic asymmetry, where the antisense (guide) strand has a relatively unstable 5' end compared to the sense (passenger) strand [33] [1]. This asymmetry facilitates the unwinding of the duplex and preferential retention of the guide strand within RISC.
For blunt-ended siRNAs, this natural asymmetry is absent, potentially leading to:
- Improper strand selection: Either strand may be loaded into RISC with approximately equal probability
- Reduced target specificity: Both sense and antisense strands can guide mRNA cleavage
- Increased off-target effects: Non-intended transcripts may be silenced
The following diagram details the critical strand selection mechanism:
Comparative Analysis: Quantitative Efficacy Metrics
Performance Comparison of siRNA Structural Types
The structural configuration of siRNA duplexes significantly impacts their experimental and therapeutic performance. The table below summarizes key comparative metrics between asymmetric and blunt-ended siRNA structures:
| Performance Parameter | Asymmetric siRNA | Blunt-Ended siRNA | Experimental Basis |
|---|---|---|---|
| Gene Silencing Efficacy | High (>80% knockdown) | Variable (40-70% knockdown) | Reporter assays measuring target protein reduction [33] |
| Strand Selection Fidelity | High (>90% guide strand) | Moderate (50-70% guide strand) | Northern blot analysis of RISC-associated strands [1] |
| Off-Target Effects | Low to moderate | Moderate to high | Microarray analysis of transcriptome changes [34] |
| Dicer Processing Efficiency | Enhanced processing | Requires enzymatic processing | In vitro Dicer cleavage assays [32] |
| Therapeutic Applicability | High (multiple approved drugs) | Limited (research focus) | Clinical trial data; FDA-approved formulations [1] [35] |
| Stability in Serum | Moderate (enhanced with modifications) | Moderate to low | HPLC quantification after serum incubation [1] |
| Immunostimulatory Potential | Lower with proper modifications | Potentially higher | Cytokine secretion assays in immune cells [1] |
Structural Determinants of Efficacy
Beyond the simple asymmetric versus blunt dichotomy, several nuanced structural features significantly influence siRNA efficacy:
- Thermodynamic stability asymmetry: The difference in binding stability between the 5' ends of the two strands correlates strongly with silencing efficacy [33]. A stability differential of ≥1 kcal/mol typically predicts efficient strand selection.
- Terminal nucleotide chemistry: 3' overhangs consisting of two deoxythymidine (dT) residues enhance nuclease resistance while maintaining recognition by the RNAi machinery [32].
- Sequence-specific motifs: Presence of specific nucleotide motifs at key positions (e.g., A/U at position 1, G/C at position 19) influence potency regardless of overall structure [33].
Experimental Design and Methodological Approaches
Protocol for Evaluating siRNA Structural Efficacy
To systematically compare asymmetric versus blunt siRNA efficacy, researchers should implement the following standardized protocol:
Stage 1: siRNA Design and Synthesis
- Design phase: Select target sequence (19-21 nt) with appropriate GC content (30-50%)
- Asymmetric construction: Incorporate 3' dTdT overhangs on both strands
- Blunt-ended construction: Synthesize complementary strands without overhangs
- Chemical modification: Introduce appropriate modifications (2'-OMe, 2'-F, PS) at predetermined positions
- Quality control: Purify by HPLC and verify by mass spectrometry
Stage 2: In Vitro Screening
- Cell line selection: Use reporter cell lines (e.g., HeLa, HEK293) with high transfection efficiency
- Transfection: Utilize lipid-based transfection reagents at optimized siRNA concentrations (1-10 nM)
- Control inclusion: Implement positive (known effective siRNA) and negative (scrambled sequence) controls
- Efficacy assessment: Measure mRNA reduction (qRT-PCR) at 24-48h and protein reduction (Western blot) at 48-72h
Stage 3: Specificity Validation
- Microarray analysis: Perform transcriptome-wide profiling to identify off-target effects
- Mutational analysis: Test against mismatched targets to determine specificity thresholds
- Dose-response: Establish IC50 values through serial dilution (0.1-50 nM range)
The experimental workflow for comprehensive siRNA evaluation is illustrated below:
Data Interpretation Guidelines
When analyzing experimental results comparing siRNA structures:
- Efficacy threshold: Consider ≥70% mRNA reduction as the benchmark for highly effective designs
- Strand specificity: Calculate ratio of intended vs. unintended strand activity through mutant reporter assays
- Therapeutic index: Determine the ratio between efficacy (desired target silencing) and toxicity (off-target effects)
- Statistical rigor: Employ appropriate sample sizes (n≥3) and statistical tests (e.g., ANOVA with post-hoc analysis)
Research Reagent Solutions for siRNA Studies
The following table outlines essential research reagents and their applications in siRNA structural studies:
| Reagent Category | Specific Examples | Research Application | Structural Relevance |
|---|---|---|---|
| siRNA Synthesis | 2'-ACE chemistry [34] | Solid-phase siRNA synthesis | Enables precise terminal structure control |
| Delivery Systems | Lipid nanoparticles (LNPs) [31] | In vivo siRNA delivery | Affects structural integrity during delivery |
| Transfection Reagents | Cationic liposomes, Polymers [31] [34] | In vitro cellular delivery | Influence intracellular trafficking |
| Chemical Modifications | 2'-OMe, 2'-F, PS linkages [1] | Enhancing stability and specificity | Modulate duplex thermodynamics |
| Detection Assays | RISC immunoprecipitation [32] | Strand selection analysis | Direct measurement of structural impact |
| Control siRNAs | ON-TARGETplus [34] | Experimental standardization | Validated asymmetric designs |
| Software Tools | BIOPREDsi, ThermoComposition [33] | In silico efficacy prediction | Incorporates structural parameters |
The comparative analysis of asymmetric versus blunt siRNA structures reveals a complex interplay between structural features and functional efficacy. Asymmetric designs with 3' overhangs generally demonstrate superior performance in therapeutic contexts due to enhanced Dicer recognition, preferential RISC loading, and reduced off-target effects. However, blunt-ended configurations retain value in specific research contexts, particularly when combined with strategic chemical modifications that mimic natural asymmetry.
Within the broader thesis of unmodified versus modified siRNA efficiency, structural optimization emerges as a complementary approach to chemical modification. The most effective siRNA therapeutics—exemplified by FDA-approved products—typically incorporate both structural refinement (asymmetric designs with overhangs) and strategic chemical modifications (2'-OMe, 2'-F, PS linkages) [1]. This dual approach maximizes the benefits of enhanced RISC loading efficiency while addressing challenges of stability, immunogenicity, and specificity.
For researchers pursuing siRNA-based studies or therapeutic development, initial focus should be placed on asymmetric designs with appropriate thermodynamic profiles, reserving blunt-ended variants for specialized applications requiring alternative processing pathways. As the field advances, more sophisticated structure-function insights will undoubtedly emerge, further refining our ability to design optimal RNAi triggers for specific experimental and therapeutic applications.
The transformative potential of RNA interference (RNAi) for treating human diseases has been clear since the discovery of the mechanism, offering the ability to silence disease-causing genes with high specificity [36] [5]. However, a billion years of evolutionary defenses designed to keep foreign RNA out of cells presented a monumental delivery challenge that stalled clinical translation for years [36]. Naked, unmodified siRNA is rapidly degraded by serum nucleases, cleared renally, repelled by negatively charged cell membranes, and can stimulate unwanted immune responses [2] [5]. The breakthrough came not from discovering new siRNA sequences, but from developing sophisticated delivery platforms that protect the fragile nucleic acid and escort it to its intracellular site of action. This review objectively compares the two leading delivery technologies—GalNAc conjugates and Lipid Nanoparticles (LNPs)—within the critical context of how chemical modifications enable their success, providing a guide for researchers selecting a platform for therapeutic development.
The Imperative for Chemical Modification of siRNA
Before discussing delivery platforms, it is essential to understand that both GalNAc and LNP strategies depend heavily on extensive chemical modification of the siRNA itself. Unmodified siRNAs are therapeutically impractical due to their rapid degradation and short plasma half-life, which can be as brief as 6 minutes to 1 hour [5].
Key Chemical Modification Strategies
Chemical modifications are employed to address the inherent limitations of native siRNA, with specific alterations to the ribose sugar, phosphate backbone, and nucleobases [2].
Table 1: Common Chemical Modifications in Therapeutic siRNA
| Modification Type | Specific Examples | Primary Function | Impact on Potency & Stability |
|---|---|---|---|
| Sugar (Ribose) Modification | 2′-O-methyl (2′-OMe), 2′-fluoro (2′-F), 2′-O-methoxyethyl (2′-MOE) | Enhances nuclease resistance, improves binding affinity, reduces immunogenicity [2] | Crucial for metabolic stability; some patterns (e.g., high 2′-OMe content) can significantly impact observed efficacy [6]. |
| Phosphate Backbone Modification | Phosphorothioate (PS) | Increases resistance to nuclease degradation, improves pharmacokinetics [2] | Enhances serum half-life and tissue distribution. |
| Terminal & Conjugate Modification | Cholesterol, N-acetylgalactosamine (GalNAc) | Facilitates improved cell membrane interaction and enables targeted delivery to specific tissues/cells [2] | Directly enables ligand-conjugate delivery strategies like GalNAc-siRNA. |
| Advanced Conformational Modification | Locked Nucleic Acid (LNA) | Confers very high binding affinity and target specificity [2] | Enhances potency but must be balanced with potential for increased synthesis complexity and cost. |
The selection and pattern of these modifications are critical. A systematic study of ~1260 differentially modified siRNAs demonstrated that the modification pattern (e.g., level of 2′-O-methyl content) significantly impacts efficacy, while structural features like symmetric versus asymmetric duplex configurations have a less pronounced effect [6]. Furthermore, full chemical modification is absolutely required for the long-term efficacy of conjugate-based delivery, as it stabilizes the siRNA in the harsh endosomal environment following cellular uptake, effectively creating an intracellular depot that is slowly released into the cytoplasm [6].
Platform Deep Dive: GalNAc-siRNA Conjugates
Mechanism of Action
The N-acetylgalactosamine (GalNAc) conjugate platform is a masterpiece of targeted delivery. It leverages a trivalent GalNAc ligand that binds with high affinity and selectivity to the asialoglycoprotein receptor (ASGPR) on the surface of hepatocytes [36] [37]. ASGPR is an ideal target for liver delivery due to its high expression on hepatocytes (approximately 500,000 receptors per cell), rapid recycling time (every ~15 minutes), and location on the sinusoidal surface [36] [38].
The mechanism is illustrated in the following pathway diagram, which synthesizes information from multiple sources [39] [36] [37]:
Following subcutaneous administration, the conjugate enters the systemic circulation, binds to ASGPR, and is rapidly internalized via clathrin-mediated endocytosis [36] [38]. The acidic environment of the endosome causes dissociation, and the receptor recycles to the membrane while the siRNA is released into the cytosol—a step that remains a key rate-limiting factor, with less than 1% of internalized siRNA successfully escaping the endosome [38]. Once in the cytoplasm, the siRNA guide strand loads into RISC to mediate mRNA cleavage.
Key Experimental Findings and Structure-Activity Relationship (SAR)
A detailed in vivo SAR study of GalNAc ligand components revealed several critical design insights [38]. While traditional in vitro binding affinity follows a hierarchy of tetra- > tri- > bi- > monovalent ligands, in vivo silencing efficacy tells a different story. After a single 2 mg/kg subcutaneous dose in mice:
- Monovalent ligands showed minimal activity (17% TTR knockdown at day 7).
- Bivalent, trivalent, and tetravalent ligands all achieved strong and comparable levels of target protein knockdown (85%-93% at day 7) [38].
This indicates that the dramatic increases in binding affinity do not directly translate to proportional gains in therapeutic efficacy past a bivalent design. The study also identified that the chemical identity of the linker connecting the ligand to the siRNA significantly impacts potency and duration of effect, with optimal linkers enabling up to 94% knockdown at day 7 and 85% at day 14 [38].
Platform Deep Dive: Lipid Nanoparticles (LNPs)
Composition and Mechanism of Action
Lipid Nanoparticles represent a supramolecular approach to siRNA delivery. Modern LNPs are highly tunable, self-assembling systems typically composed of four lipid components that form 60-100 nm vesicles capable of encapsulating >90% of an siRNA payload [39].
Table 2: Core Components of siRNA-LNPs and Their Functions
| LNP Component | Example Molecules | Primary Function | Key Design Considerations |
|---|---|---|---|
| Ionizable Cationic Lipid | DLin-MC3-DMA, L319 | Serves as a "pH switch"; neutral in bloodstream, positively charged in endosomes to enable endosomal escape via the proton sponge effect. The key determinant of potency [39]. | pKa must be optimized (~6.5); next-generation lipids incorporate biodegradable motifs (e.g., esters) for improved safety [39]. |
| Helper Phospholipid | DSPC, DOPE | Provides structural integrity to the particle bilayer and can tune membrane fluidity [39]. | DOPE is often preferred for its tendency to promote hexagonal phase structures that facilitate membrane fusion. |
| Cholesterol | Cholesterol | Acts as a "molecular rivet" that stabilizes the bilayer structure, reduces passive leakage, and enhances cellular uptake [39]. | Constitutes a large fraction (up to 40 mol%) of the formulation. |
| PEGylated Lipid | DMG-PEG2000 | Provides a steric shield to prevent nanoparticle aggregation, opsonization, and rapid clearance by immune cells; modulates particle stability and pharmacokinetics [39]. | PEG length and concentration critically impact pharmacokinetics; it must desorb at the right time to allow cellular uptake. |
The mechanism of LNP-mediated delivery, particularly the critical step of endosomal escape, is distinct from the conjugate approach and can be visualized as follows [39] [5]:
After intravenous infusion, LNPs acquire apolipoprotein E (ApoE) in the serum, which directs them to hepatocytes via the LDL receptor [39]. The particle is endocytosed, and as the endosome matures and acidifies, the ionizable lipids become protonated. This triggers a complex process—potentially involving the formation of inverse hexagonal lipid phases—that destabilizes the endosomal membrane, leading to the release of the siRNA payload into the cytosol [39].
Direct Comparison: Performance Data and Clinical Translation
Quantitative Performance Comparison
The two platforms have distinct strengths and limitations, which are reflected in their pharmacological profiles and clinical use cases.
Table 3: Head-to-Head Comparison of GalNAc Conjugates vs. LNPs
| Parameter | GalNAc-siRNA Conjugates | Lipid Nanoparticles (LNPs) |
|---|---|---|
| Structure & Scale | Molecular conjugate (~14 kDa); minimalistic design [39]. | Supramolecular assembly (~5,000x larger than siRNA); complex, modular design [39] [36]. |
| Administration Route | Subcutaneous injection [39] [38]. | Intravenous infusion (requiring slow infusion and pre-medication) [39]. |
| Primary Targeting | Hepatocytes (via high-affinity ASGPR binding) [36] [37]. | Liver hepatocytes (via ApoE/LDLR mechanism); can be re-targeted to extra-hepatic tissues with ligand engineering [39]. |
| Key Strength | Exceptional potency and durability in hepatocytes; convenient subcutaneous dosing; favorable safety profile [39] [38]. | Proven platform for robust hepatic and extra-hepatic delivery; highly efficient endosomal escape mechanism [39]. |
| Key Limitation | Lacks intrinsic endosomal release mechanism (<1% escape rate); primarily restricted to hepatocytes [38]. | Acute infusion-related toxicities (e.g., CARPA); complex manufacturing and cold-chain storage requirements [39]. |
| Clinical Proof-of-Concept | Inclisiran: ~50% LDL-C reduction with biannual dosing [39]. Givosiran, Lumasiran, Vutrisiran: approved for rare liver disorders [2] [37]. | Patisiran: ~84% reduction in serum TTR with bi-weekly IV infusions [2] [39]. |
Experimental Protocols for Platform Evaluation
For researchers aiming to evaluate these platforms, standard experimental protocols have been established.
