From Microscope to Machine: A Comprehensive Analysis of Traditional vs. AI-Based Sperm Morphology Assessment in Biomedical Research
This article provides a critical analysis for researchers and drug development professionals on the paradigm shift from traditional to AI-based sperm morphology assessment.
From Microscope to Machine: A Comprehensive Analysis of Traditional vs. AI-Based Sperm Morphology Assessment in Biomedical Research
Abstract
This article provides a critical analysis for researchers and drug development professionals on the paradigm shift from traditional to AI-based sperm morphology assessment. We explore the foundational principles of manual semen analysis and its inherent limitations, including subjectivity and high inter-observer variability. The methodological section delves into cutting-edge AI approaches, from conventional machine learning to advanced deep learning architectures like CBAM-enhanced ResNet50, which achieve over 96% accuracy. The discussion extends to troubleshooting dataset limitations and optimizing model performance, followed by rigorous validation metrics and clinical correlation studies. By synthesizing performance data, adoption trends, and future trajectories, this review serves as a technical roadmap for integrating AI-driven solutions into reproductive research and diagnostics.
The Foundation of Sperm Morphology Analysis: Principles, Limitations, and Clinical Significance
Sperm morphology assessment, the analysis of sperm size, shape, and appearance, constitutes a fundamental diagnostic component within male fertility evaluation. These analyses provide crucial insights into spermatogenesis and sperm function, informing clinical decisions for natural conception and assisted reproductive technologies (ART). For decades, traditional assessment protocols, primarily guided by the World Health Organization (WHO) laboratory manual, have established the global standard for methodology and interpretation. The inherent subjectivity and significant inter-laboratory variability of these manual techniques present considerable challenges to diagnostic consistency and clinical utility. This document details the established protocols, guidelines, and limitations of traditional sperm morphology assessment, providing a essential foundational context for the emerging paradigm of AI-based analysis.
Core WHO Guidelines and Standardized Methodology
The WHO laboratory manual serves as the principal reference for standardizing semen analysis, ensuring comparability of results across different laboratories globally. The sixth edition, published in 2021, outlines evidence-based procedures for the routine examination and processing of human semen [1].
Key Principles and Analytical Goals
The manual is designed to maintain and sustain the quality of analysis, supporting universal access to sexual and reproductive health care services. It provides detailed protocols for routine tests, with sperm morphology analysis being an integral part of the basic semen examination. The primary analytical goals are:
- Diagnostic Aid: Investigating male fertility status during an infertility workup.
- Research Tool: Monitoring spermatogenesis in clinical studies and following interventions.
- Clinical Prognostication: Providing parameters that may influence the choice of ART procedure.
A central tenet of the WHO guideline is that laboratories should establish their own reference ranges based on their specific population and methodologies, acknowledging that results can vary due to preparation techniques and staining choices [2].
Recent Re-evaluations: The BLEFCO 2025 Guidelines
A recent expert review from the French BLEFCO Group has prompted a significant re-evaluation of long-standing practices. Published in 2025, these guidelines challenge the clinical value of certain traditional assessments, suggesting a move towards simplification [3]. Their key recommendations are summarized in the table below.
Table 1: Key Recommendations from the BLEFCO 2025 Guidelines on Sperm Morphology Assessment
| Recommendation | Description | Key Rationale |
|---|---|---|
| R1: Against Detailed Analysis | Does not recommend systematic detailed analysis of individual abnormality groups during routine assessment. | Aims to simplify reporting and reduce unnecessary complexity. |
| R2: For Monomorphic Defects | Recommends qualitative or quantitative methods for detecting specific monomorphic syndromes (e.g., globozoospermia). | Critical for accurate diagnosis of severe conditions that require specific clinical management. |
| R3: Against Defect Indexes | Does not recommend the use of Teratozoospermia Index (TZI), Sperm Deformity Index (SDI), or Multiple Anomalies Index (MAI). | Insufficient evidence to demonstrate clinical utility in infertility investigation or before ART. |
| R4: For Automated Systems | Gives a positive opinion on qualified and validated automated systems based on cytological analysis after staining. | Recognizes the potential for technology to improve standardization. |
| R5: Against Prognostic Use for ART | Does not recommend using the percentage of normal forms as a prognostic criterion for selecting between IUI, IVF, or ICSI. | Challenges current practice; the overall level of evidence is low. |
Detailed Experimental Protocols for Sperm Morphology Assessment
The following section outlines the core technical workflow and methodologies prescribed for traditional sperm morphology assessment.
The process, from sample collection to final interpretation, involves multiple critical steps to ensure analytical integrity. The following diagram illustrates the complete experimental workflow.
Critical Procedural Steps
1. Sample Preparation and Staining: Sperm smears are prepared from liquefied semen and fixed for at least 15 minutes in 95% ethanol (v/v). The Papanicolaou staining method is the recommended and most widely used technique [2]. This multi-step process involves:
- Rehydration: Sequential immersion in 80% and 50% ethanol, followed by purified water.
- Nuclear Staining: Using Harris's hematoxylin for approximately 4 minutes to stain the sperm nucleus.
- Cytoplasmic Staining: Using OG-6 orange and EA-50 green to stain the cytoplasm and acrosomal region.
- Dehydration and Mounting: Final dehydration in absolute ethanol, clearing in xylene, and mounting with a coverslip [2].
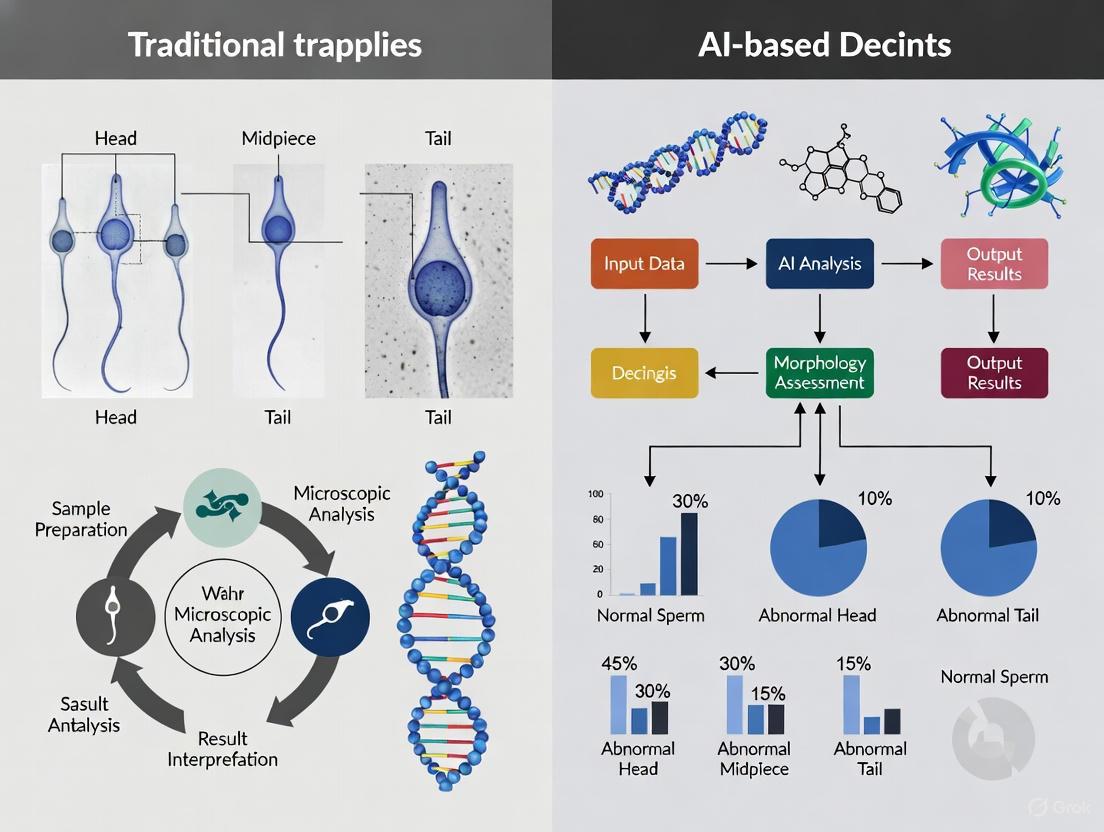
2. Microscopic Examination and Classification: Stained slides are examined under a brightfield microscope using a 100x oil immersion objective. According to WHO standards, a minimum of 200 spermatozoa should be assessed and classified [4]. The classification system is structured around the sperm's anatomical components:
- Head: Abnormalities include large, small, tapered, pyriform, round, amorphous, vacuolated (>20% of head area), or acrosome abnormalities.
- Midpiece: Abnormalities include asymmetric, thick, or thin insertion, or any broken or bent segment.
- Tail: Abnormalities include short, multiple, hairpin, broken, or bent tails.
- Excess Residual Cytoplasm: A common abnormality indicating faulty spermiogenesis.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Reagents and Materials for Traditional Sperm Morphology Assessment
| Item | Function / Application |
|---|---|
| Papanicolaou Stain Set | A multi-component stain (Hematoxylin, OG-6, EA-50) for differential staining of sperm head (nucleus and acrosome) and cytoplasmic components. Essential for detailed morphological analysis per WHO guidelines [2]. |
| 95% Ethanol (v/v) | Primary fixative for sperm smears; preserves cellular morphology and prevents degeneration prior to staining [2]. |
| Olympus CX43 Microscope | An example of a standard upright microscope equipped with a 100x oil immersion objective, essential for high-resolution imaging of spermatozoa at the required magnification [2]. |
| Microscope Camera (CMOS) | For capturing digital images of sperm for analysis, documentation, or training purposes. Specifications often include a resolution of 1920x1200 and a high frame rate for clarity [2]. |
| SSA-II Plus CASA System | An example of a Computer-Assisted Sperm Analysis system. While incorporating automation, it is used here in the context of a standardized tool to reduce subjective error in measurement, not as an AI-based system [2]. |
Quantitative Data and Reference Values
Establishing reference values is a persistent challenge in sperm morphology. The following table presents quantitative data from a 2025 study that established morphological parameters in a proven fertile population using standardized Papanicolaou staining and a CASA system for precise measurement.