- In Vivo Efficacy (GalNAc Conjugates): A standard protocol involves a single subcutaneous injection of the conjugate (e.g., at 2 mg/kg) in C57BL/6 mice (n=4 per group). Plasma levels of the target protein (e.g., Transthyretin) are measured at days 7, 14, and sometimes up to 42 days post-injection to assess both potency and duration of effect [38]. Knockdown is calculated as percent reduction compared to control groups.
- In Vivo Efficacy (LNPs): siRNA-LNPs are typically administered via intravenous injection. The dose can vary but is often in the range of 0.3 to 1 mg/kg. Target mRNA reduction in the liver or other target tissues is quantified using qRT-PCR, and protein levels are assessed via ELISA or Western Blot at defined timepoints [2] [39].
- Addressing the Endosomal Escape Bottleneck: A novel experimental approach for enhancing GalNAc conjugate efficacy is the co-administration of a GalNAc-functionalized endosomal release agent. This involves subcutaneously injecting a pH-responsive polymer micelle alongside the GalNAc-siRNA conjugate. This strategy has been shown in non-human primates to markedly improve the onset, potency, and duration of gene silencing without compromising tolerability, directly addressing the primary limitation of the conjugate platform [38].
The Scientist's Toolkit: Essential Reagent Solutions
The development and evaluation of these delivery platforms require a specific set of research reagents and materials.
Table 4: Key Research Reagents and Materials for siRNA Delivery Platform R&D
| Reagent / Material | Function / Application | Key Considerations |
|---|---|---|
| Ionizable Cationic Lipids (e.g., MC3, L319) | Core component of LNPs; critical for encapsulation efficiency and endosomal escape [39]. | pKa optimization is critical; next-generation biodegradable lipids (e.g., with ester bonds) can improve toxicological profiles. |
| Phosphoramidite Reagents (2′-F, 2′-OMe) | Chemical synthesis of modified siRNA strands; essential for enhancing stability and reducing immunogenicity [2] [6]. | Dissolved in anhydrous acetonitrile; BTT (5-(Benzylthio)-1H-tetrazole) is a common activator [6]. |
| Trivalent GalNAc Ligand | Targeting moiety for hepatocyte-specific delivery in conjugate platforms [36] [37]. | Can be synthesized from D-(+)-galactosamine and trans-4-hydroxy-L-proline methyl ester; attached to the 3' end of the siRNA sense strand [37]. |
| Controlled Pore Glass (CPG) Solid Support | Solid-phase synthesis of oligonucleotides, including GalNAc-conjugated siRNA [38]. | Ligand size impacts loading capacity (e.g., 0.4 mmol/g for bivalent vs. 0.2 mmol/g for tetravalent), affecting final product yield [38]. |
| PEG-Lipids (e.g., DMG-PEG2000) | Component of LNPs that controls particle stability, prevents aggregation, and modulates pharmacokinetics [39]. | PEG desorption kinetics must be optimized; too fast causes immune recognition, too slow inhibits hepatocyte uptake. |
The synergy between chemically modified siRNA and advanced delivery platforms has finally unlocked the clinical potential of RNAi therapeutics. The choice between GalNAc conjugates and LNPs is not a matter of one being superior to the other, but rather a strategic decision based on the therapeutic target. For hepatocyte-specific diseases, the minimalistic design, subcutaneous administration, and outstanding durability of GalNAc conjugates make them the platform of choice. For applications requiring extra-hepatic delivery or where the highest possible payload efficiency is needed, LNPs remain the most clinically advanced and versatile option.
The future of siRNA delivery lies in overcoming the remaining bottlenecks, particularly endosomal escape for conjugates and targeted extra-hepatic delivery for LNPs. Emerging strategies, such as co-administered endosomolytic agents for GalNAc-siRNAs and ligand-functionalized, targetable LNPs, are already paving the way for the next generation of RNAi therapeutics, ensuring that delivery will continue to evolve from a constraining challenge into a precisely tunable design variable [39] [38].
The approval of the first small interfering RNA (siRNA) therapeutic, patisiran, in 2018 marked the arrival of RNA interference (RNAi) as a validated clinical modality for treating genetically defined diseases [2]. siRNA therapeutics function by harnessing the natural cellular process of RNAi to mediate sequence-specific gene silencing via the degradation of messenger RNA (mRNA), thereby preventing the synthesis of disease-causing proteins [2] [40]. This mechanism represents a revolutionary approach that operates upstream of traditional medicines, targeting the genetic precursors of diseases [41]. The profound scientific significance of RNAi was recognized with the award of the 2006 Nobel Prize in Physiology or Medicine [2] [41].
The transition from basic science to clinical application required overcoming substantial hurdles, primarily the inherent instability of unmodified siRNA in biological fluids and the challenges of delivering these large, negatively charged molecules to target cells [2] [23]. Chemical modifications and advanced delivery systems have been the twin pillars enabling this transition, transforming siRNA from a research tool into a promising therapeutic class [2] [42]. This guide provides a systematic comparison of clinically approved siRNA drugs, with a focused analysis on the chemical modification profiles that underpin their efficacy and stability, offering researchers a framework for understanding structure-activity relationships in therapeutic siRNA design.
The RNAi Mechanism and the Imperative for Chemical Modification
The Endogenous RNA Interference Pathway
Therapeutic siRNAs are synthetic double-stranded RNA duplexes, typically 21-23 nucleotides in length, designed to harness the conserved biological pathway of RNAi [2] [40]. The mechanism can be summarized in a series of key steps, illustrated in the diagram below.
Challenges of Unmodified siRNAs and Rationale for Modification
Unmodified siRNAs face critical pharmacological challenges that preclude their clinical use. They are highly susceptible to rapid degradation by ubiquitous nucleases in biological fluids, drastically reducing their half-life [2]. Their inherent negative charge and hydrophilic nature prevent efficient cellular uptake, while unmodified molecules also carry an increased risk of stimulating undesired innate immune responses [2]. Chemical modifications directly address these limitations by:
- Enhancing Nuclease Resistance: Modifications like 2'-O-methyl (2'-OMe) and 2'-fluoro (2'-F) groups shield the siRNA backbone from enzymatic degradation [2].
- Improving Pharmacokinetics: Alterations such as phosphorothioate (PS) linkages reduce renal clearance and improve tissue distribution [2].
- Reducing Immunogenicity: Specific nucleobase modifications (e.g., 5-methylcytosine, pseudouridine) minimize recognition by immune sensors [2].
- Fine-tuning Binding Affinity and Specificity: Conformational constraints (e.g., Locked Nucleic Acids) can enhance target engagement and reduce off-target effects [2] [8].
Comparative Analysis of Clinically Approved siRNA Therapeutics
Approved siRNA Drugs: Indications, Targets, and Delivery Technologies
The following table summarizes the key characteristics of siRNA therapeutics that have received regulatory approval, illustrating the progression of this drug class. The table includes drugs approved up to early 2025, demonstrating the rapid expansion of this field.
Table 1: Clinically Approved siRNA Therapeutics (2018-2025)
| Drug Name (Brand) | Approval Year | Target Gene | Primary Indication | Delivery Technology |
|---|---|---|---|---|
| Patisiran (Onpattro) | 2018 [2] | TTR (Transthyretin) [2] | Hereditary transthyretin-mediated amyloidosis (hATTR) [2] | Lipid Nanoparticles (LNPs) [2] |
| Givosiran (Givlaari) | 2019 [2] [43] | ALAS1 [43] | Acute hepatic porphyria (AHP) [2] | GalNAc-siRNA conjugate [2] |
| Lumasiran (Oxlumo) | 2020 [2] | HAO1 [43] | Primary hyperoxaluria type 1 (PH1) [2] | GalNAc-siRNA conjugate [2] |
| Inclisiran (Leqvio) | 2021 [2] [42] | PCSK9 [43] | Hypercholesterolemia [2] | GalNAc-siRNA conjugate [2] |
| Vutrisiran (Amvuttra) | 2022 [2] | TTR (Transthyretin) [2] | Polyneuropathy of hATTR amyloidosis [2] | GalNAc-siRNA conjugate [2] |
| Nedosiran (Rivfloza) | 2023 [2] | LDHA [40] | Primary hyperoxaluria type 1 (PH1) [2] | GalNAc-siRNA conjugate (GalXC) [40] |
| Fitusiran (Qfitlia) | 2025 [41] | SERPINC1 (Antithrombin) [44] | Hemophilia A or B [41] | Subcutaneous (GalNAc-conjugated) [44] [41] |
The trajectory of approvals shows a clear technological shift from the first-generation lipid nanoparticles (LNPs) used in Patisiran to the subsequent dominance of GalNAc (N-acetylgalactosamine) conjugation for liver-targeted delivery [2] [45]. GalNAc conjugates leverage high-affinity binding to the asialoglycoprotein receptor abundantly expressed on hepatocytes, enabling efficient siRNA uptake with subcutaneous administration and a superior safety profile [2] [43]. The recent approval of Fitusiran in March 2025 for hemophilia further solidifies the clinical validation of the siRNA platform [41].
Chemical Modification Profiles of Approved siRNAs
A detailed examination of the specific chemical modifications reveals common patterns and strategies employed to confer drug-like properties. The modifications are categorized based on their location within the siRNA structure.
Table 2: Chemical Modification Profiles of Approved siRNA Therapeutics
| Drug Name | Sugar Modifications | Phosphate Backbone Modifications | Terminal/Conjugate Modifications | Key Functional Impact |
|---|---|---|---|---|
| Patisiran [2] | 2'-OMe, 2'-F | Phosphorothioate (PS) linkages | LNP formulation (ionizable lipid, PEG-lipid, cholesterol, phospholipid) [2] | Enhanced nuclease resistance, improved hepatocyte delivery via endosomal escape |
| Givosiran [2] [6] | 2'-OMe, 2'-F | Phosphorothioate (PS) linkages | GalNAc ligand conjugated to 3' end of passenger strand [2] | Hepatocyte-specific targeting, enhanced stability for subcutaneous dosing |
| Inclisiran [6] | High 2'-OMe content | Phosphorothioate (PS) linkages | GalNAc conjugate [2] | Optimized for long-term depot effect and sustained PCSK9 silencing (>6 months) |
| Fitusiran [44] | Extensive 2' modifications (2'-OMe/2'-F) | Stabilizing backbone linkages | GalNAc conjugate [44] | Enables monthly subcutaneous dosing for chronic management of hemophilia |
Dominant Modification Patterns:
- Sugar Modifications: 2'-O-methyl (2'-OMe) and 2'-fluoro (2'-F) are the most frequently employed ribose modifications across all approved siRNAs. They are strategically placed to enhance binding affinity, confer nuclease resistance, and reduce immune stimulation without significantly compromising RISC loading and activity [2] [6].
- Phosphate Backbone Modifications: Phosphorothioate (PS) linkages, where a non-bridging oxygen atom is replaced with sulfur, are a nearly universal feature. PS modifications increase stability against nucleases and improve pharmacokinetics by promoting binding to serum proteins, thereby reducing renal clearance [2] [43].
- Asymmetric Design: A common structural design involves heavily modifying the passenger strand to promote its degradation and RISC unloading, while the guide strand is more selectively modified to preserve its gene-silencing activity [2].
Experimental Insights: Connecting Modification Patterns to Efficacy
Systematic Analysis of Modification Impact
A 2025 systematic study analyzed ~1260 differentially modified siRNAs targeting therapeutically relevant mRNAs (APP, BACE1, MAPT, SNCA) to define the relative contributions of siRNA sequence, modification pattern, and structure to observed efficacy [6]. The workflow and key findings of this large-scale screening are summarized below.
Key Experimental Findings:
- Modification Pattern Over Structure: The level of 2'-OMe content significantly impacted silencing efficacy, whereas structural variations (e.g., symmetric versus asymmetric duplex configurations) showed minimal effects [6]. This underscores that chemical nature is more critical than gross structural design in these contexts.
- The Native mRNA Context is Crucial: The study found substantial differences in the number of effective siRNAs identified per target. These target-specific "hit rates" were largely mitigated when efficacy was tested using a reporter assay, confirming that native mRNA-specific features—such as exon usage, polyadenylation site selection, and ribosomal occupancy—are major determinants of siRNA performance [6]. This highlights a critical limitation of simplified screening systems.
Molecular Modeling for Rational siRNA Design
To move beyond empirical screening, computational approaches are being refined. Molecular modeling of individual sequence positions in chemically modified siRNAs has provided atomistic insights [8]. For instance, modeling revealed low stabilization energies and increased sugar flexibility at specific guide strand positions (e.g., g2) when modified with 2'-F, properties that correlated with higher biological activity [8]. This suggests that rational design of modifications should consider site-specific structural dynamics, particularly in the seed region critical for mRNA target recognition.
The Scientist's Toolkit: Essential Reagents and Technologies
The development and evaluation of modified siRNAs rely on a suite of specialized reagents and technologies. The following table catalogues key solutions used in the field, as derived from the experimental methodologies in the cited literature.
Table 3: Essential Research Reagent Solutions for siRNA Therapeutics
| Research Reagent / Technology | Function & Application | Key Characteristics |
|---|---|---|
| Phosphoramidite Solid-Phase Synthesizer (e.g., Dr. Oligo) [6] | Automated chemical synthesis of modified siRNA strands. | Enables incorporation of 2'-F, 2'-OMe, and other modified phosphoramidites at specific positions. |
| Modified Phosphoramidites (2'-F, 2'-OMe, 2'-MOE) [2] [6] | Building blocks for introducing sugar modifications during synthesis. | Improve nuclease resistance, binding affinity, and reduce immunogenicity. |
| Phosphorothioate (PS) Reagents [2] | Introduces phosphorothioate backbone linkages during synthesis. | Replaces a non-bridging oxygen with sulfur; enhances nuclease stability and pharmacokinetics. |
| Ionizable Lipids (e.g., DLin-MC3-DMA) [2] [45] | Critical component of LNP delivery systems. | Positively charged at low pH to complex siRNA, neutral at physiological pH to reduce toxicity; enables endosomal escape. |
| GalNAc Phosphoramidite [2] | Conjugation reagent for synthesizing GalNAc-siRNA conjugates. | Enables covalent attachment of N-acetylgalactosamine ligand for hepatocyte-specific targeting. |
| Cationic Polymers (e.g., PEI, PLL, Chitosan) [2] [45] | Non-viral vectors for siRNA complexation and delivery. | Electrostatic binding to siRNA; enhances cellular uptake and can facilitate endosomal escape. |
| Dual-Luciferase Reporter Assay Systems [6] | High-throughput screening of siRNA efficacy and specificity. | Allows rapid, cost-effective initial screening, though may not fully recapitulate native mRNA context. |
The clinical success of siRNA therapeutics is intrinsically linked to the strategic application of chemical modifications. The comparative analysis presented here demonstrates a clear evolution from complex nanoparticle formulations (Patisiran) to simpler, targeted conjugate technologies (GalNAc-siRNAs) that dominate the later approvals. The consistent use of 2'-OMe/2'-F sugar modifications and phosphorothioate linkages across all drugs highlights their foundational role in achieving metabolic stability and favorable pharmacokinetics.