Table 3: Sperm Morphological Parameters in a Fertile Population (Papanicolaou Staining) [2]
| Parameter (Abbreviation, Unit) | Description | Reference Value |
|---|---|---|
| Normal Head Morphology (%) | Percentage of sperm with morphologically normal heads. | 9.98% |
| Head Length (HL, μm) | Distance between the two furthest points along the long axis. | Provided (Precise values in source) |
| Head Width (HW, μm) | Perpendicular distance between the two furthest points on the short axis. | Provided (Precise values in source) |
| Head Area (HA, μm²) | Calculated area based on the contour of the sperm head. | Provided (Precise values in source) |
| Head Perimeter (HP, μm) | Length of the boundary surrounding the sperm head. | Provided (Precise values in source) |
| Ellipticity (L/W) | Ratio of the head length to the head width. | Provided (Precise values in source) |
| Acrosome Area (AcA, μm²) | Area of the cap-like acrosomal structure on the head. | Provided (Precise values in source) |
| Acrosome Ratio (AcR, %) | Ratio of the acrosome area to the head area. | Provided (Precise values in source) |
This study highlights that even in fertile men, the percentage of sperm with normal morphology is low, and it underscores the move towards more precise, quantitative morphometrics over subjective classification.
Limitations and Challenges of Traditional Methods
The traditional assessment framework is fraught with limitations that impact its diagnostic reliability.
- High Subjectivity and Inter-Observer Variability: The classification of sperm morphology is inherently subjective. Studies show significant disagreement even among experienced technicians, with one study noting experts agreed on only 73% of sperm images for a simple normal/abnormal classification [5].
- Lack of Standardized Training: There is no widely accepted method to train or standardize morphologists, which is a primary contributor to result variation [5]. A 2025 study demonstrated that without standardized training, novice morphologists showed high variation (Coefficient of Variation = 0.28) and low accuracy (53%-81% depending on classification complexity) [5].
- Questioned Clinical Utility: As reflected in the 2025 BLEFCO guidelines, the prognostic value of sperm morphology for selecting ART procedures (IUI, IVF, or ICSI) is now strongly questioned. The overall level of evidence is low, challenging its routine clinical application [3].
- Labor-Intensive and Low-Throughput: Manual assessment of 200+ sperm per sample is time-consuming and inefficient, creating a bottleneck in high-volume clinical or research settings [4].
Traditional sperm morphology assessment, as defined by WHO guidelines and standard laboratory practices, has provided a critical, albeit imperfect, foundation for male infertility diagnosis. Its core limitations—subjectivity, variability, and labor-intensive processes—have been rigorously documented. The recent BLEFCO guidelines signal a paradigm shift towards a simplified approach, de-emphasizing the prognostic value of detailed abnormality counts and indexes.
These acknowledged weaknesses create a clear mandate for innovation. The future of sperm morphology analysis lies in addressing these challenges through automation, standardization, and quantitative precision. This context directly paves the way for AI and deep learning-based approaches, which promise to overcome the inherent limitations of traditional methods by providing objective, high-throughput, and highly accurate analyses, ultimately enhancing diagnostic reliability for researchers, scientists, and clinicians in the field of reproductive medicine.
Semen analysis constitutes the foundational step in evaluating male fertility, with sperm morphology—the assessment of sperm size, shape, and structural characteristics—serving as a critical prognostic indicator for assisted reproductive technology (ART) outcomes [6]. Accurate morphology evaluation is essential because normal sperm morphology is strongly correlated with intact DNA and favorable clinical results, whereas abnormal morphology (teratozoospermia) is associated with reduced fertilization rates and poor embryo development [6] [7]. The World Health Organization (WHO) has established strict criteria for classifying normal sperm morphology: an oval head (length: 4.0–5.5 μm, width: 2.5–3.5 μm), an intact acrosome covering 40–70% of the head, and a single, uniform tail approximately 45 μm long without defects [6] [8]. Despite these standardized guidelines, the manual assessment of sperm morphology remains fraught with subjectivity, making it one of the most challenging and controversial parameters in semen analysis [6].
This technical guide examines the inherent limitations of manual sperm morphology analysis within the broader thesis of traditional versus AI-based assessment methodologies. For researchers and drug development professionals, understanding these limitations is paramount for developing standardized, objective approaches that can improve diagnostic consistency across laboratories and enhance the predictive value of sperm morphology for clinical outcomes.
Quantifying Subjectivity: The Evidence Base
The subjectivity inherent in manual sperm morphology assessment manifests quantitatively as significant inter-observer variability, even among trained technicians following WHO protocols. This variability undermines the reliability of fertility diagnostics and subsequent treatment decisions.
Statistical Evidence of Variability
A 2023 observational study conducted at a tertiary care institution provides compelling quantitative evidence of these limitations. The study evaluated inter-observer variability between a trained andrology technician and two academic residents by analyzing semen samples from 28 subjects. All three examiners assessed the same samples for sperm concentration, motility, vitality, and morphology according to WHO recommendations [9].
Table 1: Coefficient of Variation (CV) in Manual Semen Analysis Parameters
| Semen Parameter | Mean CV (%) | Range of CV (%) | Intraclass Correlation Coefficient (ICC) |
|---|---|---|---|
| Sperm Concentration | 6.24 | 1.2 - 23.02 | 0.982 (0.967-0.991) |
| Sperm Vitality | 10.14 | 3.68 - 26.24 | 0.955 (0.916-0.978) |
| Sperm Morphology | 2.66 | 1.05 - 5.75 | 0.490 (0.045-0.747) |
| Sperm Motility | 8.11 | 4.35 - 15.48 | 0.971 (0.945-0.986) |
The data reveals notably low inter-observer agreement for sperm morphology assessment, as evidenced by the disconcertingly low ICC of 0.490 (95% CI: 0.045-0.747) compared to other parameters [9]. While morphology demonstrated the lowest mean coefficient of variation (2.66%), this paradoxically high agreement may indicate consistent misclassification among observers rather than true precision—a phenomenon potentially reflecting systematic bias rather than reliable assessment [9].
Control chart analysis from the same study identified one measurement in sperm morphology that fell outside the statistical action control limits, with additional parameters exceeding warning limits, indicating significant deviations from expected values [9]. Bland-Altman plot analysis further confirmed substantial differences in sperm morphology assessments between observer pairs, particularly for technician versus resident 2 (T-R2) and resident 1 versus resident 2 (R1-R2) comparisons [9].
Comparative Performance Against Automated Systems
The fundamental limitations of manual analysis become particularly evident when compared to emerging automated technologies. A 2025 experimental study comparing assessment methods reported a correlation coefficient of only 0.57 between conventional semen analysis (CSA) and computer-aided semen analysis (CASA) for morphology evaluation [7]. In contrast, an artificial intelligence (AI) model demonstrated significantly stronger correlation with both CASA (r=0.88) and CSA (r=0.76), suggesting that AI more effectively captures the morphological features that human observers intend to assess but do so inconsistently [7].
Further evidence from deep learning research highlights the dramatic performance disparities between manual and automated approaches. Studies report inter-observer disagreement rates of up to 40% between expert evaluators, with kappa values as low as 0.05–0.15 indicating near-chance level agreement among trained technicians [8]. This diagnostic inconsistency has profound implications for clinical decision-making, particularly in selecting appropriate ART procedures such as IUI, IVF, or ICSI, where morphology thresholds guide treatment pathways [6].
Methodological Protocols: Manual Assessment Workflow
Understanding the sources of variability requires examination of the standard methodological protocols for manual sperm morphology assessment. The following section details the established procedures as outlined in the WHO guidelines.
Sample Preparation and Staining
Table 2: Essential Research Reagents for Sperm Morphology Assessment
| Reagent/Equipment | Function | Application Notes |
|---|---|---|
| Diff-Quik Stain | Rapid staining of sperm structures using triarylmethane dye, xanthene dye, and thiazine dye | Differentiates acrosomal (light blue) and post-acrosomal (dark blue) regions; mid-piece stains purple-red [6]. |
| Eosin-Nigrosin Stain | Vitality assessment through differential staining | Dead sperm heads appear pink; live sperm exclude stain [9]. |
| Proteolytic Enzymes (α-chymotrypsin, bromelain) | Reduce viscosity in abnormally thick samples | Incubate at 37°C for 10 minutes post-liquefaction [6]. |
| Improved Neubauer Hemocytometer | Sperm concentration calculation | Count all sperms in center 1mm×1mm area; apply dilution-specific multiplication factors [9]. |
| Ocular Micrometer | Precise measurement of sperm dimensions | Essential for accurate assessment of head size (5-6μm length, 2.5-3.5μm width) per WHO criteria [6]. |
The semen sample preparation process begins with collection in a sterile container after 2-7 days of abstinence, followed by liquefaction at 37°C for 30 minutes [6]. For viscous samples, proteolytic enzymes such as α-chymotrypsin or bromelain may be added with additional incubation for 10 minutes [6]. The liquefied sample is vortexed for 10 seconds, and a 10μL aliquot is extracted. If sperm concentration is below 2×10⁶/mL, centrifugation at 600g for 10 minutes is performed, leaving approximately 100μL of seminal plasma before gentle resuspension [6].
Smear preparation involves placing 10μL of well-mixed semen on a clean frosted slide with patient identifiers, then using a second slide at a 45° angle to create a smooth, even smear [6]. Slides are prepared in duplicate and air-dried before staining. The Diff-Quik staining protocol entails immersing the dried smear in fixative five times followed by complete drying for 15 minutes, then sequential immersion in solution I (three times for 10 seconds) and solution II (five times for 10 seconds) before rinsing in sterile water and vertical drying on absorbent paper [6]. Finally, a mounting medium such as Cytoseal is applied, and the slide is covered with a coverslip for examination.