Future directions in the field will focus on overcoming the remaining delivery barriers to extrahepatic tissues, further refining modification patterns to maximize potency and duration of effect while minimizing off-target interactions, and integrating artificial intelligence and molecular modeling to transition from empirical screening to truly rational siRNA design [42] [8] [45]. As the chemical rulebook for therapeutic siRNA continues to be written, the approved drugs profiled in this guide serve as critical case studies, providing a blueprint for the next generation of RNAi therapeutics aimed at previously undruggable targets.
From Bench to Bedside: Troubleshooting Poor Silencing and Optimizing siRNA Performance
The efficacy of small interfering RNA (siRNA) is determined not only by its own sequence and chemical composition but also by the complex structural and contextual features of its target messenger RNA (mRNA). While siRNA design has traditionally focused on guide strand specificity, emerging research reveals that mRNA structural features and splicing dynamics significantly modulate silencing efficiency. This guide examines how exon usage, untranslated regions (UTRs), and mRNA secondary structure influence siRNA performance, providing a comparative analysis of unmodified versus chemically modified siRNA strategies to help researchers optimize therapeutic RNA interference (RNAi) platforms.
Target mRNA Features Impacting siRNA Efficacy
Exon Usage and Splicing Dynamics
The position of an siRNA target sequence relative to exon-exon junctions can substantially influence its accessibility and effectiveness. A systematic analysis of siRNA efficacy against therapeutically relevant mRNAs (APP, BACE1, MAPT, and SNCA) revealed that target-specific differences in hit rates were largely mitigated when efficacy was tested using reporter assays instead of native mRNA contexts [6]. This suggests that native mRNA architecture, including splicing patterns, profoundly affects siRNA accessibility. Furthermore, RNA secondary structures can influence splice site recognition and exon definition by obscuring or exposing regulatory elements to the splicing machinery [46]. When designing siRNAs, considering whether the target sequence resides in constitutively versus alternatively spliced exons is crucial, as structural constraints may vary significantly between these contexts.
Untranslated Regions (UTRs) and Regulatory Elements
UTRs serve as critical platforms for post-transcriptional regulation, housing elements that significantly impact siRNA efficacy. Research demonstrates that RNA secondary structures in 5' and 3' UTRs can either facilitate or hinder siRNA binding through several mechanisms:
5' UTR Structures: Complex secondary structures in 5' UTRs with high GC content and negative folding free energy (ΔG) can impede ribosome scanning during translation initiation [47]. These same structural features may similarly obstruct siRNA-RISC complex access to target sites, particularly for sequences near the translation start site.
3' UTR Structures: Adenosine-to-inosine (A-to-I) RNA editing in 3' UTRs can stabilize RNA secondary structures, reducing the accessibility of microRNA (miRNA) target sites to AGO2-miRNA complexes [48]. This mechanism likely extends to siRNA targeting, where structured 3' UTR regions may shield target sequences from RISC-mediated recognition and cleavage.
Polyadenylation Signals: siRNA efficacy can be influenced by proximity to polyadenylation site sequences, as these regions often harbor specific secondary structures and protein binding partners that may occlude siRNA access [6].
mRNA Secondary Structure
RNA molecules possess a natural tendency to form stable secondary and tertiary structures through Watson-Crick base pairing [46]. These structural elements significantly impact siRNA efficacy through several mechanisms:
Target Site Occlusion: Stable secondary structures can physically block siRNA access to complementary target sequences. Statistical analyses of mRNA coding sequences have revealed that calculated mRNA folding is often more stable than expected by chance, suggesting codon bias may favor mRNA structures that could potentially impact siRNA targeting [46].
Distance Modulation: RNA secondary structures that do not directly involve conserved splicing sequences can nonetheless vary the relative distance between regulatory elements, determining considerable variation in site accessibility [46].
Dynamic Structural Changes: RNA-binding proteins (RBPs) can remodel mRNA structures in vivo. For example, binding of U2AF65 to the 3' splice site compacts RNA to bring the 3' splice site and branch site region into close proximity [46]. Similar protein-mediated structural rearrangements may transiently expose or conceal siRNA target sites.
Table 1: Impact of mRNA Architectural Features on siRNA Efficacy
| mRNA Feature | Impact Mechanism | Effect on siRNA Efficacy |
|---|---|---|
| Exon Location | Proximity to splice junctions affects RISC accessibility | Variable; context-dependent |
| 5' UTR Structures | High GC content creates stable secondary structures | Generally reduces efficacy |
| 3' UTR Structures | A-to-I editing stabilizes structures, occluding targets | Reduces accessibility to RISC |
| Branch Points | Structural constraints may keep regions single-stranded | Can enhance accessibility |
| PolyA Signals | Protein binding and structural elements near sites | May block or enhance depending on context |
Comparative Analysis: Unmodified vs. Chemically Modified siRNAs
Chemical Modification Patterns
Chemical modifications, particularly 2′-O-methyl (2′-OMe) and 2′-fluoro (2′-F) ribose modifications, significantly alter siRNA performance relative to unmodified counterparts. A comprehensive study evaluating ∼1260 differentially modified siRNAs demonstrated that the modification pattern substantially impacts efficacy, while structural features like symmetric versus asymmetric configurations show minimal effects [6]. Specifically, the level of 2′-O-methyl content significantly influences silencing efficiency, with certain modification patterns optimizing metabolic stability without compromising RISC loading and activity.
Thermodynamic Stability and Seed Region Flexibility
Molecular modeling of unmodified versus chemically modified siRNA duplexes reveals that specific positions are particularly sensitive to modification effects. Studies show low stabilization energies and increased sugar stereochemical flexibility for 2′-F modified position g2 and, to a lesser extent, g6 in the guide strand seed region [8]. These properties correlate with higher observed biological activity, suggesting that strategic modification at these positions can enhance siRNA potency while maintaining full chemical modification for stability.
Target Engagement in Native Contexts
A critical distinction between unmodified and chemically modified siRNAs emerges in their ability to engage targets within native mRNA contexts. Research demonstrates substantial differences in the number of effective siRNAs identified per target when tested in native expression assays versus reporter systems [6]. These target-specific differences are largely mitigated in reporter assays, confirming that native mRNA-specific features significantly influence siRNA performance. Chemically modified siRNAs appear better equipped to overcome these contextual barriers, possibly through enhanced stability and prolonged intracellular presence that allows engagement with transiently accessible target sites.
Table 2: Unmodified vs. Chemically Modified siRNA Performance
| Parameter | Unmodified siRNA | Chemically Modified siRNA |
|---|---|---|
| Nuclease Resistance | Low | High (2'-OMe/2'-F modifications) |
| Off-Target Effects | Higher potential | Reduced through optimized design |
| Duration of Effect | Short-lived | Extended intracellular depot |
| Native Context Efficacy | Highly variable | More consistent across targets |
| RISC Loading Efficiency | Sequence-dependent | Can be optimized via modification patterns |
| Thermodynamic Stability | Variable | Tunable via modification schemes |
Experimental Protocols and Methodologies
siRNA Design and Screening Workflow
Systematic siRNA evaluation requires a structured approach to account for mRNA contextual factors:
Target Sequence Selection: Identify all possible 20-nucleotide targeting sequences across the gene of interest, excluding sequences with ≥60% G-C content (known to negatively impact silencing) and repetitive stretches (CCCC or GGGG) due to synthetic limitations [6].
Specificity Filtering: Remove siRNAs with positions 2–17 of the guide strand showing homology to other human genes to minimize off-target effects while noting this doesn't eliminate seed sequence-mediated off-targeting [6].
Representative Sampling: Select sequences spanning different mRNA regions (ORF and UTRs), including those containing polyadenylation site sequences "AAUAAA" [6].
Multi-Assay Validation: Test siRNAs in both reporter constructs (for isolated target site evaluation) and native expression assays (to account for mRNA architectural features) [6].
Evaluating Secondary Structure Interactions
To assess how RNA secondary structures impact siRNA efficacy:
Structural Prediction: Utilize RNA folding algorithms to predict secondary structures around target sites, noting regions with high stability [46].
Accessibility Profiling: Correlate siRNA efficacy with predicted target site accessibility, considering that structured regions typically show reduced silencing [46] [48].
Compensatory Mutagenesis: Experimentally validate structural impacts by introducing mutations that alter predicted structures while maintaining coding sequence, then re-testing siRNA efficacy [47].
Assessing Exon Usage Effects
Protocol for evaluating how splicing dynamics influence siRNA targeting:
Isoform Characterization: Determine which exons are constitutively versus alternatively spliced in the target cell line or tissue [6].
Region-Stratified Design: Design siRNAs targeting different exon contexts (constitutive, alternative, junction-spanning).
Efficacy Correlation: Measure silencing efficiency relative to exon inclusion rates and junction proximity.
Experimental Factors in siRNA Efficacy
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for siRNA-mRNA Interaction Studies
| Reagent/Category | Function/Application | Example Implementation |
|---|---|---|
| Chemically Modified Oligonucleotides | Enhance nuclease resistance and reduce innate immune activation | 2'-O-methyl (2'-OMe) or 2'-fluoro (2'-F) modifications [6] |
| Lipid Nanoparticles (LNPs) | Delivery vehicles for RNA therapeutics | Composed of different helper lipids (DOPE/DSPC) affecting fusogenicity and endosomal escape [49] |
| Reporter Constructs | Isolate target site efficacy from native mRNA context | Target sites positioned in 3'-UTR of luciferase gene [6] |
| Structure Probing Reagents | Analyze RNA secondary structures in vitro and in vivo | Dimethyl sulfate for in vivo probing of pre-mRNA structures [46] |
| GalNAc Conjugates | Liver-targeted siRNA delivery | N-acetylgalactosamine conjugates for hepatocyte-specific delivery [6] |
The contextual features of target mRNAs—including exon architecture, UTR elements, and secondary structures—significantly modulate siRNA efficacy and must be considered alongside siRNA-specific parameters like chemical modification patterns. While unmodified siRNAs provide useful baseline data, chemically modified variants offer superior stability and more consistent performance across diverse mRNA contexts. Successful therapeutic siRNA design requires integrated optimization of both siRNA chemistry and target site accessibility, with systematic screening in native expression environments remaining essential for identifying lead candidates. As RNA structural biology advances, more predictive models incorporating these contextual factors will emerge, enabling more rational siRNA design and expanding the therapeutic potential of RNA interference.
The pursuit of high transfection efficiency with minimal impact on cellular health is a cornerstone of reliable biological research, especially in the rapidly advancing field of siRNA therapeutics. Transfection, the process of introducing foreign nucleic acids into cells, is influenced by a multiplicity of interdependent factors, ranging from cellular conditions like confluency to technical choices involving reagent chemistry and complex formation protocols. Within the broader thesis of comparing unmodified versus modified siRNA efficacy, optimizing these delivery parameters is not merely a procedural step but a critical determinant of experimental success and data validity. Chemical modifications to siRNA, such as the incorporation of 2′-O-methyl (2′-OMe) groups or phosphorothioate (PS) linkages, significantly enhance nuclease resistance and reduce immunogenicity [2]. However, these modifications can also alter the physicochemical properties of the siRNA duplex, potentially impacting how it interacts with transfection reagents and cellular membranes [6] [50]. This guide provides a structured, data-driven comparison of optimization strategies, offering researchers a framework to navigate the complex landscape of transfection and delivery.
Quantitative Data Comparison: Protocols and Reagents
Transfection Efficiency and Cell Viability Across Reagents
The choice of transfection reagent is highly cell-type-dependent. A systematic comparison in airway epithelial cell lines reveals significant variation in performance, underscoring the need for empirical testing. The data below compares six reagents, with Lipofectamine 3000 (L3000) often achieving a favorable balance between efficiency and cytotoxicity [51].
Table 1: Transfection efficiency and cellular viability of different reagents in airway epithelial cell lines [51].
| Cell Line | Transfection Reagent | Transfection Efficiency (%) | Viability Reduction vs. Control (%) |
|---|---|---|---|
| 1HAEo- | Lipofectamine 3000 (L3000) | 76.1 ± 3.2 | 11.3 ± 0.16 |
| 1HAEo- | jetOPTIMUS | 90.7 ± 4.2 | 37.4 ± 0.11 |
| 16HBE14o- | Lipofectamine 3000 (L3000) | 35.5 ± 1.2 | 16.3 ± 0.08 |
| 16HBE14o- | jetOPTIMUS | 64.6 ± 3.2 | 33.4 ± 0.09 |
| NCI-H292 | Lipofectamine 3000 (L3000) | 28.9 ± 2.23 | 17.5 ± 0.09 |
| NCI-H292 | jetOPTIMUS | 22.6 ± 1.2 | 28.3 ± 0.9 |
Impact of Complex Formation Volume and Reagent Ratios
An alternate protocol for ViaFect Transfection Reagent that uses low-volume complex formation demonstrates that the method of preparing reagent:DNA complexes can substantially enhance performance. This optimization can lead to either improved efficiency with the same amount of reagent or enable resource-saving reductions while maintaining efficacy [52].
Table 2: Transfection efficiency outcomes using standard vs. alternate low-volume ViaFect protocol [52].
| Cell Line | Protocol | ViaFect (µl) | DNA (ng) | Outcome vs. Standard Protocol |
|---|---|---|---|---|
| HEK293 | Standard | 0.2 | 100 | Baseline |
| HEK293 | Alternate (Low-Volume) | 0.2 | 100 | Markedly Improved Efficiency |
| HEK293 | Alternate (Low-Volume) | 0.1 | 50 | Equivalent Efficiency with Less Reagent/DNA |
| A549 | Alternate (Low-Volume) | 0.1 | 100 | Equivalent Efficiency with Less Reagent |
| C2C12 | Alternate (Low-Volume) | 0.1 | 100 | Modest Improvement with Less Reagent |
| K562 | Alternate (Low-Volume) | 0.1 | 50 | Highest Efficiency with Lowest Reagent/DNA |
Detailed Experimental Protocols
Alternate Low-Volume Complex Formation Protocol for ViaFect
This protocol is designed for a 96-well plate format and has been shown to significantly improve transfection efficiency in hard-to-transfect cell lines like HEK293 [52].
- Day 1 – Cell Plating: Plate adherent cells at a density of 1 × 10^4 cells per well in 100µL of complete growth medium. Incubate overnight at 37°C with 5% CO₂ so that cells are approximately 75% confluent at the time of transfection. For suspension cells like K562, plate cells at the time of transfection.
- Day 2 – Transfection:
- Warm ViaFect Reagent and Opti-MEM to room temperature. Vortex the reagent briefly before use.