Morphology Evaluation and Classification
Stained smears are examined under a bright-field microscope with 100× objective and 10× eyepiece, using immersion oil with a refractive index of 1.52 for optimal sharpness [6]. The evaluation requires scoring at least 200 spermatozoa across multiple fields, with all borderline forms classified as abnormal [6]. According to strict Tygerberg criteria, a spermatozoon must conform to all normal morphological characteristics: a smooth, regularly contoured oval head measuring 5-6μm in length and 2.5-3.5μm in width, with a well-defined acrosome covering 40-70% of the head area and containing no more than two small vacuoles occupying ≤20% of the head area [6]. The mid-piece must be slender, regular, approximately the same length as the head, and aligned with its axis, while the tail should be uniform and approximately 45μm long [6]. Any sperm with excess residual cytoplasm larger than one-third of the head area is classified as abnormal [6]. The reference threshold for morphologically normal forms is ≥4% according to the most recent WHO guidelines [6].
Diagram 1: Manual Analysis Workflow and Variability Sources
Technological Solutions: AI and Automated Approaches
The documented limitations of manual analysis have accelerated development of automated solutions, ranging from computer-assisted semen analysis (CASA) to advanced artificial intelligence systems.
Computer-Assisted Semen Analysis (CASA)
Traditional CASA systems were designed to objectively measure sperm concentration and motility but proved unreliable for morphology evaluation [8]. These systems typically operate by analyzing video recordings of semen samples, using algorithms for segmentation, localization, and tracking of sperm cells [10]. Open-source alternatives like OpenCASA have emerged, offering modules for motility, morphometry, membrane integrity, and guidance mechanism analysis while providing customizable platforms for method validation and development [11]. However, these systems still face challenges in capturing the subtle morphological features essential for accurate classification.
Artificial Intelligence and Deep Learning
Recent advances in artificial intelligence have demonstrated remarkable potential for overcoming the limitations of both manual assessment and traditional CASA systems. Deep learning frameworks combining Convolutional Block Attention Module (CBAM) with ResNet50 architecture and deep feature engineering have achieved test accuracies of 96.08±1.2% on benchmark datasets, representing significant improvements of 8.08% over baseline CNN performance [8]. These AI models minimize subjectivity through automated feature extraction and classification, with processing times reduced from 30-45 minutes per sample for manual analysis to under one minute [8].
A particularly promising development is the emergence of AI models capable of assessing unstained live sperm morphology using confocal laser scanning microscopy at low magnification [7]. This approach maintains sperm viability post-assessment, enabling immediate use in ART procedures—a significant advantage over traditional methods that require staining and fixation, rendering sperm unusable for further treatments [7].
Diagram 2: AI-Based Assessment Workflow and Advantages
The inherent limitations of manual sperm morphology analysis—subjectivity, inter-observer variability, lengthy processing times, and diagnostic inconsistency—represent significant challenges in male fertility assessment and reproductive research. Quantitative evidence demonstrates concerning levels of disagreement among even trained technicians, with intraclass correlation coefficients as low as 0.490 for morphology assessment [9]. These limitations have profound implications for clinical decision-making, particularly in selecting appropriate assisted reproductive technologies and predicting treatment outcomes.
The emerging paradigm of AI-based sperm morphology analysis offers a promising solution to these challenges, providing objective, standardized assessment with superior accuracy and significantly reduced processing times [7] [8]. For researchers and drug development professionals, understanding these technological transitions is essential for advancing reproductive medicine and developing next-generation diagnostic tools. Future directions should focus on validating AI systems across diverse clinical settings, establishing standardized protocols for automated analysis, and integrating these technologies into comprehensive male fertility assessment platforms.
Sperm morphology assessment, the evaluation of the size and shape of spermatozoa, has been a cornerstone of male fertility evaluation for decades. Its integration into clinical practice is based on the premise that the presence of a sufficient proportion of normally formed sperm is indicative of healthy spermatogenesis and is correlated with the ability to achieve fertilization and pregnancy [12]. Since the introduction of the first World Health Organization (WHO) laboratory manual in 1980, the criteria for defining 'normal' sperm morphology have continuously evolved, shifting from lenient to stricter thresholds, with the most recent 6th edition establishing a reference value of ≥4% normal forms [12] [13]. Despite its historical prominence, the clinical utility and prognostic value of sperm morphology in predicting both natural and assisted reproductive outcomes remain a subject of significant debate among clinicians and researchers [12]. This debate is fueled by the parameter's poor analytical reliability and conflicting evidence regarding its independent predictive power [12]. The contemporary landscape is further complicated by the emergence of artificial intelligence (AI) and machine learning (ML) technologies, which promise to revolutionize morphology assessment by introducing unprecedented levels of objectivity, speed, and accuracy [7]. This whitepaper provides an in-depth technical analysis of the prognostic value of traditional sperm morphology evaluation, frames it within the context of emerging AI-based methodologies, and details the experimental protocols shaping the future of fertility assessment.
Traditional Sperm Morphology Assessment
Evolution of Assessment Criteria and Standards
The methodology for sperm morphology assessment has undergone significant refinement. Initial evaluations used liberal criteria, with the first WHO manual (1980) setting the lower reference limit at 50% normal forms [12]. The subsequent introduction and adoption of the Kruger (Tygerberg) strict criteria represented a paradigm shift, characterizing sperm with even borderline abnormalities as "morphologically abnormal" [12] [13]. This evolution culminated in the detailed systematic approach of the WHO 6th Edition manual (2021), which defines a normal spermatozoon as having a smooth, oval head with a well-defined acrosome covering 40–70% of the head area, a midpiece that is slender and aligned with the head axis, and a tail of uniform caliber that is approximately ten times the length of the head without sharp bends [12] [13]. The current reference value of ≥4% normal forms is derived from the 5th percentile of a fertile population [13].
A critical challenge in traditional morphology assessment is high inter-laboratory variability. To ensure reliable and reproducible results, the WHO 6th Edition mandates rigorous standardization [12] [13]. This includes the use of trained personnel who participate in continuous internal and external quality control programs. The manual also emphasizes the importance of proper staining techniques (e.g., Papanicolaou, Diff-Quik) and detailed characterization of specific defects in the head, neck/midpiece, tail, and cytoplasmic residues, rather than simply reporting a single "abnormal" category [12].
Factors Influencing Sperm Morphology
Sperm morphology can be adversely affected by a range of environmental, occupational, and clinical factors, although the evidence for some associations remains heterogeneous.
Table 1: Factors Impacting Sperm Morphology and Evidence Quality
| Factor Category | Specific Factor | Reported Effect on Morphology | Evidence Quality & Notes |
|---|---|---|---|
| Lifestyle & Environmental | Cigarette Smoking | -1.37% to -1.88% difference in normal forms (conflicting data) [12] | Meta-analysis of 20 studies; conclusion confounded by semen analysis method. |
| Cannabis Use | No significant association with teratozoospermia found [12] | Meta-analysis of three large studies. | |
| Alcohol Consumption | Lower percentage of normal sperm, dose-dependent effect [12] | Meta-analysis of 11 studies. | |
| Air Pollution | Significant association with teratozoospermia [12] | -- | |
| Cell Phone Radiation | Potential negative effect, but results are conflicting [12] | Heat and radiation from devices kept in front pockets may be culprits. | |
| Anatomic & Health | Varicocele | Mean improvement of 6.1% in normal forms after repair [12] | Meta-analysis of prospective studies; results were inconsistent across studies. |
| Febrile Illness | Reductions in normal morphology post-illness [12] | Disruption of testicular thermoregulation. | |
| Bacterial Infections (e.g., Ureaplasma urealyticum) | Detrimental effect on morphology [12] | Semen microbiome is a nascent field of study. |
Prognostic Value in Fertility Outcomes
The clinical correlation between sperm morphology and fertility outcomes is complex and varies significantly depending on the mode of conception.
Natural Conception: Data on the prognostic value of sperm morphology for natural pregnancy is sparse. The Longitudinal Investigation of Fertility and the Environment (LIFE) study found that the percentage of abnormal morphology was associated with a small but statistically significant increase in the time to pregnancy. However, this association was not retained after controlling for other semen parameters, such as sperm concentration, suggesting that morphology is not an independent predictor of natural fecundity [12]. Notably, even men with 0% normal forms have demonstrated the ability to conceive naturally, indicating that morphology alone should not be used to preclude natural conception potential [12].
Intrauterine Insemination (IUI): The prognostic value of sperm morphology in IUI cycles is a subject of discussion. A key determinant appears to be the inseminated motile count (IMC). Evidence suggests that when the IMC is below one million, a normal sperm morphology of >4% can help achieve cumulative live birth rates comparable to cases with a higher IMC [13]. However, a meta-analysis found no difference in clinical pregnancy rates between patient subgroups with normal forms of >4%, ≤4%, and <1% when the total motile sperm count (TMSC) was above 10 million [13]. Female age is a critical interacting variable; for women older than 35 years, normal sperm morphology below 5% may predict poor IUI outcomes [13].
Assisted Reproductive Technology (ART):
- Conventional IVF (cIVF): Studies generally agree that fertilization rates can be negatively impacted by a low percentage of morphologically normal sperm [12] [13]. Some reports also indicate a lower rate of high-quality embryo formation [13]. However, the impact on ultimate pregnancy and live birth rates is less clear, with several large studies failing to find a significant association [12] [13].
- Intracytoplasmic Sperm Injection (ICSI): During ICSI, the embryologist actively selects a single sperm for oocyte injection, theoretically bypassing many natural selection barriers. Consequently, the prognostic value of overall semen morphology parameters for ICSI outcomes is considered limited [13]. The focus shifts from the population-level analysis to the selection of a single, morphologically optimal spermatozoon for injection.