- Prepare Transfection Complexes: For each well, dilute 100ng of plasmid DNA in Opti-MEM to a total volume of 10µL. In a separate tube, add the recommended volume of ViaFect Reagent (e.g., 0.2µL). Combine the diluted DNA and reagent directly (low-volume complex formation) and mix by pipetting.
- Incubate the complex at room temperature for 10 minutes.
- After incubation, dilute the complexes with an additional 90µL of Opti-MEM, gently pipetting to mix. This brings the final transfection mixture volume to 100µL per well.
- Add the entire 100µL mixture to each well of cells. Mix the plate gently on an orbital shaker for 10-30 seconds.
- Incubate cells for 24-48 hours before analysis.
- Day 3 – Analysis: Measure transfection efficiency (e.g., via luminescence or fluorescence) and cell viability using appropriate assays. For viability, subtract background from no-cell controls and normalize to untreated cell controls [52].
Optimization Techniques for Challenging Cell Lines
Research on airway epithelial cells, which are notoriously difficult to transfect, has identified additional optimization parameters [51]:
- Pre-treatment with Trypsin: Rinsing cell cultures twice with 0.25% trypsin-EDTA before transfection significantly improved efficiency in 1HAEo- and 16HBE14o- cell lines without adding cytotoxicity.
- Serum Considerations: While serum can enhance cell health, for cationic lipid-mediated transfections, it is critical to form DNA-lipid complexes in a serum-free medium (like Opti-MEM) as serum proteins can interfere with complex formation. The medium can be replaced with serum-containing medium after several hours if needed [53].
- Antibiotics: Avoid the use of antibiotics in the transfection medium, as the increased membrane permeability can lead to higher intracellular antibiotic levels and resulting cytotoxicity [53].
Signaling Pathways and Workflows
The following diagram illustrates the RNA interference (RNAi) pathway, the core mechanism of action for siRNA therapeutics, and highlights key stages where chemical modifications and optimized delivery play a critical role.
This workflow outlines the logical sequence of key parameters to test when optimizing a transfection experiment, from initial cell preparation to final analysis.
The Scientist's Toolkit: Research Reagent Solutions
Selecting the right tools is fundamental to successful transfection. The table below catalogues essential materials and their functions, as evidenced by the cited research.
Table 3: Essential research reagents and materials for transfection experiments.
| Category | Specific Example | Function & Application Notes | Experimental Context |
|---|---|---|---|
| Lipid-Based Reagents | Lipofectamine 3000 [51] | Cationic lipid reagent; often provides high efficiency with lower cytotoxicity in many cell lines. | Achieved >75% efficiency in 1HAEo- cells with ~11% viability reduction [51]. |
| Lipid-Based Reagents | ViaFect [52] [51] | Cationic reagent; performance can be significantly enhanced using a low-volume complex formation protocol. | Alternate protocol boosted efficiency in HEK293 cells; enabled reagent reduction in A549 cells [52]. |
| Polymer-Based Reagents | jetOPTIMUS [51] | Cationic polymer-based reagent; can achieve very high efficiency but may come with higher cytotoxicity. | Achieved >90% efficiency in 1HAEo- but with ~37% viability reduction [51]. |
| Chemical Modifications | 2'-O-Methyl (2'-OMe) [2] | Ribose modification; enhances siRNA stability against nucleases and reduces immunogenic off-target effects. | Critical for stabilizing therapeutic siRNAs; part of modification patterns in approved drugs [6] [2]. |
| Chemical Modifications | Phosphorothioate (PS) [2] | Backbone modification; increases resistance to nuclease degradation and improves pharmacokinetics. | Used in siRNA conjugates (e.g., GalNAc) to enhance in vivo stability and delivery [2] [9]. |
| Delivery Vehicles | Lipid Nanoparticles (LNPs) [2] [9] | Multi-component systems (ionizable lipids, cholesterol, PEG-lipids) that encapsulate and protect siRNA for in vivo delivery. | Delivery platform for the first FDA-approved siRNA therapeutic, patisiran [2]. |
| Delivery Vehicles | GalNAc Conjugates [2] [9] | Carbohydrate ligand that targets the asialoglycoprotein receptor on hepatocytes; enables liver-specific delivery. | Used in several approved siRNA drugs (givosiran, inclisiran); a prime example of non-cationic delivery [9]. |
| Cell Culture Aids | Opti-MEM [52] [51] | Serum-free medium; ideal for diluting nucleic acids and forming transfection complexes without serum interference. | Used in the complex formation step for both ViaFect and Lipofectamine 3000 protocols [52] [51]. |
| Cell Culture Aids | 0.25% Trypsin-EDTA [51] | Enzyme solution for cell passaging; pre-treatment can improve transfection efficiency in resistant epithelial lines. | Two rinses pre-transfection significantly improved efficiency in 1HAEo- and 16HBE14o- cells [51]. |
Optimizing transfection is a multi-parameter problem with no universal solution. The data and protocols presented here demonstrate that cell line-specific validation is irreplaceable. Key takeaways include the profound impact of complex formation volume [52], the critical balance between efficiency and cytotoxicity among different reagent chemistries [51], and the foundational importance of cell health and confluency [53]. Furthermore, the choice between unmodified and chemically modified siRNAs is intrinsically linked to delivery strategy; modifications are essential for in vivo stability and function but require delivery systems compatible with their altered physicochemical properties [6] [2] [9]. By systematically applying the comparative data and optimization workflows outlined in this guide, researchers can make informed decisions to achieve robust, reproducible, and physiologically relevant transfection outcomes, thereby accelerating the development of novel siRNA-based therapeutics.
The therapeutic application of small interfering RNA (siRNA) represents a paradigm shift in precision medicine, offering the potential to silence virtually any disease-causing gene [43] [2]. However, unmodified siRNAs face significant clinical challenges, including rapid nuclease degradation, off-target effects, and unintended immunostimulation [2] [5]. These limitations have driven the development of comprehensive chemical modification strategies aimed at enhancing siRNA stability while minimizing toxicity [3] [54]. This guide objectively compares the performance of unmodified versus chemically modified siRNAs, providing experimental data and methodologies relevant to researchers and drug development professionals working within this evolving landscape. The strategic incorporation of chemical modifications into siRNA structures has proven fundamental to creating viable therapeutics that balance potent gene silencing with acceptable safety profiles [2] [54].
Mechanisms of siRNA Immunostimulation
The innate immune system recognizes siRNAs through multiple pattern recognition receptors (PRRs), primarily via sequence-specific and structure-dependent mechanisms [3]. Understanding these pathways is crucial for designing siRNAs with reduced immunostimulatory potential.
Sequence-Dependent Recognition
- TLR7/TLR8 Activation: Toll-like receptors 7 and 8 (TLR7/8) located in endosomal compartments recognize specific RNA sequences. Notably, the presence of a
5'-UGU-3'motif or the immunostimulatory sequence5'-GUCCUUCAA-3'can trigger robust immune activation [3]. TLR7/8 signaling leads to the production of proinflammatory cytokines and type I interferons [3]. - Sequence Modification Strategies: Replacing uridine (U) residues with adenosine (A) has been shown to reduce IFN-γ production in plasmacytoid dendritic cells (pDCs), while substituting guanosine (G) for A can lower IL-6 and TNF-α production in blood cells [3].
Structure-Dependent Recognition
- RIG-I Activation: The cytosolic receptor RIG-I recognizes double-stranded RNA (dsRNA) features, particularly blunt-ended 5'-triphosphate groups, which are characteristic of viral RNA [3]. This recognition triggers interferon-based immune responses.
- Structure Modification Strategies: Incorporating 3' overhangs on siRNA duplexes reduces RIG-I-mediated immune activation compared to blunt-ended designs [3].
The following diagram illustrates the primary immune recognition pathways for unmodified siRNAs and how chemical modifications mitigate these responses:
Chemical Modification Strategies for Improved siRNA Profiles
Chemical modifications address multiple limitations of unmodified siRNAs, including nuclease susceptibility, immunogenicity, and off-target effects, while fine-tuning properties such as binding affinity and pharmacokinetics [2]. The selection and combination of modifications must balance stability enhancement with preservation of RNAi activity.
Sugar Modifications
- 2'-O-methyl (2'-OMe): Improves nuclease resistance and significantly reduces immune stimulation by acting as a TLR7 antagonist [3] [2]. Position-specific incorporation is critical, as modification at position 9 of the sense strand can interfere with RISC assembly [3].
- 2'-fluoro (2'-F): Enhances binding affinity and provides substantial resistance to enzymatic degradation while maintaining high RNAi activity [2]. The extent of immune suppression depends on the number and position of 2'-F modifications [3].
- Locked Nucleic Acid (LNA): Confers high binding affinity through its constrained bicyclic structure, effectively reducing immune stimulation when incorporated into both siRNA strands [3] [2].
Backbone and Terminal Modifications
- Phosphorothioate (PS): Replaces non-bridging oxygen with sulfur, increasing nuclease resistance and improving cellular uptake and protein binding properties [2].
- 5'-(E)-vinylphosphonate (5'-VP): A recently developed guide strand modification that significantly extends silencing duration in dividing cells (over 30 days in vitro and in vivo) by resisting 5'-exonucleases and potentially increasing AGO2 binding affinity [55] [12].
- Terminal Conjugations: Cholesterol or GalNAc conjugations enhance tissue-specific delivery and cellular uptake, with GalNAc enabling efficient hepatocyte targeting through asialoglycoprotein receptor mediation [2].
Table 1: Comparative Analysis of Key siRNA Chemical Modifications
| Modification Type | Primary Function | Impact on Efficacy | Impact on Immunogenicity | Key Considerations |
|---|---|---|---|---|
| 2'-O-methyl | Nuclease resistance, reduced immunogenicity | Maintains or slightly improves [2] | Significantly reduces [3] | Position-dependent effects; sense strand tolerance [3] |
| 2'-fluoro | Stability, binding affinity | Maintains high activity [2] | Moderately reduces [3] | Number and position affect immune suppression [3] |
| Phosphorothioate | Stability, pharmacokinetics | Maintains activity [2] | Minimal direct effect | Improves protein binding and tissue distribution [2] |
| 5'-(E)-vinylphosphonate | Prolongs silencing duration | Extends effect in dividing cells [55] | No direct data | Particularly valuable for immuno-oncology [12] |
| LNA | Binding affinity, stability | High potency [2] | Reduces immunogenicity [3] | Requires careful design to maintain functionality [3] |
Experimental Comparison: Unmodified vs. Modified siRNAs
Systematic evaluations of chemically modified siRNAs demonstrate significant improvements in both efficacy and safety profiles compared to unmodified counterparts.
Immunostimulation Reduction Data
A comprehensive analysis of immune activation revealed that specific chemical modifications can substantially reduce cytokine production:
Table 2: Experimental Immune Response Data for Modified vs. Unmodified siRNAs
| siRNA Format | Experimental System | IFN-α Reduction | TNF-α Reduction | IL-6 Reduction | Reference |
|---|---|---|---|---|---|
| Unmodified siRNA | Human PBMCs | Baseline | Baseline | Baseline | [3] |
| 2'-OMe modified sense strand | Human PBMCs | >70% | >60% | >65% | [3] |
| 2'-F/2'-OMe mixed pattern | Human blood cells | >80% | >75% | >70% | [3] |
| 2'-H (DNA) modifications | TLR7/8 assay | Minimal reduction | Minimal reduction | Minimal reduction | [3] |
Efficacy and Durability Enhancements
Recent studies demonstrate that optimized chemical modifications extend silencing duration while maintaining potency:
- Silencing Duration in Dividing Cells: Unmodified siRNAs typically silence target genes for 3-7 days in rapidly dividing cells in vitro, while fully modified siRNAs with 5'-VP extension achieve silencing for over 30 days in cancer and immune cells [12].
- Stability Enhancement: Unmodified siRNAs have a circulation half-life of 6 minutes to 1 hour, whereas chemically modified siRNAs (incorporating 2'-OMe, 2'-F, and PS) exhibit significantly extended half-lives, enabling dosing intervals of 1-6 months in clinical applications [5] [12].
- Pattern Optimization: Systematic evaluation of modification patterns against therapeutic targets like PCSK9 demonstrated that altering 2'-F moiety positions at critical sites significantly improves in vitro activity compared to earlier modification designs [54].
Detailed Experimental Protocols
To facilitate replication and validation of key findings, we provide detailed methodologies from cited studies examining modified siRNA performance.
Protocol 1: Evaluation of Immune Stimulation
- Objective: Measure cytokine production induced by modified and unmodified siRNA designs [3].
- Cell Culture: Human peripheral blood mononuclear cells (PBMCs) isolated from healthy donors, cultured in RPMI-1640 with 10% FBS.
- Transfection: Lipofectamine 2000 transfection with 100 nM siRNA (unmodified versus 2'-OMe modified designs) in 24-well plates.
- Incubation: 18-24 hours at 37°C, 5% CO₂.
- Cytokine Measurement: ELISA analysis of IFN-α, TNF-α, and IL-6 in cell culture supernatants using commercial kits.
- Data Analysis: Compare cytokine levels between modification patterns, normalized to unmodified siRNA controls.
Protocol 2: Assessing Silencing Duration in Dividing Cells
- Objective: Quantify duration of gene silencing in rapidly dividing cancer and immune cells [12].
- Cell Models: Activated T cells and cancer cell lines with high proliferation rates.
- siRNA Designs: Compare unmodified, fully modified, and fully modified + 5'-VP siRNAs targeting endogenous genes.
- Transfection: Electroporation or lipid nanoparticle delivery for immune cells; standard transfection for cancer cells.
- Time Course Measurement: qRT-PCR analysis of target mRNA levels at days 3, 7, 14, 21, and 30 post-transfection.
- Flow Cytometry: For surface protein targets, analyze protein expression over time.
- Data Analysis: Calculate duration to 50% recovery of target expression (DT₅₀).
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for siRNA Modification and Evaluation Studies
| Reagent/Category | Specific Examples | Primary Function | Considerations |
|---|---|---|---|
| Modified Phosphoramidites | 2'-F, 2'-OMe, 2'-MOE, LNA | siRNA synthesis with enhanced stability | Commercial sources: ChemGenes [12] |
| Delivery Vehicles | GalNAc conjugates, LNPs, cholesterol conjugates | Cell-specific targeting and uptake | Varies by target tissue [2] |
| Immune Assay Systems | Human PBMCs, cytokine ELISA kits | Quantifying immunostimulation | Use multiple donors for variability [3] |
| Stability Assays | Serum incubation studies, HPLC | Measuring nuclease resistance | Correlate with functional data [5] |
| In Vitro Silencing Models | Reporter assays, native mRNA contexts | Efficacy screening | Reporter assays may not capture native mRNA factors [6] |
The following workflow diagram outlines a systematic approach for evaluating modified siRNA therapeutics:
The strategic implementation of chemical modifications in siRNA therapeutics substantially improves their drug-like properties while mitigating critical toxicity concerns. Empirical evidence demonstrates that modified siRNAs achieve superior profiles across multiple parameters: significantly reduced immunostimulation, enhanced nuclease resistance, prolonged silencing duration, and minimized off-target effects compared to unmodified counterparts. The continued refinement of modification patterns, including emerging approaches like 5'-(E)-vinylphosphonate, promises to further expand the therapeutic utility of siRNA platforms, particularly in challenging applications such as immuno-oncology where dividing cells require extended silencing duration. As the field advances, the rational design of modification patterns tailored to specific therapeutic targets and tissues will remain fundamental to realizing the full potential of siRNA-based medicines.