The Rise of AI in Morphology Assessment
Limitations of Traditional Analysis and the Rationale for AI
The subjective nature of traditional visual assessment, combined with its high inter-operator variability, represents a major limitation to its reliability and prognostic power [12] [14]. This variability stems from the challenging and fatiguing task of classifying sperm based on complex, multi-parameter criteria. Artificial intelligence, particularly deep learning, offers a paradigm shift by providing a means for fully automated, objective, and highly reproducible sperm morphology analysis [7]. Furthermore, AI models can be developed to assess unstained, live sperm under lower magnifications, a capability that is impossible with traditional methods and is crucial for selecting viable sperm for clinical procedures like ICSI without compromising cellular integrity [7].
Development and Validation of an AI Model for Live Sperm
A landmark 2025 study by Thongkittidilok et al. developed and validated an in-house AI model for assessing the morphology of unstained, live sperm, providing a direct comparison with traditional methods [7].
Experimental Protocol:
- Sample Collection: Semen samples were collected from 30 healthy volunteers (aged 18-40) after 2-7 days of sexual abstinence.
- Imaging: A novel, high-resolution dataset was created. Unstained sperm were imaged using confocal laser scanning microscopy at 40x magnification in Z-stack mode (0.5 μm interval, 2 μm total range), generating high-quality images of live cells.
- Data Annotation and Categorization: Embryologists and researchers manually annotated over 12,000 sperm images. Each sperm was categorized into one of nine classes based on strict WHO 6th Edition criteria: one "normal" class and eight "abnormal" classes (e.g., abnormal head, vacuole, aberrant neck, abnormal tail). Normal morphology was confirmed only if the sperm met all criteria across five consecutive frames.
- AI Model Training: A deep learning model (ResNet50) was trained using transfer learning on a dataset of 9,000 images (4,500 normal, 4,500 abnormal). The model was trained to minimize the difference between its predictions and the expert annotations.
- Comparison and Validation: The performance of the AI model in quantifying the percentage of normal forms was compared against Computer-Aided Sperm Analysis (CASA) of stained sperm and Conventional Semen Analysis (CSA) by trained personnel.
Results: The AI model demonstrated superior performance, showing a stronger correlation with CSA (r = 0.76) than CASA showed with CSA (r = 0.57). Most notably, the correlation between the AI model and CASA was the highest (r = 0.88). The model achieved a test accuracy of 93%, with high precision and recall for both normal and abnormal sperm classes. Its processing speed was extremely fast, at approximately 0.0056 seconds per image, enabling rapid analysis [7].
Standardized Training Tools Augmented by Machine Learning
Addressing the root cause of variability in traditional analysis, another 2025 study developed a 'Sperm Morphology Assessment Standardisation Training Tool' based on machine learning principles to train novice morphologists [14]. The experiment demonstrated that untrained users initially achieved only 53% accuracy when using a detailed 25-category classification system. However, with the aid of visual aids and repeated training over four weeks, their accuracy significantly improved to 90%, and their diagnostic speed increased. This research highlights how AI-driven tools can be used not only for direct analysis but also to enhance human expertise, standardizing morphology assessment across laboratories and improving the reliability of traditional methods [14].
Comparative Analysis & The Scientist's Toolkit
Table 2: Comparative Analysis: Traditional vs. AI-Based Sperm Morphology Assessment
| Feature | Traditional Assessment | AI-Based Assessment |
|---|---|---|
| Basis of Assessment | Visual inspection by trained human personnel [12]. | Automated analysis by a trained deep learning model [7]. |
| Subjectivity | High, significant inter-operator variability [12] [14]. | Low, fully objective and reproducible [7]. |
| Sample Preparation | Requires staining (e.g., Papanicolaou, Diff-Quik) and fixation, rendering sperm non-viable [12] [7]. | Can be performed on unstained, live sperm, preserving viability [7]. |
| Magnification | High magnification (100x oil immersion) required [7]. | Can be performed at lower magnifications (e.g., 40x) with high-resolution imaging [7]. |
| Analysis Speed | Slow, labor-intensive process [14]. | Extremely fast (milliseconds per sperm) [7]. |
| Data Output | Percentage of normal and broadly abnormal forms; limited sub-categorization in practice. | Detailed classification into multiple normal and abnormal categories; quantitative and granular data [7]. |
| Clinical Integration | Standard of care, but prognostic value is debated [12]. | Emerging technology with potential to enhance ART outcomes via superior sperm selection [7]. |
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials and Reagents for Sperm Morphology Research
| Item | Function / Application | Technical Notes |
|---|---|---|
| Papanicolaou Stain | Recommended staining method for traditional morphology assessment. Provides best overall visibility of all sperm regions [13]. | Validated against WHO standards; requires proper technical validation if alternative stains (e.g., Diff-Quik, Shorr) are used [13]. |
| Diff-Quik Stain | A rapid Romanowsky-type stain variant for traditional morphology. Used for fixing and staining sperm smears for CASA or manual assessment [7]. | Allows for quicker processing than Papanicolaou but must be validated. |
| Confocal Laser Scanning Microscope | High-resolution imaging of unstained, live sperm for AI model development. Creates Z-stack images to capture 3D morphological details [7]. | Crucial for creating high-quality datasets for training AI models on live cells. |
| LabelImg Program | Open-source graphical image annotation tool. Used to manually draw bounding boxes and label sperm images for supervised machine learning [7]. | Creates the ground-truth dataset essential for training and validating AI models. |
| Pre-annotated Sperm Datasets (e.g., HSMA-DS, SCIAN-MorphoSpermGS) | Benchmark datasets for training and validating AI models. Contain hundreds to thousands of pre-classified sperm images [7]. | Limitations include low resolution or limited sample size, driving the need for novel, high-quality datasets. |
| Sperm Morphology Standardisation Training Tool | A tool based on machine learning principles to train novice morphologists, reducing subjectivity and improving accuracy in traditional assessment [14]. | Demonstrated significant improvement in classifier accuracy and diagnostic speed. |
Visualization of Workflows
Traditional Sperm Morphology Assessment Workflow
Diagram 1: Traditional assessment workflow.
AI-Based Sperm Morphology Assessment Workflow
Diagram 2: AI-based assessment workflow.
The prognostic value of sperm morphology in fertility outcomes is nuanced and context-dependent. While traditional assessment provides a foundational metric for male fertility evaluation, its utility as an independent predictor of success, particularly in assisted reproduction, is limited by subjectivity, variability, and a weak correlation with clinical pregnancy endpoints outside of its effect on fertilization rates in cIVF. The emergence of AI and machine learning is poised to address these fundamental limitations. AI models offer a paradigm shift towards objective, rapid, and highly detailed morphological analysis. Crucially, the ability to assess unstained, live sperm opens new avenues for selecting the most viable spermatozoa for ART procedures, potentially improving fertilization rates and embryo quality. For researchers and drug development professionals, the future lies in leveraging these advanced AI tools to discover novel, quantitative morphological biomarkers that are more tightly correlated with functional sperm competence and ultimate reproductive success. The integration of AI into both diagnostic practice and laboratory training promises to standardize and enhance the prognostic power of sperm morphology in the evolving landscape of reproductive medicine.
The assessment of sperm morphology represents a critical diagnostic procedure in male fertility evaluation. For decades, this analysis remained entrenched in manual methodologies characterized by significant subjectivity and inter-laboratory variability. The emergence of automated solutions marks a paradigm shift from these traditional approaches, driven by converging advancements in imaging technology, computational power, and artificial intelligence (AI). This whitepaper delineates the historical context of sperm morphology analysis and examines the core technological drivers catalyzing its automation, providing researchers and drug development professionals with a technical framework for understanding this transition within the broader thesis of traditional versus AI-based assessment.
Historical Context: From Manual Microscopy to Initial Automation
The history of semen analysis spans centuries, with the first observation of spermatozoa by Johan Ham and Antony van Leeuwenhoek in 1677 representing the foundational milestone [15]. For the next three centuries, analysis relied exclusively on manual microscopy without standardized protocols.
The Standardization Era and Manual Morphology Assessment
The pivotal development in modern semen analysis arrived with the publication of the World Health Organization (WHO) Laboratory Manual for the Examination and Processing of Human Semen in 1980 [16]. This manual, and its subsequent revisions in 1987, 1992, 1999, 2010, and 2021, established standardized procedures for the global community. The manual assessment of sperm morphology, as prescribed, involves a trained technician visually classifying over 200 spermatozoa into normal or abnormal categories based on strict criteria defining irregularities in the head, midpiece, and tail [17]. Despite standardization, this process suffers from inherent limitations:
- Subjectivity and Variability: The classification is highly dependent on the technician's expertise and experience, leading to substantial inter- and intra-observer variability [18] [4].
- Time-Intensive Workflow: The manual evaluation of hundreds of sperm cells per sample is laborious and limits laboratory throughput [19].
- Qualitative Limitations: Human assessment struggles to quantify subtle morphological features and patterns that may have clinical significance [7].
The First Wave of Automation: Computer-Aided Sperm Analysis (CASA)
Initial automation efforts focused on Computer-Aided Sperm Analysis (CASA) systems. These systems, evolving over approximately 40 years, integrated optical microscopes with digital cameras and basic image-processing software to provide automated assessments of sperm concentration and motility [19]. However, their capability for fully automated morphology analysis remained limited. Early CASA systems had a restricted ability to accurately distinguish spermatozoa from cellular debris and to classify midpiece and tail abnormalities, often producing unsatisfactory results due to limited image quality [18]. This initial wave of automation set the stage for more sophisticated AI-driven solutions by highlighting the need for advanced pattern recognition algorithms.
Technological Drivers Behind Modern Automated Solutions
The transition from manual and semi-automated systems to contemporary AI-powered platforms has been driven by several key technological advancements.
Core AI and Machine Learning Paradigms
The most significant driver is the maturation of artificial intelligence, particularly in machine learning (ML) and deep learning (DL).