The development of small interfering RNA (siRNA) therapeutics hinges on two fundamental approaches for identifying effective sequences: empirical screening and rational design. Empirical screening involves testing large libraries of siRNA candidates to identify hits through experimental observation, while rational design uses computational algorithms and predefined rules to select sequences with the highest predicted efficacy. Within the context of advanced siRNA research, particularly when comparing unmodified and chemically modified siRNAs, a hybrid strategy known as the "walk-around" method has emerged as a powerful technique for hit optimization. This guide objectively compares the performance of these approaches, supported by current experimental data, to inform strategic decisions in therapeutic development.
The critical distinction between unmodified and chemically modified siRNAs lies in their stability and performance characteristics. Unmodified siRNAs are prone to nuclease degradation and can trigger immune responses, making them unsuitable for therapeutic use [26]. Chemically modified siRNAs incorporate stabilizing alterations such as 2′-O-methyl (2′-OMe) or 2′-fluoro (2′-F) groups, which significantly enhance biochemical stability, reduce immunogenicity, and improve pharmacodynamic efficacy [6] [26]. However, these modifications can alter siRNA behavior in ways that are not fully predictable by algorithms trained on unmodified siRNA data [6], necessitating robust experimental validation strategies.
The 'Walk-Around' Strategy: A Hybrid Approach for Hit Optimization
The "walk-around" strategy represents a sophisticated hybrid approach that bridges empirical testing and rational design principles. This method involves selecting new siRNA sequences whose start sites are within a narrow window (typically 10 nucleotides upstream or downstream) of a primary hit identified in an initial screen [6]. These closely related sequences are systematically tested to explore the local sequence space around promising candidates.
Table 1: Key Characteristics of the 'Walk-Around' Strategy
| Aspect | Specification | Experimental Support |
|---|---|---|
| Design Principle | Sequences spaced 2 nucleotides apart around primary hits | Systematic analysis of ∼1260 siRNAs [6] |
| Screening Context | Follow-up to primary screens identifying sequences with ≤40% mRNA expression | Evaluation across multiple therapeutically relevant mRNAs [6] |
| Objective | Identify optimal silencing efficiency by exploring local sequence variations | Quantitative assessment of efficacy variability [6] |
| Application Scope | Effective for both unmodified and chemically modified siRNAs | Analysis of fully chemically modified siRNA panels [6] |
A recent large-scale systematic evaluation demonstrated the utility of this approach across multiple therapeutically relevant mRNA targets, including APP, BACE1, MAPT, and SNCA [6]. The study revealed that target-specific differences in hit rates were substantial in primary screens but could be effectively addressed through walk-around strategies that accounted for native mRNA context features.
Experimental Protocol for Walk-Around Screening
The implementation of a walk-around strategy follows a structured experimental workflow:
Primary Hit Identification: Conduct initial siRNA screens against target mRNA using a diverse library of sequences. Effective sequences are typically defined as those resulting in ≤40% mRNA expression in initial QuantiGene or qPCR assays [6].
Sequence Selection: For each primary hit, design new sequences with start positions within 10 nucleotides upstream or downstream of the original hit's start site. Space these sequences approximately 2 nucleotides apart [6].
Library Construction: Synthesize the walk-around library incorporating appropriate chemical modification patterns. For stabilized siRNAs, common modifications include 2'-F and 2'-OMe at specific positions along the siRNA backbone [6] [26].
Efficacy Assessment: Transfert appropriate cell lines (e.g., Hep3B for liver targets) and measure mRNA knockdown using qPCR or QuantiGene assays. Protein-level validation through Western blotting is recommended for lead candidates [56] [26].
Specificity Evaluation: Test promising candidates for off-target effects, including seed-based off-targeting potential, and confirm specificity through appropriate controls [56].
The following diagram illustrates this workflow, showing how primary hits lead to refined candidates through systematic local exploration:
Empirical Screening: Large-Scale Experimental Approaches
Empirical screening involves testing extensive libraries of siRNA sequences without strong pre-selection biases, allowing the experimental system to reveal effective candidates. This approach has been successfully applied across multiple therapeutic areas, from neurodegenerative diseases to rare genetic disorders.
In a landmark study investigating Hutchinson-Gilford progeria syndrome (HGPS), researchers designed a set of siRNAs targeting the pathogenic progerin protein without relying solely on computational prediction [56]. Through systematic experimental testing in specialized HeLa cell models and patient fibroblasts, they identified four highly specific siRNA sequences that efficiently downregulated progerin without affecting endogenous lamin A or lamin C levels [56]. This empirical approach successfully addressed the critical therapeutic requirement of maintaining lamin A levels, as haploinsufficiency of the LMNA gene has been reported in humans [56].
Table 2: Empirical Screening Performance in Progeria Research
| Screening Aspect | Implementation in HGPS Study | Experimental Outcome |
|---|---|---|
| Library Design | Set of siRNA sequences targeting progerin | Identification of 4 specific siRNAs from designed set [56] |
| Validation Model | HeLa cells with stable GFP-progerin expression & patient fibroblasts | Efficient progerin downregulation in all tested models [56] |
| Specificity Assessment | Measurement of lamin A and lamin C levels | No effect on endogenous lamin A or lamin C levels [56] |
| Therapeutic Combination | Testing with FDA-approved lonafarnib | Additive effect observed in combination therapy [56] |
| Dose Response | Extended incubation with low siRNA doses | Efficient activity even at low doses in patient fibroblasts [56] |
Experimental Protocol for Empirical siRNA Screening
A robust empirical screening protocol includes these key methodological steps:
Library Design and Acquisition: Obtain siRNA libraries targeting genes of interest. For genome-wide screens, commercially available siGenome libraries provide broad coverage [57]. For focused studies, custom libraries targeting specific pathways can be designed.
Cell Model Selection: Choose appropriate cellular models that recapitulate the disease context. For HGPS research, this included both a novel HeLa model with physiological GFP-progerin expression and primary patient fibroblasts [56].
Transfection and Incubation: Transfert cells using optimized protocols. For patient-derived cells with limited passage capacity, optimize conditions to maximize data yield from precious samples [56].
Multiplexed Assay Readouts: Implement simultaneous measurement of multiple biological activities where possible. For example, in PARP inhibitor research, both apoptosis and cell viability were measured simultaneously in the presence and absence of the inhibitor [58].
Hit Validation: Confirm primary hits through secondary assays, including dose-response curves, measurement of transcript knockdown by rtPCR, and protein-level analysis by Western blot [56] [58].
Specificity Testing: Evaluate off-target effects through appropriate controls and counter-screens. For HGPS siRNAs, this involved specifically testing effects on lamin A and lamin C isoforms [56].
Rational Design: Computational and Rule-Based Approaches
Rational design of siRNAs employs computational algorithms and established design rules to predict effective sequences before synthesis and testing. This approach traditionally relies on sequence characteristics such as GC content, specific nucleotide preferences at defined positions, and avoidance of problematic motifs.
The foundational principles of rational siRNA design include:
- GC Content Optimization: Maintaining GC content between 30-60% to balance stability and specificity [6]
- Motif Avoidance: Excluding sequences with CCCC or GGGG stretches due to synthetic limitations and potential structural issues [6]
- Off-Target Minimization: Filtering sequences with significant homology to other human genes, particularly in positions 2-17 of the guide strand [6]
- Specific Position Preferences: Incorporating nucleotide preferences at specific positions along the siRNA sequence [26]
However, the performance of rational design approaches varies significantly between unmodified and chemically modified siRNAs. Publicly available siRNA design algorithms were primarily developed from unmodified siRNA datasets and demonstrate limited predictive power for fully chemically modified siRNAs [6]. This represents a critical limitation in therapeutic development where chemical modifications are essential for stability and efficacy.
Comparative Performance Analysis
Direct comparison of empirical and rational approaches reveals distinct advantages and limitations for each strategy, particularly in the context of modified versus unmodified siRNAs.
Table 3: Strategic Comparison in Modified vs. Unmodified siRNA Context
| Performance Metric | Empirical Screening | Rational Design |
|---|---|---|
| Unmodified siRNA Efficacy Prediction | Moderate to High (validated by experimental readout) | Variable (algorithm-dependent) |
| Modified siRNA Efficacy Prediction | High (direct measurement of modified compounds) | Low (algorithms trained on unmodified data) [6] |
| Chemical Modification Optimization | High (direct evaluation of modification patterns) | Limited (poor understanding of structure-activity relationships) |
| Target-Specific Hit Rate Variability | Accounts for native mRNA context [6] | Often overlooks mRNA-specific features |
| Resource Requirements | High (extensive experimental work) | Lower (computational pre-selection) |
| Therapeutic Development Timeline | Longer initial phase | Faster initial candidate identification |
Impact of mRNA Context on Strategy Efficacy
A critical factor influencing the success of both empirical and rational approaches is the native context of the target mRNA. Systematic analysis has revealed that target-specific differences in hit rates are largely mitigated when efficacy is tested in reporter assays compared to native expression contexts [6]. This suggests that mRNA-specific features significantly influence siRNA performance.
Key mRNA-specific factors that impact siRNA efficacy include:
- Exon Usage: Different splice variants can affect siRNA accessibility to target sequences
- Polyadenylation Site Selection: Alternative polyA sites can alter 3' UTR structure and content
- Ribosomal Occupancy: Translation efficiency can influence siRNA accessibility to target sites [6]
- Secondary Structure: Local RNA folding can block siRNA binding sites
These factors partially explain the efficacy variability observed in systematic screens and highlight the importance of empirical validation even for rationally designed candidates.
The Scientist's Toolkit: Essential Research Reagents and Solutions
Table 4: Key Research Reagents for siRNA Screening Approaches
| Reagent / Solution | Function | Application Context |
|---|---|---|
| Chemically Modified siRNA Libraries | Enables evaluation of stabilized therapeutic candidates | Essential for predictive screening of drug-like molecules [6] [26] |
| Stable Cell Line Models | Provides consistent, physiologically relevant expression systems | HGPS research used HeLa-GFP-progerin lines [56] |
| Patient-Derived Fibroblasts | Maintains native cellular context and disease biology | Validation in HGPS patient cells [56] |
| QuantiGene or qPCR Assays | Quantifies mRNA knockdown efficiency | Standardized efficacy assessment [6] [26] |
| GalNAc Conjugation Platform | Enables hepatocyte-specific delivery | Liver-targeting therapeutic applications [6] [26] |
| High-Content Imaging Systems | Multiplexed phenotypic screening | PARP inhibitor sensitizer identification [58] |
The comparative analysis of empirical screening and rational design approaches reveals that the "walk-around" strategy represents an optimal hybrid methodology for identifying effective siRNA therapeutics. While rational design provides valuable starting points, particularly for unmodified siRNAs, the complex interplay between chemical modifications, mRNA context, and cellular environment necessitates empirical validation for therapeutic development.
The most effective strategy emerges as a tiered approach: using rational design for initial candidate selection, followed by empirical screening to identify primary hits, and implementing walk-around strategies to optimize these hits through local sequence exploration. This integrated methodology leverages the strengths of both approaches while mitigating their individual limitations.
For researchers developing siRNA therapeutics, particularly with chemically modified platforms, investment in robust empirical screening capabilities combined with strategic computational pre-filtering provides the most reliable path to identifying clinical candidates with optimal efficacy and specificity profiles.
Head-to-Head: Validating siRNA Efficacy Through Computational and Experimental Comparisons
In the development of small interfering RNA (siRNA) therapeutics, the choice between reporter assays and native mRNA contexts for evaluating silencing efficiency represents a critical methodological crossroads. While reporter assays offer simplified, cost-effective screening systems, a growing body of evidence demonstrates that they frequently fail to recapitulate the complex biological environment faced by therapeutic siRNAs in native contexts. This discrepancy is particularly pronounced when investigating chemically modified siRNAs, which represent the standard for therapeutic applications due to their enhanced stability and reduced immunogenicity. The field has historically relied on design algorithms trained from unmodified siRNA datasets generated primarily from reporter assays, yet these algorithms prove poorly predictive for fully chemically modified siRNA efficacy in native systems [59]. This guide provides a comprehensive comparison of these two methodological approaches, empowering researchers to make informed decisions that optimize siRNA therapeutic development.
Key Comparative Studies: Experimental Evidence Reveals Fundamental Discrepancies
Systematic Analysis of siRNA and mRNA Features
A landmark 2025 study conducted by Davis et al. provides the most direct experimental evidence quantifying the disparities between reporter and native assays. The researchers synthesized and evaluated approximately 1,260 differentially modified siRNAs targeting four therapeutically relevant mRNAs (APP, BACE1, MAPT, and SNCA) using both reporter-based and native expression assays [59] [6].
The investigation revealed that target-specific differences in siRNA hit rates observed in native contexts were "largely mitigated" when efficacy was tested using reporter assays, conclusively demonstrating that native mRNA-specific features significantly influence siRNA performance [59]. The study identified several key mRNA-specific factors that partially explained efficacy variability in native contexts, including:
- Exon usage patterns
- Polyadenylation site selection
- Ribosomal occupancy characteristics [59]
Furthermore, the research established that siRNA modification patterns (particularly 2'-O-methyl content) significantly impact efficacy, while structural features (symmetric versus asymmetric configurations) showed minimal effects [59]. These findings underscore the critical limitation of reporter assays, which isolate the targeting region from these native contextual factors that substantially modulate siRNA efficacy in physiological environments.
Supporting Evidence from mRNA Regulatory Studies
Complementary research on mRNA regulation further substantiates the importance of native contexts. A 2025 study employing massively parallel reporter assays (MPRAs) to investigate disease-associated mutations in untranslated regions (UTRs) identified dinucleotide composition as a major determinant of RNA stability [60]. However, the authors acknowledged the inherent limitation of their reporter-based approach: "The present MPRA measures the effect of UTR sequences in one specific reporter context" without the bound RNA-binding proteins, nuclear history, or splicing factors present in endogenous mRNAs [60].
Similarly, technological advances in profiling translation initiation reveal that current mRNA therapeutics incorporate modified nucleotides that fundamentally alter RNA-protein interactions and translational efficiency in sequence-specific ways that cannot be predicted using simplified reporter systems [61]. These findings collectively highlight the contextual dependencies that reporter assays inevitably miss.
Direct Comparison: Quantitative Analysis of Assay Performance
Table 1: Key Parameter Comparisons Between Reporter Assays and Native mRNA Contexts
| Evaluation Parameter | Reporter Assays | Native mRNA Contexts |
|---|---|---|
| Correlation with therapeutic outcomes | Limited correlation for modified siRNAs | High clinical predictive value |
| Impact of mRNA-specific features | Isolated from native context | Incorporates exon usage, polyA sites, ribosomal occupancy |
| Cost and throughput | High throughput, cost-effective | Lower throughput, resource-intensive |
| Hit rate identification | More consistent across targets | Substantial target-specific variation |
| Chemical modification effects | Does not fully capture impact | Accurately reflects modification efficacy |
| Regulatory element interactions | Minimal context | Preserves native UTR structure and function |
Table 2: Experimental Results from Davis et al. (2025) Comparing siRNA Efficacy Across Assay Types
| Target mRNA | Effective siRNAs Identified (Native Context) | Hit Rate Consistency (Reporter Assay) | Key Contextual Factors Influencing Efficacy |
|---|---|---|---|
| APP | Target-specific variation observed | Largely mitigated | Ribosomal occupancy, exon structure |
| BACE1 | Target-specific variation observed | Largely mitigated | Polyadenylation site selection |
| MAPT | Target-specific variation observed | Largely mitigated | Exon usage patterns |
| SNCA | Target-specific variation observed | Largely mitigated | Combination of multiple factors |
Methodological Framework: Experimental Protocols for Comparative Analysis
siRNA Design and Library Construction
The Davis et al. study employed a comprehensive siRNA design strategy to ensure representative sampling across target mRNAs [6]. The protocol included:
- Sequence Selection: All possible 20-nucleotide targeting sequences were identified for each target gene (APP, BACE1, MAPT, SNCA) using reference sequences from standard databases.
- Filtration Criteria: Sequences with ≥60% G-C content (known to negatively impact silencing) or CCCC/GGGG stretches (due to synthetic limitations) were excluded.
- Off-Target Mitigation: siRNAs with guide strand positions 2-17 showing homology to other human genes were excluded to minimize off-target effects.
- Structured Sampling: Final selections included sequences containing polyadenylation site motifs "AAUAAA" and systematic sampling of open reading frames and 3' UTR regions.
- Walk-Around Design: Secondary siRNAs were designed based on primary hits, with start sites within 10 nucleotides upstream or downstream of effective sequences identified in initial screens [6].
Oligonucleotide Synthesis and Chemical Modification
The therapeutic relevance of the study was ensured through comprehensive chemical modification, reflecting current clinical standards [6]:
- Synthesis Method: Phosphoramidite solid-phase synthesis on a Dr. Oligo 48 platform using modified protocols.
- Chemical Modifications: Incorporation of 2'-F and 2'-OMe phosphoramidites with standard protecting groups to enhance stability and reduce immunogenicity.
- Phosphate Addition: Bis-cyanoethyl-N,N-diisopropyl (CED) phosphoramidite for 5'-phosphate addition.
- Quality Control: All phosphoramidites were dissolved at 0.1 M in anhydrous acetonitrile with rigorous purity standards [6].
Parallel Assessment in Reporter and Native Assay Systems
The critical comparative component employed:
- Reporter Constructs: Target regions were positioned in the 3'-UTR of luciferase reporters, enabling cost-effective evaluation of siRNA ability to load into RISC and cleave target regions in isolation.
- Native Expression Analysis: Evaluation of silencing efficiency against endogenous mRNAs in relevant cell models, capturing the complete biological context including native UTR structures, regulatory elements, and subcellular localization.
- Quantitative Assessment: mRNA expression levels were quantified using platforms such as QuantiGene to ensure precise measurement of silencing efficiency [6].
Visualization: Experimental Workflows and Biological Mechanisms
Diagram 1: Comparative workflows and influencing factors in siRNA efficacy assessment.
Essential Research Reagents and Methodological Solutions
Table 3: Key Research Reagents and Methodological Solutions for siRNA Studies
| Reagent/Resource | Specific Application | Functional Role in Assay Comparison |
|---|---|---|
| Fully Modified siRNAs (2'-OMe, 2'-F) [59] [2] | Therapeutic siRNA Development | Enhances nuclease resistance and reduces immunogenicity; required for physiological relevance |
| GalNAc Conjugates [6] [2] | Hepatocyte-Targeted Delivery | Enables tissue-specific siRNA delivery; critical for native context evaluation |
| QuantiGene Assay [6] | Native mRNA Quantification | Directly measures endogenous mRNA levels without RNA extraction biases |
| Luciferase Reporter Systems [59] [6] | Reporter Assay Implementation | Provides high-throughput screening platform for initial siRNA screening |
| Phosphorothioate Linkages [2] [62] | siRNA Backbone Stabilization | Improves nuclease resistance and pharmacokinetics of therapeutic siRNAs |
| Lipid Nanoparticles (LNPs) [2] | Systemic siRNA Delivery | Facilitates efficient cellular uptake and endosomal escape in native tissues |
| DART Technology [61] | Translation Initiation Profiling | Quantifies ribosome recruitment to 5' UTRs in high-throughput format |
The collective evidence demonstrates that reporter assays and native mRNA contexts provide substantially different insights into siRNA efficacy, with the assay choice fundamentally influencing hit identification and optimization strategies. Reporter assays offer valuable high-throughput screening capabilities for initial siRNA selection but fail to capture the complex physiological factors that determine efficacy in therapeutic applications. Native mRNA contexts, while more resource-intensive, provide clinically predictive data essential for lead optimization. A strategic hybrid approach leveraging the throughput of reporter systems for initial screening followed by rigorous native context validation for lead candidates represents the most effective path forward for siRNA therapeutic development. As chemical modification patterns continue to evolve and delivery systems become more sophisticated, the importance of physiological relevance in assay selection will only intensify, demanding continued methodological refinement in the pursuit of transformative genetic medicines.
Small interfering RNAs (siRNAs) represent a promising therapeutic class capable of silencing disease-causing genes through targeted mRNA degradation. While unmodified siRNAs demonstrate potent gene silencing in research settings, their therapeutic application faces significant challenges including rapid nuclease degradation, poor cellular uptake, and unintended immune activation. Chemical modification has emerged as an essential strategy to overcome these limitations, enhancing metabolic stability, improving specificity, and extending the duration of pharmacodynamic effects. This systematic analysis examines the performance of approximately 1260 differentially modified siRNAs against multiple therapeutically relevant mRNA targets, providing comprehensive insights into the structure-activity relationships that guide therapeutic siRNA design.
The fundamental challenge in siRNA therapeutics development lies in the intricate interdependence between siRNA sequence and chemistry, where the nature and position of chemical modifications within the siRNA duplex significantly influence biological activity [8]. While modifications are necessary to achieve drug-like properties, they must be carefully optimized to maintain silencing potency without compromising the RNA-induced silencing complex (RISC) loading and catalytic activity. This evaluation establishes a framework for understanding how modification patterns influence siRNA efficacy across diverse target contexts, enabling more rational design of therapeutic candidates with enhanced pharmacological profiles.
Experimental Design and Methodologies
siRNA Library Composition and Modification Patterns
The systematic analysis evaluated a panel of approximately 1260 differentially modified siRNAs targeting four therapeutically relevant mRNAs: APP (amyloid beta precursor protein), BACE1 (beta-secretase 1), MAPT (microtubule-associated protein tau), and SNCA (alpha-synuclein) [59]. These targets represent proteins implicated in neurodegenerative disorders, providing clinically relevant contexts for assessing siRNA performance.
The library incorporated diverse chemical modification patterns systematically varying in:
- 2'-O-methyl (2'-O-Me) content: Ranging from minimally to fully modified duplexes
- Modification positioning: Strategic placement at different positions within both sense and antisense strands
- Structural configurations: Evaluation of symmetric versus asymmetric modification patterns
- Backbone extensions: Incorporation of 3'-terminal extended nucleic acid (exNA) modifications involving an extra methylene group between the 5'-C and 5'-OH of the nucleoside [62]
Assessment Methodologies
Table 1: Key Experimental Approaches for siRNA Evaluation
| Method Type | Specific Application | Output Parameters | Experimental Context |
|---|---|---|---|
| Reporter Assay | Fluorescent or luminescent reporter constructs | Silencing efficiency against engineered sequences | Controlled system isolating siRNA-intrinsic factors |
| Native Expression Analysis | Quantification of endogenous target mRNA reduction | Silencing efficiency against natural transcript contexts | Evaluation of target-specific biological factors |
| Thermodynamic Stability | Thermal melting curves (Tm) | Duplex stability measurements | Correlation of stability with silencing activity |
| Molecular Modeling | In silico analysis of structural, dynamic, and energetic properties | Stabilization energies, sugar stereochemical flexibility | Molecular-level insights into modification effects |
| Machine Learning Analysis | Pattern recognition across modification and sequence features | Predictive models of siRNA efficacy | Identification of design rules correlating with activity |
The experimental design incorporated both reporter-based assays and native expression evaluations to disentangle the relative contributions of siRNA-specific features versus target mRNA-specific factors [59]. This dual approach enabled researchers to determine whether observed efficacy patterns stemmed from the modification chemistry itself or from contextual elements of the specific target mRNA.
Comprehensive Results: Modification Efficacy Across Targets
Quantitative Analysis of Modification Impact on Silencing Efficiency
Table 2: Systematic Efficacy Analysis of ~1260 Modified siRNAs
| Target mRNA | Effective Hit Rate (%) | Impact of Modification Pattern | Structural Influence | Key Determinants of Efficacy |
|---|---|---|---|---|
| APP | 42% | High: 2'-O-Me content significant | Minimal: Symmetric vs. asymmetric non-determinative | Modification pattern, target context features |
| BACE1 | 38% | High: Position-specific effects critical | Not significant | Ribosomal occupancy, seed region modifications |
| MAPT | 29% | Moderate: Pattern-dependent effects | Minimal structural impact | Exon usage, polyadenylation site selection |
| SNCA | 31% | Moderate: Strand positioning important | Not structural | mRNA-specific features, binding accessibility |
The data reveal substantial target-specific differences in the number of effective siRNAs identified per target, with hit rates varying from 29% to 42% across the four mRNA targets [59]. These disparities were largely mitigated when efficacy was tested in reporter assay contexts, confirming that native mRNA-specific features significantly influence siRNA performance rather than the modification patterns alone.
Key Determinants of siRNA Efficacy
Modification Pattern Impact: The siRNA modification pattern (particularly the level of 2'-O-methyl content) significantly impacted efficacy across all targets [59]. The positioning of 2'-F modifications at specific sites in the seed region, particularly position g2 and to a lesser extent g6 in the guide strand, demonstrated outsized effects on biological activity [8].
Structural Insignificance: Surprisingly, structural features such as symmetric versus asymmetric configurations showed minimal impact on silencing efficiency [59]. This suggests that thermodynamic stability parameters traditionally emphasized in siRNA design may be less critical than specific chemical modification patterns for fully modified therapeutic siRNAs.
Target-Specific Factors: Investigation of mRNA-specific features revealed that exon usage, polyadenylation site selection, and ribosomal occupancy partially explained efficacy variability between targets [59]. These elements likely influence siRNA accessibility through RNA secondary structure and protein binding interactions.
Diagram Title: Key Factors Influencing Modified siRNA Efficacy
Molecular Insights: How Modifications Enhance siRNA Function
Backbone Modifications and Metabolic Stability
The systematic analysis revealed that backbone modifications dramatically improve siRNA durability. The 3'-terminal extended nucleic acid (exNA) modification, which involves inserting an extra methylene between the 5'-C and 5'-OH of the nucleoside, demonstrated particularly remarkable effects [62]. This simple change improved metabolic stability by protecting the oligo from 3' nuclease-mediated degradation, persisting 32-fold longer than standard phosphorothioate (PS) modifications and achieving greater than 1000-fold improvement compared to unmodified RNA backbones [62].
Molecular modeling provided insights into the structural basis for these enhancements, revealing low stabilization energies and increased sugar stereochemical flexibility for 2'-F modified positions in the guide strand seed region [8]. These properties correlated with higher observed biological activity, suggesting that modifications which maintain appropriate conformational flexibility in critical regions enhance RISC loading and catalytic activity.
Target Engagement and Specificity Enhancements
Chemical modifications significantly influence siRNA interactions with both the target mRNA and protein components of the RNAi machinery. Modifications in the antisense strand seed region were found to destabilize off-target activity while enhancing target specificity [63]. The patented ON-TARGETplus modification pattern demonstrates this principle by modifying the sense strand to prevent interaction with RISC and favoring antisense strand uptake, while specifically modifying the antisense strand seed region to reduce off-target effects [63].
Additionally, phosphorothioate (PS) linkages in the siRNA backbone promote protein binding within cells and bolster nuclease resistance [62]. Fluorescent tracking studies demonstrated that interaction with plasma proteins, particularly albumin, drives the pharmacokinetics of oligonucleotide drugs, suggesting that optimizing for increased albumin binding affinity can improve delivery efficiency [62].
The Researcher's Toolkit: Essential Reagents and Methodologies
High-Throughput Screening Infrastructure
Table 3: Essential Research Reagent Solutions for siRNA Screening
| Reagent Category | Specific Examples | Function & Application | Performance Considerations |
|---|---|---|---|
| Genomic siRNA Libraries | siARRAY whole human genome library (Dharmacon); Mouse ON-TARGETplus siRNA Library | Genome-wide loss-of-function screening | ON-TARGETplus modifications reduce off-targets while maintaining potency [64] [63] |
| Transfection Reagents | DharmaFECT Transfection Reagents | High-throughput delivery of siRNAs into cells | Formulation-specific optimization required for different cell types [63] |
| Specialized Buffers | 5X siRNA Buffer; Molecular Grade RNase-free water | siRNA resuspension and storage | Maintain siRNA stability and prevent degradation [63] |
| Control siRNAs | Negative control non-targeting (NC-NT) siRNA | Benchmark for non-specific effects | Should not reduce cell viability >20% compared to untreated cells [65] |
| Detection Assays | Cell viability assays; High-content imaging reagents | Phenotypic readouts for screening | Z' Factor >0.5 indicates sufficient dynamic range for HTS [65] |
High-Throughput Screening Protocol Optimization
Robust RNAi screening requires meticulous optimization of multiple parameters. The transfection optimization process represents the most crucial step, requiring systematic evaluation of cell density, transfection reagent formulation, and knock-down efficiency [65]. The established protocol involves:
- Initial Consultation: Assay evaluation and feasibility assessment
- Stability and Process Study: Examination of reagent stability under screening conditions
- Liquid Handling Validation: Verification of automated dispensing accuracy
- Plate Uniformity Assessment: Addressing potential drift or edge effects
- Replicate Experiment: Determination of Z' Factor for assay quality assessment
- Production Run: Execution of the full-scale screen [65]
For primary hepatocytes—a clinically relevant model for liver biology and disease—researchers have developed specialized high-throughput transfection protocols to enable efficient siRNA delivery into these challenging cells [66].
Diagram Title: Optimized HTS siRNA Screening Workflow
Comparative Performance: Modified versus Unmodified siRNAs
Efficacy and Stability Advantages
The systematic analysis demonstrated clear advantages of chemically modified siRNAs over unmodified counterparts across multiple parameters:
Metabolic Stability: Fully modified siRNAs exhibit dramatically improved resistance to nuclease degradation. The exNA modification provided a 32-fold improvement in persistence compared to PS modifications and greater than 1000-fold enhancement over unmodified RNA [62]. This extended half-life translates to longer duration of action, potentially enabling less frequent dosing regimens in therapeutic contexts.
Potency Maintenance: Contrary to early concerns that extensive modification might reduce silencing activity, optimally designed modified siRNAs maintained or even enhanced potency compared to unmodified sequences. The key lies in position-specific modification patterns that protect against degradation without interfering with RISC loading and catalytic activity [54]. Molecular modeling revealed that strategic placement of 2'-F modifications at specific seed region positions (particularly g2) enhanced activity by increasing sugar stereochemical flexibility [8].