- Classical Machine Learning: Early automated approaches utilized conventional ML algorithms such as Support Vector Machines (SVM), K-means clustering, and decision trees [4]. These models often relied on manually engineered features—shape-based descriptors, Hu moments, Zernike moments, and Fourier descriptors—to classify sperm heads into categories like normal, tapered, or pyriform [4]. While achieving accuracies up to 90% in some studies for head classification, their performance was limited by their dependence on these handcrafted features and their inability to holistically analyze the entire sperm structure (head, midpiece, and tail) in an integrated manner [4].
- Deep Learning and Convolutional Neural Networks (CNNs): Deep learning has superseded classical ML by automatically learning hierarchical feature representations directly from raw pixel data. Convolutional Neural Networks (CNNs) are now the cornerstone of modern sperm morphology analysis systems [18] [19] [4]. Studies have demonstrated the successful application of CNN architectures, including ResNet50, for the classification of unstained live sperm and for detailed morphological categorization based on the David classification [18] [7]. This shift from manual feature engineering to automated feature learning represents the primary technological leap enabling robust and accurate automation.
Table 1: Evolution of Algorithmic Approaches in Sperm Morphology Analysis
| Technological Era | Representative Algorithms | Feature Extraction Method | Primary Strengths | Primary Limitations |
|---|---|---|---|---|
| Classical Machine Learning | Support Vector Machine (SVM), K-means, Decision Trees | Manual engineering (e.g., shape, texture, moments) | Interpretability; efficiency with structured data [19] | Limited performance; inability to analyze complete sperm structure [4] |
| Deep Learning | Convolutional Neural Networks (CNNs), ResNet50 | Automated learning from raw image data | High accuracy; holistic analysis of entire sperm cell [18] [7] | "Black-box" nature; requires large, annotated datasets [19] |
Data Availability and Dataset Curation
The robustness of DL models is inherently dependent on large, high-quality, annotated datasets for training [19] [4]. The creation of dedicated, publicly available sperm image datasets has been a critical technological enabler. Notable examples include:
- SMD/MSS (Sperm Morphology Dataset/Medical School of Sfax): Comprises 1,000 images extended to 6,035 via augmentation, classified by experts according to the modified David classification (12 defect classes) [18].
- MHSMA (Modified Human Sperm Morphology Analysis Dataset): Contains 1,540 images of sperm heads, used for feature extraction related to acrosome, shape, and vacuoles [4].
- SVIA (Sperm Videos and Images Analysis): A larger dataset with ~125,000 annotated instances for detection and ~26,000 segmentation masks [4].
To overcome the challenge of limited data, researchers extensively use data augmentation techniques such as rotations, flips, and color variations to artificially expand dataset size and improve model generalizability [18].
Advanced Imaging and Processing Hardware
Improvements in imaging technologies provide the high-quality input data essential for AI analysis. Confocal laser scanning microscopy, for example, allows for the acquisition of high-resolution, z-stack images at low magnification, enabling the detailed analysis of unstained, live sperm—a crucial requirement for clinical use in assisted reproductive technologies [7]. Furthermore, the accessibility of powerful graphics processing units (GPUs) has made the training of complex, computationally intensive DL models feasible in clinical and research settings.
Detailed Experimental Protocols in AI-Based Morphology Assessment
The implementation of an AI-based sperm morphology analysis system follows a structured experimental pipeline. The following protocols are synthesized from recent key studies.
Protocol 1: CNN-Based Classification of Stained Sperm (SMD/MSS Dataset)
This protocol outlines the methodology for developing a multi-class classifier for stained sperm images [18].
1. Sample Preparation and Image Acquisition:
- Prepare semen smears from samples with a concentration of at least 5 million/mL, stained per WHO guidelines (e.g., RAL Diagnostics kit or Diff-Quik) [18] [7].
- Acquire images of individual spermatozoa using a microscope with a 100x oil immersion objective in bright-field mode and a digital camera [18].
2. Expert Annotation and Ground Truth Establishment:
- Have each image independently classified by multiple experienced embryologists according to a standardized classification system (e.g., modified David classification) [18].
- Compile a ground truth file containing the image name, classifications from all experts, and morphometric data. Resolve discrepancies through consensus or by establishing an agreement threshold (e.g., Total Agreement: 3/3 experts) [18].
3. Image Pre-processing and Augmentation:
- Clean images by handling missing values and outliers.
- Normalize pixel values and resize all images to a uniform dimension (e.g., 80x80 pixels) [18].
- Apply data augmentation techniques (e.g., rotation, scaling, flipping) to balance morphological classes and increase the effective size of the training set [18].
4. Model Training and Evaluation:
- Partition the augmented dataset into training (80%) and testing (20%) subsets [18].
- Implement a CNN architecture (e.g., custom Python model using TensorFlow/PyTorch) for multi-class classification.
- Train the model on the training set and evaluate its performance on the unseen test set using metrics such as accuracy, precision, and recall [18].
AI Classification Workflow for Stained Sperm
Protocol 2: AI Assessment of Unstained Live Sperm via Confocal Microscopy
This protocol describes a method for analyzing live, unstained sperm, preserving their viability for use in Assisted Reproductive Technology (ART) [7].
1. Sample Collection and Preparation:
- Collect semen samples from donors after 2-7 days of sexual abstinence.
- Dispense a 6 µL droplet onto a two-chamber slide with a 20 µm depth [7].
2. Confocal Image Acquisition:
- Capture images using a confocal laser scanning microscope (e.g., LSM 800) at 40x magnification in confocal mode (Z-stack) [7].
- Set the Z-stack interval to 0.5 µm, covering a total range of 2 µm to ensure all focal planes are captured [7].
3. Manual Annotation and Labeling:
- Manually annotate well-focused sperm images using a program (e.g., LabelImg), drawing bounding boxes around each sperm [7].
- Categorize sperm into classes (e.g., normal vs. abnormal) based on WHO criteria for unstained sperm, assessing head shape, vacuoles, neck, and tail across all Z-stack frames [7].
4. Deep Learning Model Development and Validation:
- Employ a transfer learning approach using a pre-trained architecture like ResNet50 [7].
- Fine-tune the model on the dataset of annotated, unstained sperm images.
- Validate the model's performance against manual annotations by embryologists, reporting metrics such as test accuracy, precision, and recall. Compare the AI's assessment of normal morphology rates against CASA and conventional semen analysis (CSA) [7].
Live Sperm Analysis via Confocal AI
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials and Reagents for Automated Sperm Morphology Research
| Item/Category | Function/Application | Specific Examples / Notes |
|---|---|---|
| Microscopy Systems | Image acquisition for model training and validation. | Bright-field microscope with 100x oil objective [18]; Confocal Laser Scanning Microscope (e.g., LSM 800) for live, unstained sperm [7]. |
| CASA Systems | Provides benchmark data and automated morphometry; often used for comparison. | IVOS II (Hamilton Thorne) with morphology software [7]. |
| Staining Kits | Provides contrast for traditional and some AI-based analysis of fixed sperm. | RAL Diagnostics kit [18]; Diff-Quik stain [7]. |
| Annotation Software | Manual labeling of sperm images to create ground truth datasets. | LabelImg program [7]. |
| AI/ML Frameworks | Development, training, and validation of deep learning models. | Python 3.8 with deep learning libraries (e.g., TensorFlow, PyTorch) [18]. |
| Public Datasets | Training and benchmarking models; facilitates reproducibility. | SMD/MSS [18]; MHSMA [4]; SVIA [4]. |
The automation of sperm morphology assessment is the product of a necessary evolution away from subjective manual methods, driven decisively by the maturation of deep learning, the strategic curation of annotated datasets, and advancements in imaging technology. While challenges remain, including model generalizability and the "black-box" nature of some complex algorithms, the trajectory is clear. The emerging paradigm offers the promise of objective, standardized, and high-throughput analysis. For researchers and drug development professionals, understanding these historical contexts and technological drivers is essential for leveraging these tools to advance reproductive medicine and develop novel therapeutic interventions.
AI Methodologies in Sperm Analysis: From Machine Learning to Deep Neural Networks
Within the broader research on traditional versus AI-based sperm morphology assessment, conventional machine learning (ML) represents a critical evolutionary step. Before the rise of deep learning, these methods formed the technological backbone for automating the analysis of sperm cells, relying heavily on human expertise to identify and quantify meaningful patterns [20] [4]. This technical guide details the core components of these conventional approaches: the manual craft of feature engineering and the application of classic classification algorithms, framed within the specific context of male fertility diagnostics.
Sperm morphology analysis is a cornerstone of male infertility assessment, with abnormal morphology strongly correlated with reduced fertility rates [8]. Traditional manual evaluation is notoriously subjective, time-consuming, and suffers from significant inter-observer variability, highlighting the need for objective, automated methods [4]. While deep learning has recently advanced the field, conventional ML approaches established the foundational principles for this automation, leveraging feature engineering and robust classifiers to standardize the process [21].
Feature Engineering in Sperm Morphology Analysis
Feature engineering is the process of transforming raw data into features that better represent the underlying problem to predictive models. In the context of sperm morphology, this involves converting raw pixel values from sperm images into quantitative descriptors that capture essential morphological characteristics [4].
Core Feature Types and Techniques
The following table summarizes the primary categories of features engineered for conventional ML-based sperm morphology analysis.
Table 1: Feature Engineering Techniques for Sperm Morphology
| Feature Category | Description | Specific Examples | Application in Sperm Analysis |
|---|---|---|---|
| Shape-Based Descriptors | Quantify the geometric properties of the sperm head, midpiece, and tail. | Area, perimeter, eccentricity, length, width, elongation [4] [21]. | Head length-to-width ratio is critical for identifying normal oval heads (1.5–2) [8]. |
| Texture & Intensity Features | Capture surface characteristics and staining patterns. | Grayscale intensity, histogram statistics, edge density [20]. | Differentiating acrosome regions, detecting vacuoles, or identifying staining irregularities. |
| Mathematical Moment Invariants | Advanced shape descriptors that are invariant to rotation, scale, and translation. | Hu moments, Zernike moments, Fourier descriptors [4] [21]. | Providing a robust, compact representation of complex head shapes (e.g., tapered vs. pyriform) [21]. |
The process of feature engineering extends beyond simple extraction. As with general machine learning principles, feature selection is a critical subsequent step to identify the most informative features, reduce dimensionality, and prevent overfitting [22]. Techniques such as Principal Component Analysis (PCA) transform the original features into a set of linearly uncorrelated components, while methods like Recursive Feature Elimination (RFE) or Mutual Information scoring can select the most predictive subset of features [8] [22].