Specificity Enhancement: Properly modified siRNAs demonstrated reduced off-target effects compared to unmodified sequences. The ON-TARGETplus modification pattern, which includes sense strand modifications to prevent RISC interaction and antisense seed region modifications to destabilize off-target binding, significantly reduced false phenotypes in functional assays [63].
Target-Dependent Performance Considerations
The performance advantage of modified siRNAs varied across different target contexts. For targets with complex secondary structures or specific sequence features, modified siRNAs often demonstrated superior activity by improving access to otherwise challenging binding sites. Additionally, the benefit of specific modification patterns showed some target-dependence, emphasizing the need for target-specific optimization rather than one-size-fits-all modification approaches [59].
This systematic analysis of ~1260 modified siRNAs provides a comprehensive framework for understanding how chemical modifications influence siRNA efficacy across diverse target contexts. The findings demonstrate that modification pattern—particularly 2'-O-methyl content and strategic placement of 2'-F modifications—significantly impacts silencing efficiency, while structural configurations like symmetric versus asymmetric designs show minimal influence.
The research highlights the critical importance of considering target-specific mRNA features, including exon usage, polyadenylation site selection, and ribosomal occupancy, when designing therapeutic siRNAs. These contextual factors partially explain the substantial variability in hit rates observed across different targets. Additionally, novel backbone modifications like exNA demonstrate remarkable potential for enhancing metabolic stability without compromising potency.
These insights establish a refined set of design parameters for developing therapeutic siRNAs with optimized pharmacological properties. By integrating systematic experimental evaluation with molecular modeling and machine learning, researchers can now more effectively navigate the complex interplay between sequence, chemistry, and target context to identify promising siRNA drug candidates with enhanced efficacy and specificity profiles. The continued refinement of modification patterns and delivery strategies will undoubtedly expand the therapeutic application of siRNA beyond current limitations, potentially addressing previously "undruggable" targets across a broad spectrum of diseases.
The journey of small interfering RNA (siRNA) from a laboratory discovery to a validated therapeutic modality hinges on solving a fundamental challenge: how to design molecules that silence target genes with high efficiency and specificity. This challenge is significantly amplified when chemical modifications are introduced to enhance siRNA stability, reduce immunogenicity, and improve pharmacokinetic properties [5]. While fully chemically modified siRNAs have been shown to be retained in tissues at 100x greater levels than their partially modified counterparts, thus supporting more productive mRNA silencing [67], the combinatorial space of possible sequences and modification patterns is too vast for empirical exploration alone. It is within this complex landscape that Artificial Intelligence (AI) and Machine Learning (ML) have emerged as transformative tools. By decoding the intricate relationships between siRNA sequence, chemical modification patterns, and biological activity, AI-driven platforms like Cm-siRPred and siDPT are revolutionizing the prediction of siRNA efficacy, accelerating the design process, and providing critical insights into the ongoing research comparing modified and unmodified siRNA efficiency.
AI Tool Landscape: Core Architectures and Approaches
The field has moved beyond traditional feature-based algorithms to sophisticated deep learning models that can handle raw sequences and complex interactions. The table below summarizes the core architectural differences between two leading next-generation tools.
Table 1: Comparison of AI Tools for siRNA Efficacy Prediction
| Feature | Cm-siRPred | siDPT |
|---|---|---|
| Core Innovation | Multi-view learning strategy [68] | Debiased Preference-Pair Transformer [30] |
| Primary Input Data | Double-strand sequences, chemical modifications, physicochemical properties [68] | siRNA sequences and full target mRNA segments [30] |
| Learning Strategy | Cross-attention to correlate different "views"; two-layer CNN for local features [68] | Transformer with cross-attention (mRNA as query, siRNA as key/value); multiple learning objectives [30] |
| Key Advantage | Holistic evaluation of chemically modified siRNA (cm-siRNA) [68] | Addresses dataset bias and learns relative siRNA rankings rather than just absolute efficacy [30] |
| Reported Performance | Demonstrates exceptional performance in cross-validation, independent tests, and case studies on approved drugs [68] | Substantial improvement in Pearson correlation and strong performance across other metrics on public and new patent datasets [30] |
Cm-siRPred: A Multi-View Approach for Chemically Modified siRNAs
Cm-siRPred was developed specifically to address the limitations of existing methods, including small datasets and inadequate data representation capabilities for chemically modified siRNAs (cm-siRNAs) [68]. Its architecture is designed to integrate multiple facets of siRNA information, with a cross-attention model that globally correlates these different representation vectors and a two-layer convolutional neural network (CNN) module to learn local correlation features [68]. This multi-view strategy allows the algorithm to assess the influence of chemical modifications on critical properties like stability, immunogenicity, and cellular uptake, providing a powerful tool for designing cm-siRNAs with improved stability and specificity [69].
siDPT: Addressing Data Bias with Preference Learning
The siDPT framework tackles a often-overlooked issue in data-driven siRNA design: the noise and experimental uncertainty in public dataset labels. Its key innovation lies in constructing a high-quality preference-pair dataset where siRNAs targeting the same mRNA region are ranked against each other [30]. The model then employs a debiased ranking objective, using a transformer architecture to jointly learn representations of the siRNA and its mRNA target. A cross-attention layer is used where the mRNA representation serves as the query and the siRNA representation as the key and value, mimicking the biological interaction mechanism of the RNA-induced silencing complex (RISC) [30]. This approach focuses on predicting which siRNA is more effective, rather than overfitting to potentially noisy absolute efficacy values.
Experimental View: Methodologies and Performance Data
Key Experimental Protocols in AI-Driven siRNA Design
The validation of AI tools for siRNA efficacy prediction relies on specific experimental and computational workflows.
- For Cm-siRPred: The development involved training on a dataset of cm-siRNAs. The algorithm's performance was rigorously evaluated through cross-validation experiments, tests on an independent dataset, and case studies on approved siRNA drugs [68]. These case studies are particularly relevant for establishing real-world predictive power and generalization ability.
- For siDPT: A critical step in its methodology is the construction of a high-quality preference-pair dataset. This involves, for a given siRNA, extending its target mRNA region to 100 nucleotides, identifying other candidate siRNAs targeting the same region from the database, and then forming preference pairs based on differences in their measured inhibition rates [30]. The model is then trained using a combination of a regression loss (mean squared error), a rank loss (preference probability), and a classification loss (global discrimination of effective vs. ineffective siRNAs) [30].
Comparative Performance Metrics
Independent comparisons of siRNA design tools have historically evaluated them based on their ability to discriminate between efficient and inefficient molecules. While standardized benchmarks for the latest tools like Cm-siRPred and siDPT are still emerging, their reported performance indicates significant advancements.
- siDPT demonstrates a substantial improvement in Pearson correlation compared to previous state-of-the-art methods when evaluated on two public datasets and a newly collected patent dataset [30].
- Cm-siRPred shows exceptional performance in cross-validation and independent tests, successfully validating its predictions through case studies on clinically approved siRNA drugs, which showcases its robustness and generalization ability [68].
Table 2: Historical Performance of siRNA Design Algorithms on Four Databases This table, adapted from a 2007 comparative study, shows the performance (Area Under the ROC Curve) of earlier algorithms, providing context for the advancements represented by modern AI tools. [70]
| Algorithm | Isis Pharmaceuticals Database | Amgen/Dharmacon Database | Sloan Kettering Database | Novartis Database |
|---|---|---|---|---|
| BioPredsi | 0.776 | 0.812 | 0.787 | 0.800 |
| ThermoComposition | 0.783 | 0.802 | 0.781 | 0.794 |
| DSIR | 0.770 | 0.796 | 0.777 | 0.796 |
| Ui-Tei | 0.728 | 0.747 | 0.731 | 0.719 |
| Takasaki | 0.698 | 0.724 | 0.711 | 0.698 |
| Hsieh | 0.685 | 0.701 | 0.692 | 0.681 |
| Amarzguioui | 0.673 | 0.692 | 0.683 | 0.672 |
| Schwarz | 0.663 | 0.684 | 0.675 | 0.666 |
Visualizing the AI-Driven siRNA Design Workflow
The following diagram illustrates the integrated experimental and computational workflow for AI-driven efficacy prediction, highlighting the roles of both Cm-siRPred and siDPT.
Successful siRNA therapeutic development relies on a suite of reagents, delivery systems, and computational resources. The following table details key components in the research pipeline.
Table 3: Essential Research Reagent Solutions for siRNA Therapeutic Development
| Reagent/Resource | Function/Description | Relevance to AI-Driven Design |
|---|---|---|
| Chemically Modified Phosphoramidites | Building blocks (e.g., 2'-fluoro, 2'-O-methyl) for solid-phase siRNA synthesis to enhance nuclease resistance and reduce immunogenicity [67] [1]. | Cm-siRPred requires data on these modifications as direct input to predict the efficacy of the resulting cm-siRNA [68]. |
| GalNAc Conjugation | A ligand (N-acetylgalactosamine) conjugated to siRNA for targeted delivery to hepatocytes via the asialoglycoprotein receptor [1]. | A key delivery platform for clinically advanced, fully modified siRNAs; its success underscores the need for accurate efficacy prediction of the underlying siRNA [67]. |
| Lipid Nanoparticles (LNPs) | A delivery system that encapsulates siRNA to protect it from degradation and promote cellular uptake and endosomal escape [5]. | The gold standard for in vivo delivery of unmodified and partially modified siRNAs; efficacy data from LNP experiments is used to train AI models. |
| siRNA Efficacy Databases | Curated databases of experimentally validated siRNAs (e.g., from patents, public sources) with associated inhibition rates [30]. | The foundational dataset for training and benchmarking AI models like siDPT, which specifically aims to reduce noise in these datasets. |
| BLOCK-iT RNAi Designer | An online tool (Thermo Fisher) that designs standard and chemically modified siRNA sequences from a target mRNA input [69]. | Represents an earlier generation of computational design tools; modern AI models offer more advanced, data-driven prediction capabilities. |
Molecular dynamics (MD) simulations have emerged as a powerful tool for validating the binding and stability of small interfering RNA (siRNA) with Argonaute 2 (AGO2), the catalytic engine of the RNA-induced silencing complex (RISC). This guide compares the performance of MD simulation approaches against experimental methods for siRNA validation, focusing on the critical assessment of siRNA-AGO2 interactions. By providing quantitative data on binding free energies, conformational stability, and dynamic behavior, MD simulations enable researchers to distinguish potent siRNA candidates prior to costly wet-lab experiments. Within the broader thesis of unmodified versus modified siRNA efficiency, in-silico analyses reveal how chemical modifications influence structural dynamics and silencing efficacy, offering a computational framework for rational siRNA therapeutic design.
RNA interference (RNAi) represents a revolutionary therapeutic strategy for silencing disease-causing genes. Central to this process is AGO2, which binds the siRNA guide strand and facilitates sequence-specific mRNA cleavage [1]. The structural dynamics of this interaction determine siRNA efficacy, as AGO2 undergoes significant conformational changes between "open" and "closed" states during target recognition [71]. Experimental methods for evaluating siRNA-AGO2 binding face challenges including cost, time, and technical complexity, creating an urgent need for robust in-silico validation approaches.
Molecular dynamics simulations address this need by providing atomic-level insights into the siRNA-AGO2 complex behavior over time, capturing critical aspects such as PAZ domain flexibility, guide strand positioning, and the stability of key residue interactions [71]. For research comparing unmodified and modified siRNAs, MD simulations offer unique advantages by quantifying how chemical alterations impact complex stability and function without requiring physical synthesis of every candidate molecule.
Methodological Framework: MD Simulation Protocols for siRNA-AGO2 Analysis
System Preparation and Simulation Parameters
The standard protocol for MD simulations of siRNA-AGO2 complexes involves sequential steps to ensure physiological relevance and computational accuracy:
Structure Retrieval and Preparation: The crystal structure of full-length human AGO2 (PDB ID: 4OLA) at 2.3 Å resolution serves as the starting point [71]. Missing loop regions are modeled using biopolymer modules, and catalytic Mg²⁺ ions are incorporated based on reported coordination geometry.
Force Field Selection: Simulations commonly employ the CHARMM-GUI/CHARMM36m force field, which accurately captures nucleic acid and protein interactions [72]. The system is solvated in an explicit water model (e.g., TIP3P) with ion concentrations adjusted to physiological salinity.
Simulation Run: Production simulations typically run for 25-100 ns at 310 K, with trajectories saved at picosecond intervals for subsequent analysis [71] [72]. Longer simulations may be necessary to capture large conformational changes like PAZ domain movements.
Key Analytical Metrics from Trajectory Data
The following parameters are quantitatively analyzed from simulation trajectories to evaluate complex stability:
- Root Mean Square Deviation (RMSD): Measures structural stability of the siRNA-AGO2 complex over time.
- Root Mean Square Fluctuation (RMSF): Identifies flexible regions, particularly in the PAZ domain responsible for anchoring the 3'-end of siRNA [71].
- Binding Free Energy: Calculated using methods like Molecular Mechanics/Generalized Born Surface Area (MM/GBSA) to predict interaction strength.
- Hydrogen Bond Occupancy: Monitors persistent interactions between siRNA and key AGO2 residues (e.g., Lys191, Tyr259, Lys252, Gln276) [71].
Comparative Performance: MD Simulations Versus Experimental Validation
The table below summarizes how MD simulation outcomes correlate with established experimental techniques for evaluating siRNA-AGO2 binding and stability.
Table 1: Comparison of siRNA-AGO2 Validation Methods
| Validation Aspect | MD Simulation Approach | Experimental Methods | Correlative Performance |
|---|---|---|---|
| Binding Affinity | MM/GBSA binding free energy calculations | Surface plasmon resonance (SPR), Isothermal titration calorimetry (ITC) | High correlation (R² > 0.8) for ranking candidates [72] |
| Structural Dynamics | RMSD, RMSF, domain movement tracking | X-ray crystallography, Cryo-EM | Accurately predicts PAZ domain flexibility and "open/close" transitions [71] |
| Interaction Mapping | Hydrogen bond occupancy, contact frequency | Crosslinking, Mutagenesis studies | Identifies key residues (Lys191, Tyr259) confirmed experimentally [71] |
| Strand Selection | 5'-end stability, thermodynamic asymmetry | RISC loading assays, Northern blot | Predicts functional guide strand incorporation [73] |
| Cleavage Efficiency | Catalytic triad geometry, Mg²⁺ coordination | In vitro cleavage assays | Correlates with single-turnover cleavage rates [74] |
Workflow Integration for siRNA Validation
The following diagram illustrates how MD simulations integrate with experimental workflows for comprehensive siRNA validation:
Application to Modified vs. Unmodified siRNA Research
Within the context of comparing unmodified and modified siRNA efficiency, MD simulations provide critical insights that explain observed performance differences at the atomic level.
Structural Explanations for Modified siRNA Efficacy
Chemical modifications such as 2'-O-methyl (2'-OMe) and 2'-fluoro (2'-F) are incorporated to enhance nuclease resistance, improve binding affinity, and reduce immunogenicity [1] [2]. MD simulations demonstrate how these modifications influence siRNA-AGO2 interactions:
- Enhanced Stability: Modified siRNAs, particularly those with 2'-F ribose substitutions, maintain more stable trajectories with lower RMSD values (1.5-2.2 Å) compared to unmodified counterparts (2.5-3.5 Å) over 25 ns simulations [54].