Conventional Classification Algorithms
Once discriminative features are engineered, they serve as input to classification algorithms that assign sperm into predefined morphological categories, such as normal, tapered, pyriform, small, or amorphous [21].
Prominent Algorithms and Performance
The table below outlines key algorithms and their documented performance in peer-reviewed studies on sperm morphology classification.
Table 2: Conventional Classification Algorithms in Sperm Morphology Analysis
| Algorithm | Key Characteristics | Reported Performance |
|---|---|---|
| Support Vector Machine (SVM) | Finds the optimal hyperplane to separate different classes in a high-dimensional feature space. Effective for binary and multi-class problems [4]. | - A Bayesian Density Estimation model with SVM achieved 90% accuracy classifying sperm heads [4].- Another study yielded 88.59% AUC-ROC and precision above 90% for good/bad head classification [4]. |
| Cascade Ensemble of SVMs (CE-SVM) | A multi-stage approach using specialized SVMs for different classification subtasks to improve overall accuracy [21]. | Achieved an average true positive rate of 58% on a dataset requiring expert agreement [21]. |
| k-Nearest Neighbors (k-NN) | A simple, instance-based learning algorithm that classifies a sample based on the majority class among its k-nearest neighbors in the feature space. | Used in conjunction with Principal Component Analysis for human sperm health diagnosis [21]. |
| Decision Trees | A hierarchical model of decisions and their possible consequences, creating a tree-like structure that is relatively easy to interpret. | Listed among the archetypal algorithms (along with k-means and SVM) applied in the field, though often limited by handcrafted features [4]. |
Experimental Protocols and Workflows
A standardized experimental pipeline is crucial for the reproducible application of conventional ML to sperm morphology analysis. The following workflow details the key stages from sample preparation to model evaluation.
Detailed Experimental Methodology
1. Sample Preparation and Staining
- Smears are prepared from semen samples according to World Health Organization (WHO) guidelines [18].
- Staining is typically performed using Romanowsky-type stains (e.g., Diff-Quik) or specific kits (e.g., RAL Diagnostics) to enhance contrast and cellular detail [7] [18].
2. Data Acquisition and Pre-processing
- Images are captured using a microscope equipped with a digital camera, often with a 100x oil immersion objective for high magnification [18].
- The CASA (Computer-Assisted Semen Analysis) system's morphometric tool can be used to determine initial measurements of head width/length and tail length [18].
- Pre-processing steps are critical. As noted in research, this can include:
- Denoising: Techniques like wavelet denoising are applied to remove noise signals from poorly lit or stained images [8].
- Normalization/Standardization: Numerical features are brought to a common scale to prevent dominance by features with large magnitudes. Images may be resized to a standard resolution [18].
3. Expert Annotation and Ground Truth Establishment
- Each spermatozoon is manually classified by multiple experienced experts following a standardized classification system (e.g., WHO criteria, modified David classification) [18].
- A ground truth file is compiled for each image, containing the image name, classifications from all experts, and morphometric dimensions. This file is essential for supervised learning [18].
4. Feature Engineering Pipeline
- Feature Extraction: Shape-based descriptors (area, perimeter), texture features, and moment invariants (Hu, Zernike) are extracted from each segmented sperm cell [4] [21].
- Feature Selection: Techniques like PCA or mutual information-based selection are employed to reduce noise and dimensionality, retaining the most informative features for model training [8] [22].
5. Model Training and Validation
- The dataset is partitioned, typically with 80% used for training and 20% held out for testing [18].
- Classifiers like SVM are trained on the feature vectors from the training set.
- Performance is validated using k-fold cross-validation (e.g., 5-fold) to ensure robustness and avoid overfitting [8].
Visual Workflow Diagram
The following diagram illustrates the logical flow of the conventional machine learning pipeline for sperm morphology analysis.
The Scientist's Toolkit: Research Reagent Solutions
The experimental protocols rely on a suite of specific reagents and tools. The following table details essential items and their functions in the context of conventional ML-based sperm morphology analysis.
Table 3: Essential Research Reagents and Materials
| Item | Function/Application |
|---|---|
| Diff-Quik Stain | A Romanowsky-type stain variant used to stain fixed sperm smears, enhancing the contrast and visibility of cellular structures (head, acrosome, midpiece, tail) for subsequent imaging and feature extraction [7]. |
| RAL Diagnostics Stain | A commercial staining kit used for preparing semen smears, providing consistent coloration for morphological assessment [18]. |
| CASA System (e.g., IVOS II) | A Computer-Assisted Semen Analysis system used for initial image acquisition, cell tracking, and providing preliminary morphometric measurements (head dimensions, tail length) that can inform feature engineering [7] [18]. |
| SVM Classifiers (with RBF/Linear Kernels) | The core algorithmic tool for the final classification step. SVMs use the engineered features to build a model that distinguishes between different morphological classes of sperm [8] [4]. |
| Feature Selection Algorithms (e.g., PCA, Chi-square) | Statistical and algorithmic tools used post-feature-extraction to identify and retain the most discriminative features, improving model performance and efficiency [8]. |
Conventional machine learning approaches, built upon meticulously engineered features and robust classifiers like SVMs, laid the essential groundwork for the automation of sperm morphology analysis. These methods demonstrated significant success in reducing subjectivity and establishing quantitative benchmarks [4] [21]. However, their fundamental limitation lies in the dependency on manual feature extraction, a process that is not only cumbersome and time-consuming but also inherently limited by human design, which can restrict the model's ability to learn more complex and subtle morphological patterns [20] [8]. This key shortcoming paved the way for the next paradigm shift in the field: the adoption of deep learning models capable of automated, end-to-end feature learning and classification.
The assessment of cellular morphology represents a critical challenge across numerous biomedical disciplines, perhaps nowhere more consequentially than in the field of male fertility, where sperm morphology analysis is a cornerstone diagnostic. Traditional manual assessment methods are plagued by inherent subjectivity, significant inter-observer variability, and labor-intensive processes [4] [5]. Within this context, artificial intelligence has emerged as a transformative technology, with Convolutional Neural Networks (CNNs) standing as the fundamental architecture powering this revolution. In 2025, CNNs are projected to be the engine behind a computer vision market worth over $25 billion, capable of identifying objects in images with over 99% accuracy—a rate that often surpasses human performance [23]. This technical guide provides an in-depth examination of core deep learning architectures—CNNs, ResNet50, and the Convolutional Block Attention Module (CBAM)—framed within their groundbreaking application to automated sperm morphology assessment. By elucidating both the theoretical foundations and practical implementations of these technologies, this review equips researchers and clinicians with the knowledge necessary to leverage AI for overcoming long-standing limitations in morphological analysis.
Core Architectural Principles
Convolutional Neural Networks: Biological Inspiration
CNNs are specifically designed to process pixel data, mimicking the hierarchical pattern recognition of the human visual cortex [23]. When you look at an object, your brain first identifies simple shapes like edges and corners, then combines these into more complex patterns like textures and objects. CNNs operate on this same principle: their early layers learn basic features like colors and edges, deeper layers combine these into more complex patterns like textures, and the final layers recognize whole objects [23]. This biological inspiration makes CNNs uniquely suited for image analysis tasks, including the complex morphological assessment required in sperm analysis.
Table 1: Fundamental Layers in a Convolutional Neural Network
| Layer Type | Primary Function | Technical Operation | Biological Analogy |
|---|---|---|---|
| Convolutional | Feature detection | Applies filters/kernels across input image to create feature maps | Simple cell receptive fields in V1 |
| Activation (ReLU) | Introduces non-linearity | Applies element-wise activation function (e.g., max(0,x)) | Neural firing threshold |
| Pooling | Dimensionality reduction | Downsamples feature maps (max, average) | Complex cell spatial invariance |
| Fully Connected | Classification | Connects all neurons between layers for final prediction | Higher cognitive integration |
CNN Data Processing Pipeline
The transformation of raw pixel data into actionable classifications follows a sophisticated, multi-stage pipeline that acts as a digital assembly line for visual understanding [23]. Modern optimized networks can classify an image in just milliseconds—faster than the blink of an eye—through this highly efficient process:
- Input Processing: The image is converted into a grid of numerical values representing pixel color and brightness [23].
- Feature Extraction: The data passes through repeating cycles of convolution, activation, and pooling layers. With each cycle, the network detects increasingly complex features—from simple edges to textures to object parts [23].
- Flattening: The extracted 2D feature maps are transformed into a single, long vector of numbers, lining up all evidence for final assessment [23].
- Classification: This feature vector is fed into fully connected layers that weigh all evidence and calculate probability scores for different outcomes [23].
Training and Optimization Fundamentals
Training a CNN is a complex optimization process where the network learns to minimize its prediction errors. The network makes initial guesses about images, compares these to known correct answers, and calculates an error score using a loss function [23]. Through backpropagation, the network then works backward through its layers to identify which internal connections contributed most to the error, adjusting its parameters accordingly [23]. A critical challenge in this process is overfitting, where the network memorizes training examples rather than learning generalizable features. This is addressed through regularization techniques like dropout (randomly turning off parts of the network during training) and data augmentation (creating more training data by rotating, flipping, or cropping existing images) [23]. These techniques force the network to learn robust features that generalize to new data—a crucial capability for clinical applications where sample variability is high.
Advanced Architectures for Complex Morphological Assessment
ResNet50: Overcoming Deep Network Limitations
As networks grow deeper to capture more complex features, they encounter the vanishing gradient problem, where weight updates become infinitesimally small during backpropagation, effectively halting learning in early layers. The ResNet (Residual Network) architecture, specifically ResNet50 with its 50 layers, introduces a groundbreaking solution: skip connections [24] [25]. These connections create "highways" that allow gradients to flow directly through layers by implementing identity mapping. Rather than hoping each layer perfectly learns a desired underlying mapping, ResNet layers instead learn residual functions—the difference between input and output. If a layer has nothing useful to add, the residual approaches zero, and the skip connection dominates. This elegant architecture enables training of previously unmanageable deep networks while improving both performance and training efficiency [25].
Attention Mechanisms: The CBAM Innovation
While deeper networks capture more features, not all features contribute equally to the final decision. Attention mechanisms address this by dynamically highlighting important features while suppressing less relevant ones, mimicking human cognitive focus [24] [26]. The Convolutional Block Attention Module (CBAM) is a lightweight, effective attention mechanism that sequentially applies both channel attention (identifying "what" is important) and spatial attention (identifying "where" important features are located) [24] [26]. In medical imaging applications like sperm morphology assessment, CBAM helps networks focus on structurally significant regions—such as sperm heads, midpieces, and tails—while ignoring background noise or artifacts [24]. This capability is particularly valuable in complex biological images where multiple structures compete for diagnostic relevance.
Integrated Architectures: GM-CBAM-ResNet
Recent research has explored integrating multiple architectural innovations to create highly efficient models. The GM-CBAM-ResNet architecture incorporates both the Ghost Module (GM) for parameter reduction and CBAM for attention-driven feature refinement within a ResNet framework [24]. The Ghost Module reduces computational redundancy by generating some feature maps through cheap linear operations on existing ones rather than through expensive convolution [24]. When combined with CBAM's attention mechanism, this creates a lightweight yet highly accurate architecture ideal for clinical deployment where computational resources may be limited. On benchmark datasets, GM-CBAM-ResNet has demonstrated a 45.4% reduction in parameters while improving diagnostic accuracy by approximately 5% compared to standard ResNet [24].
Application to Sperm Morphology Assessment
Limitations of Conventional Assessment
Traditional sperm morphology assessment faces significant challenges that impact diagnostic reliability and clinical utility. The process remains highly subjective, with studies showing that expert morphologists agree on normal/abnormal classification for only 73% of sperm images [5]. This inter-observer variability stems from the complex nature of morphological classification, which requires simultaneous evaluation of head, neck, and tail abnormalities across numerous defect categories [4] [5]. Manual assessment is also time-consuming, with trained morphologists taking approximately 4.9–7.0 seconds per image classification even after extensive training [5]. These limitations have created an urgent need for automated, objective assessment methods that can deliver consistent, reproducible results across clinical laboratories.
Deep Learning Implementation Frameworks
Multiple research teams have developed sophisticated deep learning frameworks specifically for sperm morphology assessment. The following experimental protocols represent current state-of-the-art approaches:
Protocol 1: ResNet50 Transfer Learning for Stained Sperm Morphology
- Dataset: 6035 images of individual spermatozoa extended from 1000 original images through data augmentation [27]
- Sample Preparation: Sperm images acquired using MMC CASA system, stained with Diff-Quik following standard protocols [27]
- Annotation: Expert classification by three morphologists based on modified David classification [27]
- Model Architecture: ResNet50 with transfer learning, customized final classification layer [27]
- Training: Fine-tuning on sperm dataset, class imbalance addressing through weighted loss function [27]
- Performance: Accuracy ranging from 55% to 92% across different morphological categories [27]
Protocol 2: Custom CNN for Unstained Live Sperm Assessment
- Dataset: 21,600 images with 12,683 annotated unstained sperm [7]
- Sample Preparation: Sperm imaged live using confocal laser scanning microscopy at 40× magnification in LSM Z-stack mode [7]
- Annotation: Manual bounding box annotation by embryologists using LabelImg program [7]
- Model Architecture: Custom deep learning model for simultaneous detection and classification [7]
- Training: 150 epochs, batch size 32, Adam optimizer with learning rate 0.001 [7]
- Performance: 93% test accuracy, precision 0.95/recall 0.91 for abnormal sperm, precision 0.91/recall 0.95 for normal sperm [7]
Table 2: Performance Comparison of AI Models for Sperm Morphology Assessment
| Model Architecture | Dataset Characteristics | Accuracy | Precision | Recall | Processing Speed |
|---|---|---|---|---|---|
| ResNet50 Transfer Learning [27] | 6035 stained sperm images | 55–92% (category dependent) | N/R | N/R | N/R |
| Custom CNN (Unstained) [7] | 21,600 unstained sperm images | 93% | 0.91–0.95 | 0.91–0.95 | 0.0056 s/image |
| GM-CBAM-ResNet [24] | ECG images (architectural benchmark) | ~5% improvement over baseline | N/R | N/R | 45.4% parameter reduction |
Research Reagent Solutions
Table 3: Essential Research Materials for AI-Based Sperm Morphology Analysis
| Reagent/Equipment | Specification | Application Function |
|---|---|---|
| Confocal Laser Scanning Microscope [7] | LSM 800, 40× magnification, Z-stack interval 0.5μm | High-resolution imaging of unstained live sperm |
| Computer-Aided Semen Analysis (CASA) | IVOS II, Hamilton Thorne [7] | Standardized sperm concentration and motility assessment |
| Diff-Quik Stain | Romanowsky stain variant [7] | Sperm staining for conventional morphology assessment |
| LabelImg Program | Python-based annotation tool [7] | Manual bounding box annotation for dataset creation |
| Phase Contrast Optics | Standard compound microscope [5] | Live sperm visualization without staining |
| LEJA Slides | 20μm preparation depth, 026855, SC-20-01-C [7] | Standardized chamber slides for semen analysis |
Comparative Analysis and Validation
Performance Benchmarking
Validation studies demonstrate that AI-based sperm morphology assessment correlates strongly with established methods. One study comparing an in-house AI model with Computer-Aided Semen Analysis (CASA) and Conventional Semen Analysis (CSA) found the AI model showed the strongest correlation with CASA (r = 0.88), followed by CSA (r = 0.76) [7]. The correlation between CASA and CSA was weaker (r = 0.57), suggesting AI models may potentially exceed conventional methods in consistency [7]. The same study found the AI model achieved a test accuracy of 93% after 150 epochs of training, with precision of 0.95 and recall of 0.91 for detecting abnormal sperm morphology [7]. These results indicate that well-designed deep learning systems can meet or exceed expert-level performance while providing greater standardization.
Diagnostic Efficiency Metrics
The implementation of deep learning systems dramatically improves diagnostic workflow efficiency. While trained human morphologists require 4.9–7.0 seconds to classify a single sperm image [5], optimized AI models can process images in approximately 0.0056 seconds per image—nearly 1000 times faster [7]. This acceleration enables comprehensive analysis of larger sperm populations, potentially improving the statistical reliability of morphology assessments. Furthermore, AI systems maintain this performance consistently without fatigue or drift in assessment criteria, addressing a significant limitation of human-based morphological analysis [5].
Future Directions and Clinical Implementation
Emerging Architectural Innovations
The field of deep learning continues to evolve rapidly, with several emerging architectures showing promise for medical imaging applications. The Dense Skip-Attention method represents a significant advancement that establishes connections between all attention modules within a network, forcing the model to learn interactive attention features across the entire architecture [26]. This approach enhances performance without significantly increasing computational complexity, maintaining minimal impact on both parameters and operations [26]. Similarly, the ECA (Efficient Channel Attention) mechanism optimizes the traditional squeeze-and-excitation approach by avoiding channel dimensionality reduction, thereby better preserving information while maintaining efficiency [25]. These innovations point toward increasingly sophisticated yet computationally efficient architectures ideally suited for clinical deployment.
Clinical Translation Challenges
Despite impressive technical capabilities, several challenges remain for widespread clinical adoption of AI-based sperm morphology assessment. The "black box" problem—the difficulty in interpreting how deep learning models arrive at specific decisions—represents a particular concern in clinical medicine where diagnostic reasoning must often be explained [23]. Additionally, dataset limitations including low resolution, limited sample sizes, and insufficient morphological categories continue to constrain model generalizability [4]. Future research must focus on developing more comprehensive, multi-center datasets and creating explainable AI techniques that provide transparent diagnostic rationale. As these technical and validation challenges are addressed, deep learning architectures—particularly optimized networks like CBAM-enhanced ResNet50—are poised to become indispensable tools for standardized, objective sperm morphology assessment, potentially transforming the diagnostic landscape in reproductive medicine.
The integration of advanced deep learning architectures—particularly CNN frameworks, ResNet50, and attention mechanisms like CBAM—represents a paradigm shift in sperm morphology assessment. These technologies offer a solution to the long-standing challenges of subjectivity, variability, and inefficiency that have plagued conventional morphological analysis. By providing standardized, automated classification with accuracy approaching or exceeding human experts, these systems have the potential to significantly improve diagnostic consistency in male fertility assessment. The architectural principles and implementation frameworks detailed in this technical review provide researchers and clinicians with both the theoretical foundation and practical roadmap for leveraging these transformative technologies in both research and clinical settings.
The assessment of sperm morphology has long been a cornerstone of male fertility evaluation. Traditional methods, as outlined by the World Health Organization (WHO), require sperm to be fixed and stained before analysis under high magnification (100×), a process that renders them non-viable for subsequent clinical use [7]. This approach, while established, is plagued by significant subjectivity and variability, with results often differing based on the technician's skill and interpretation [28]. This manual process is not only labor-intensive and time-consuming but also leads to substantial variations between individuals and across laboratories, undermining the standardization of sperm quality criteria and the accuracy of male fertility evaluations [29].