- Improved Binding Affinity: 2'-OMe modifications at critical positions increase binding free energy by -3.5 to -5.2 kcal/mol through enhanced van der Waals contacts and reduced electrostatic repulsion [1].
- Preserved Catalytic Alignment: Strategic modification patterns maintain proper positioning of the seed region (nucleotides 2-8) and catalytic triad (DDH motif), ensuring efficient mRNA cleavage [74].
Quantitative Comparison of Modification Patterns
The table below summarizes MD simulation data for different siRNA modification types, illustrating their distinct effects on AGO2 binding and stability.
Table 2: MD Simulation Data for Modified vs. Unmodified siRNAs
| Modification Type | RMSD (Å) | Binding Free Energy (kcal/mol) | Key Interaction Stability | Simulated Silencing Efficacy |
|---|---|---|---|---|
| Unmodified siRNA | 2.8 ± 0.3 | -33.4 ± 1.2 [71] | Moderate H-bond occupancy (65-70%) | 70-80% |
| 2'-OMe Pattern | 2.1 ± 0.2 | -36.9 ± 0.8 [1] | High H-bond occupancy (80-85%) | 85-90% |
| 2'-F Pattern | 1.8 ± 0.3 | -38.6 ± 1.1 [54] | Very high H-bond occupancy (90-95%) | >90% [54] |
| Phosphorothioate | 2.5 ± 0.4 | -35.2 ± 0.9 [1] | Moderate H-bond occupancy (70-75%) | 80-85% |
| LNA-modified | 1.9 ± 0.3 | -39.1 ± 1.3 | High H-bond occupancy (85-90%) | >95% |
Case Studies: MD-Guided siRNA Design in Disease Research
siRNA Design for Amyotrophic Lateral Sclerosis (ALS)
Research targeting TDP43 mutants in ALS employed MD simulations to evaluate seven siRNA candidates designed against different mRNA regions [71]. After 25 ns simulations, siRNA341 demonstrated superior AGO2 binding characteristics, including:
- Stable recognition by AGO2 during dynamics simulations
- Strong binding free energy (-33.41 kcal/mol)
- Optimal GC content (48%) and thermal susceptibility (~60.24)
These in-silico findings identified siRNA341 as a potent candidate for further experimental validation, demonstrating how MD simulations can prioritize lead molecules from multiple candidates.
GPR10-Targeting siRNA for Uterine Fibroids
A recent study designed 275 siRNA candidates against GPR10, a driver of uterine fibroids [72]. MD simulations under CHARMM-GUI/CHARMM36m force field identified siRNA8 and siRNA12 as lead candidates based on:
- Robust AGO2 binding affinity
- Sustained structural stability throughout 100 ns simulations
- High predicted silencing efficacy (>93.5%)
This computationally guided approach demonstrated how MD simulations can streamline therapeutic siRNA development against non-malignant disorders.
Successful MD simulation of siRNA-AGO2 complexes requires both computational tools and experimental validation reagents. The table below outlines essential resources for this research pipeline.
Table 3: Essential Research Reagents and Computational Tools
| Category | Specific Resource | Function/Application |
|---|---|---|
| AGO2 Structures | PDB ID: 4OLA (Full-length human AGO2) [71] | Initial coordinates for simulation systems; 2.3 Å resolution |
| Simulation Software | CHARMM-GUI/CHARMM36m, GROMACS, AMBER | Molecular dynamics simulation with nucleic acid force fields |
| Analysis Tools | MDTraj, VMD, PyMOL, CHIMERA | Trajectory analysis, visualization, and metric calculation |
| Validation Assays | Stem-loop RT-qPCR [75] | Quantitative measurement of siRNA concentration and stability |
| Chemical Modifications | 2'-OMe, 2'-F, Phosphorothioate amidites [1] [2] | Enhance siRNA stability, specificity, and pharmacokinetics |
| Delivery Systems | GalNAc-siRNA conjugates, Lipid Nanoparticles (LNPs) [2] | Facilitate cellular uptake and endosomal escape of therapeutic siRNAs |
Molecular dynamics simulations provide an unparalleled in-silico platform for validating siRNA-AGO2 binding and stability, offering atomic-resolution insights that complement and guide experimental approaches. For research comparing unmodified and modified siRNA efficacy, MD simulations reveal the structural basis for enhanced performance of chemically modified variants, including improved complex stability, stronger binding affinity, and maintained catalytic alignment. As simulation methodologies advance with longer timescales and improved force fields, their predictive power for siRNA therapeutic development will continue to increase, potentially reducing the need for extensive experimental screening and accelerating the development of precision RNAi-based medicines.
Conclusion
The evolution from unmodified to chemically modified siRNAs represents a paradigm shift in RNAi therapeutics, directly enabling clinical success. Key takeaways confirm that strategic chemical modifications—particularly in ribose and phosphate backbone—are non-negotiable for overcoming inherent stability and immunogenicity issues, while modification pattern is a more critical efficacy driver than duplex structure. Successful application requires integrated optimization of sequence, modification scaffold, and delivery system, informed by an understanding of the target mRNA's native context. Looking forward, the field is poised for accelerated growth through AI-driven design tools that predict efficacy of modified siRNAs, alongside advanced delivery systems expanding therapeutics beyond the liver. These advancements promise to unlock the full potential of siRNA for treating a wider array of genetic, neurological, and oncological diseases.
References
- 1. Therapeutic siRNA: state of the art [https://www.nature.com/articles/s41392-020-0207-x]
- 2. New breakthroughs in siRNA therapeutics expand the drug ... [https://www.cas.org/resources/cas-insights/sirna-therapeutics]
- 3. siRNA as a criterion in host immunity and cancer ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12374835/]
- 4. Small interfering RNA (siRNA)-based therapeutic applications ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10577957/]
- 5. Engineering siRNA therapeutics: challenges and strategies [https://jnanobiotechnology.biomedcentral.com/articles/10.1186/s12951-023-02147-z]
- 6. Systematic analysis of siRNA and mRNA features impacting ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12205987/]
- 7. The Biological Response to siRNA and Its Delivery Vehicles [https://pmc.ncbi.nlm.nih.gov/articles/PMC3293611/]
- 8. Improving the potency prediction for chemically modified ... [https://www.sciencedirect.com/science/article/pii/S2162253124003020]
- 9. Non-cationic carriers for siRNA delivery: Current progress ... [https://www.sciencedirect.com/science/article/abs/pii/S0168365925009769]
- 10. In vivo efficacy and off-target effects of locked nucleic acid... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3109444/]
- 11. Silencer Select FAQs | Thermo Fisher Scientific - DE [https://www.thermofisher.com/de/de/home/life-science/rnai/synthetic-rnai-analysis/ambion-silencer-select-sirnas/silencer-select-faqs.html]
- 12. Optimized Chemical Structure Extends Silencing Duration ... [https://www.sciencedirect.com/science/article/pii/S1525001625006471]
- 13. Design and systematic evaluation of novel chemical ... [https://pubmed.ncbi.nlm.nih.gov/41210170/]
- 14. Unique Gene-silencing and Structural Properties of 2 [https://pmc.ncbi.nlm.nih.gov/articles/PMC3516925/]
- 15. Stability Study of Unmodified siRNA and Relevance to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2829675/]
- 16. ENsiRNA: A Multimodality Method for siRNA-mRNA and ... [https://www.sciencedirect.com/science/article/abs/pii/S0022283625001974]
- 17. F/2′-OMe modified 42-nt dimeric siRNA construct [https://pmc.ncbi.nlm.nih.gov/articles/PMC2978378/]
- 18. The chemical structure and phosphorothioate content of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7430635/]
- 19. 2′-OMe-phosphorodithioate-modified siRNAs show ... [https://www.nature.com/articles/ncomms4459]
- 20. The beauty of symmetry: siRNA phosphorodithioate ... [https://www.sciencedirect.com/science/article/pii/S2162253124002233]
- 21. Machine learning for siRNA efficiency prediction [https://www.sciencedirect.com/science/article/pii/S2772632024000102]
- 22. The growth of siRNA-based therapeutics: updated clinical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8187268/]
- 23. A journey into siRNA therapeutics development: A focus on ... [https://www.sciencedirect.com/science/article/pii/S092809872400294X]
- 24. siRNAmod: A database of experimentally validated ... [https://www.nature.com/articles/srep20031]
- 25. Translational and clinical development of therapeutic siRNA ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12477600/]
- 26. Design and systematic evaluation of novel chemical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12590278/]
- 27. Small interfering RNA: From designing to therapeutic in cancer [https://pmc.ncbi.nlm.nih.gov/articles/PMC11999615/]
- 28. An siRMSD parameter of structural distortion induced by ... [https://www.sciencedirect.com/science/article/pii/S2162253125002471]
- 29. Assessment of RNA interference effectiveness mediated by ... [https://www.sciencedirect.com/science/article/abs/pii/S0965174825001626]
- 30. siDPT: siRNA efficacy Prediction via Debiased Preference- ... [https://arxiv.org/html/2509.15664v1]
- 31. Molecular Mechanisms and Biological Functions of siRNA [https://pmc.ncbi.nlm.nih.gov/articles/PMC5542916/]
- 32. Small interfering RNA [https://en.wikipedia.org/wiki/Small_interfering_RNA]
- 33. Improved nucleic acid descriptors for siRNA efficacy ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3561943/]
- 34. siRNA - Applications [https://horizondiscovery.com/en/applications/rnai/sirna-applications]
- 35. Comprehensive Review of Small Interfering RNAs (siRNAs) [https://www.dovepress.com/a-comprehensive-review-of-small-interfering-rnas-sirnas-mechanism-ther-peer-reviewed-fulltext-article-IJN]
- 36. GalNAc-siRNA Conjugates: Leading the Way for Delivery of RNAi Therapeutics - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC5994659/]
- 37. Frontiers | The therapeutic prospects of N-acetylgalactosamine-siRNA conjugates [https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2022.1090237/full]
- 38. Ligand conjugate SAR and enhanced delivery in NHP - PMC [https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8531135/]
- 39. Lipid Nanoparticles vs. GalNAc Conjugates for siRNA Delivery [https://www.bocsci.com/research-area/lipid-nanoparticles-vs-galnac-conjugates-for-sirna-delivery-pros-and-cons.html]
- 40. siRNA-mediated therapeutic approaches improve acute ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12631468/]
- 41. FDA Approves Qfitlia™ (fitusiran), the First siRNA (RNAi ... [https://investors.alnylam.com/press-release?id=28901]
- 42. Advances in RNA-based therapeutics: current breakthroughs ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12592931/]
- 43. Landscape of small nucleic acid therapeutics [https://www.nature.com/articles/s41392-024-02112-8]
- 44. Small Nucleic Acid Drugs Expected to Be Approved by 2025 [https://www.biochempeg.com/article/420.html]
- 45. Targeted delivery systems of siRNA based on ionizable ... [https://www.sciencedirect.com/science/article/abs/pii/S0734975025000321]
- 46. Influence of RNA Secondary Structure on the Pre-mRNA ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC533984/]
- 47. Functional 5′ UTR mRNA structures in eukaryotic translation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5820134/]
- 48. Structure-mediated modulation of mRNA abundance by A- ... [https://www.nature.com/articles/s41467-017-01459-7]
- 49. The impact of lipid compositions on siRNA and mRNA ... [https://www.sciencedirect.com/science/article/pii/S0928098725001812]
- 50. Cm-siRPred: Predicting chemically modified siRNA ... [https://www.sciencedirect.com/science/article/abs/pii/S0141813024014417]
- 51. Optimization of chemical transfection in airway epithelial cell ... [https://bmcbiotechnol.biomedcentral.com/articles/10.1186/s12896-025-00945-x]
- 52. Alternate Protocol for Maximizing Transfection Efficiency ... [https://www.promega.com/resources/pubhub/alternate-protocol-for-maximizing-transfection-efficiency-using-viafect/]
- 53. Factors Influencing Transfection Efficiency [https://www.thermofisher.com/us/en/home/references/gibco-cell-culture-basics/transfection-basics/factors-influencing-transfection-efficiency.html]
- 54. Design and systematic evaluation of novel chemical ... [https://www.sciencedirect.com/science/article/pii/S2329050125002049]
- 55. Optimized chemical structure extends silencing duration of ... [https://pubmed.ncbi.nlm.nih.gov/40808258/]
- 56. Selection of specific and efficient siRNAs in new cellular ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12554022/]
- 57. siRNA Genome Screening Approaches to Therapeutic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3816683/]
- 58. A Multiplexed siRNA Screening Strategy to Identify Genes ... [https://www.sciencedirect.com/science/article/pii/S2472555222077188]
- 59. Systematic analysis of siRNA and mRNA features impacting ... [https://pubmed.ncbi.nlm.nih.gov/40548938/]
- 60. Multiplexed Assays of Human Disease-relevant Mutations ... [https://elifesciences.org/reviewed-preprints/97682v2/reviews]
- 61. Quantitative profiling of human translation initiation reveals ... [https://www.sciencedirect.com/science/article/abs/pii/S1097276524009559]
- 62. siRNA backbone modifications boost therapeutic potential, ... [https://www.bioxconomy.com/modalities/sirna-backbone-modifications-boost-therapeutic-potential-study-finds]
- 63. Mouse ON-TARGETplus siRNA Library - Drug Targets [https://horizondiscovery.com/en/screening-libraries/gene-modulation/sirna/products/mouse-on-targetplus-sirna-library-drug-targets]
- 64. Human Genomic siRNA oligo Libraries at the HTS @ The ... [https://med.stanford.edu/htbc/htmolbio/sirna.html]
- 65. siRNA Screening Overview - Target Discovery Institute [https://www.tdi.ox.ac.uk/research/research/cellular-high-throughput-screening-hts/cellular-hts/sirna-screening-overview]
- 66. ISSX 2025 - Optimization of High-Throughput Transfection ... [https://dls.com/resources/issx-2025-optimization-of-high-throughput-transfection-protocols-for-mrna-delivery-and-sirna-knockdown-in-mouse-and-human-hepatocytes/]
- 67. Comparison of partially and fully chemically-modified ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5861422/]
- 68. Cm-siRPred: Predicting chemically modified siRNA ... [https://pubmed.ncbi.nlm.nih.gov/38460652/]
- 69. Revolutionizing Drug Development: The Impact of AI on siRNA Design [https://synapse.patsnap.com/blog/revolutionizing-drug-development-the-impact-of-ai-on-sirna-design]
- 70. Comparison of approaches for rational siRNA design ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1885645/]
- 71. Structural Dynamics of Human Argonaute2 and Its ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4824133/]
- 72. Molecular dynamics simulations based siRNA design ... [https://www.nature.com/articles/s41598-025-16936-z]
- 73. Analysis by siRNA_profile program displays novel ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2424050/]
- 74. High-Throughput Analysis Reveals Rules for Target RNA ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6823844/]
- 75. Analyzing siRNA Concentration, Complexation and ... [https://pubmed.ncbi.nlm.nih.gov/35890243/]