Artificial intelligence (AI) is poised to revolutionize this field by introducing objective, automated, and highly accurate analysis methods. A key advancement is the ability to analyze live, unstained sperm, facilitating the immediate selection of viable sperm for assisted reproductive technology (ART) procedures such as intracytoplasmic sperm injection (ICSI) [7]. This technical guide delves into two groundbreaking AI applications: the morphological analysis of unstained live sperm and the prediction of sperm fertilization competence from the egg's perspective, framing them within the broader thesis of overcoming the limitations inherent in traditional assessment methods.
AI for Live, Unstained Sperm Morphology Analysis
Core Methodology and Technological Workflow
The fundamental innovation in this domain is the use of confocal laser scanning microscopy to capture high-resolution images of live, unstained sperm at a lower magnification (40×) [7]. This technology generates Z-stack images at intervals of 0.5 μm, covering a total range of 2 μm, which allows for detailed subcellular examination without compromising sperm viability [7].
The subsequent analytical workflow is powered by deep learning. Researchers have developed frameworks that integrate multiple algorithms for comprehensive analysis. A prominent approach involves:
- Sperm Tracking: An improved FairMOT tracking algorithm incorporates the distance, angle of sperm head movement, and the Intersection over Union (IOU) value of the head target detection frame into the cost function of the Hungarian matching algorithm. This significantly enhances the accuracy of tracking individual sperm in motion [30].
- Morphological Segmentation: The BlendMask method is used to segment individual sperm, followed by the use of SegNet to separate the head, midpiece, and principal piece of each sperm [30]. This allows for a detailed, component-wise morphological assessment.
Other methodologies utilize transfer learning with established deep neural networks like ResNet50, which are trained on novel datasets of annotated sperm images to classify sperm as normal or abnormal based on WHO criteria [7]. The model processes images through multiple convolutional layers to extract hierarchical features, which are then used for classification.
The diagram below illustrates the integrated workflow of this AI-powered analysis system.
Quantitative Performance and Validation
The performance of these AI models in classifying unstained sperm morphology has been rigorously validated against traditional methods. The following table summarizes key performance metrics from recent studies.
Table 1: Performance Metrics of AI Models for Unstained Sperm Morphology Analysis
| Model / Study Feature | Reported Performance Metric | Comparative Outcome |
|---|---|---|
| In-house AI Model (ResNet50) [7] | Test Accuracy: 0.93Precision (Abnormal): 0.95Recall (Normal): 0.95 | Strongest correlation with CASA (r = 0.88), followed by Conventional Semen Analysis (r = 0.76) |
| Multidimensional Framework [30] | Morphological Accuracy: 90.82%High Consistency with Manual Microscopy | Validated on 1,272 samples across multiple tertiary hospitals |
| Processing Speed [7] | ~0.0056 seconds per image~139.7 seconds for 25,000 images | Enables high-throughput, real-time clinical analysis |
A prospective study further demonstrated the clinical utility of an AI-enabled computer-assisted semen analyzer (CASA), which showed statistically significant improvements (p < 0.05) in postoperative sperm parameters for patients undergoing varicocelectomy, underscoring its concordance with manual analysis and value in clinical decision-making [31].
AI for Fertilization Competence Prediction
Core Methodology and Technological Workflow
Moving beyond basic morphology, a pioneering AI model developed by HKUMed addresses a more complex question: which sperm possess the actual capacity to fertilize an egg? This model evaluates sperm quality from the egg's perspective by focusing on the crucial first step of fertilization—the binding of sperm to the zona pellucida (ZP), the outer coat of the egg [29].
The ZP selectively binds to sperm with normal morphology, intact chromosomes, and fertilization capability, acting as a natural screening mechanism [29]. The AI model was trained using advanced deep-learning techniques on a dataset of over 1,000 sperm images to recognize the subtle morphological features associated with this binding capability [29]. From 2022 to 2024, the model was rigorously validated on over 40,000 sperm images from 117 men diagnosed with infertility or unexplained infertility [29]. The results confirmed a strong correlation between the proportion of sperm capable of binding to the ZP and the success rate of ART procedures. A critical clinical threshold was established at 4.9%; men with a lower percentage of ZP-binding sperm are considered at higher risk for fertilization failure during IVF [29].
The logical process of how this AI model bridges traditional analysis and functional competence prediction is outlined below.
Quantitative Performance and Validation
This novel approach has demonstrated exceptional accuracy in clinical validation, offering a direct solution to the limitations of conventional semen analysis.
Table 2: Performance and Application of the AI Model for Fertilization Competence
| Feature | Detail |
|---|---|
| Validation Accuracy [29] | Exceeded 96% |
| Clinical Parameter | Percentage of sperm capable of binding to the Zona Pellucida (ZP) |
| Diagnostic Threshold [29] | < 4.9% (indicates high risk of fertilization failure in IVF) |
| Clinical Value | Serves as a novel diagnostic tool for issues conventional analysis may overlook; allows for tailored treatment plans [29] |
The Scientist's Toolkit: Essential Research Reagents and Materials
The development and implementation of these advanced AI models rely on a foundation of specific laboratory instruments, reagents, and computational resources. The following table details key components of the research toolkit for this field.
Table 3: Key Research Reagent Solutions for AI-Based Sperm Analysis
| Item Name | Function / Application | Specific Examples / Notes |
|---|---|---|
| Confocal Laser Scanning Microscope | High-resolution, Z-stack imaging of live, unstained sperm [7] | LSM 800; used at 40x magnification in confocal mode [7] |
| Standardized Slide Systems | Preparing semen samples of consistent depth for imaging [7] | LEJA slides (20 μm preparation depth) [7] |
| AI-CASA Systems | Automated, AI-powered semen analysis for concentration, motility, and morphology [31] | LensHooke X1 PRO; IVOS II (Hamilton Thorne) [31] |
| Image Annotation Software | Manual labeling of sperm images for training supervised AI models [7] | LabelImg program [7] |
| Deep Learning Frameworks | Developing and training custom neural network models for classification and segmentation | ResNet50, BlendMask, SegNet, FairMOT [7] [30] |
| High-Performance Computing | Processing large image datasets (thousands to millions of images) within clinically viable timeframes [7] | Required for model training and high-throughput analysis |
Discussion and Future Directions
The integration of AI into sperm analysis marks a paradigm shift from subjective, destructive assessment to objective, non-invasive, and functionally relevant evaluation. The abilities to analyze live sperm without staining and to predict fertilization competence address long-standing limitations in male infertility diagnosis and treatment [7] [29]. These technologies not only improve diagnostic accuracy but also directly enhance ART outcomes by enabling the selection of the highest quality sperm for procedures like ICSI.
Future efforts will focus on multicenter validation trials to ensure robustness across diverse patient populations and clinical environments [32]. Furthermore, the integration of AI into automated sperm selection systems for IVF/ICSI is a key developmental trajectory [32]. As noted in recent reviews, for AI to achieve widespread clinical adoption, it must "inspire trust, integrate seamlessly into workflows and deliver real benefits," ensuring that embryologists and clinicians remain central to an augmented, more efficient ART process [33].
The evaluation of sperm quality is a cornerstone in the diagnosis and treatment of male infertility, which contributes to approximately 50% of infertility cases worldwide [34]. For decades, conventional semen analysis has relied on manual assessment by embryologists and technicians, a approach plagued by subjectivity, high inter-observer variability, and inherent inefficiencies [34] [4]. Computer-Aided Sperm Analysis (CASA) systems initially promised automation and standardization; however, these systems often struggled with accurately distinguishing sperm from similar-sized debris and offered limited analysis of complex morphological features [34] [19]. The integration of Artificial Intelligence (AI), particularly machine learning (ML) and deep learning (DL), is now revolutionizing CASA systems by enabling more accurate, objective, and high-throughput evaluations of key sperm parameters, including motility, morphology, and DNA integrity [19].
This transformation is critical within assisted reproductive technologies (ART), where the selection of a single sperm from millions for procedures like intracytoplasmic sperm injection (ICSI) carries profound implications for success rates [35]. Traditional sperm morphology assessment requires staining and examination under high magnification (100×), a process that renders sperm unusable for subsequent procedures [7]. AI-driven CASA systems overcome this limitation by capable of analyzing unstained, live sperm with high reliability, thereby improving the selection of high-quality sperm for fertility treatments [7]. This technical guide explores the core architectures, experimental validations, and practical implementations of these advanced AI-CASA systems, framing them within the broader research context of moving from traditional subjective methods to objective, algorithm-enhanced precision medicine in reproductive biology [19].
AI Technologies Powering Next-Generation CASA
The evolution from traditional CASA to AI-enhanced systems is marked by a shift from simple image processing to sophisticated pattern recognition and predictive modeling. At the heart of this revolution are deep learning algorithms, especially convolutional neural networks (CNNs), which excel at processing complex image and video data to extract nuanced features beyond human discernment [19].
Deep Learning for Sperm Morphology Analysis
Conventional machine learning approaches for sperm morphology analysis relied on manually engineered features—such as shape descriptors, grayscale intensity, and texture patterns—which often proved inadequate for the vast heterogeneity of sperm forms [4]. Deep learning models, particularly the ResNet50 architecture used in recent studies, automatically learn hierarchical feature representations directly from pixel data, enabling comprehensive assessment of sperm head, neck, and tail structures without human bias [7]. One in-house AI model demonstrated exceptional performance in classifying unstained live sperm, achieving a test accuracy of 93%, with precision and recall rates for abnormal sperm morphology reaching 0.95 and 0.91, respectively [7].
These models require extensive training on high-quality annotated datasets. Recent research has utilized confocal laser scanning microscopy at 40× magnification to create high-resolution Z-stack images, covering a range of 2 μm with a 0.5 μm interval [7]. This approach generates detailed image sets of 512 × 512 pixels, with each capture containing 2-3 sperm, enabling the model to reconstruct three-dimensional morphological features from two-dimensional images [7]. The model's processing capability of approximately 0.0056 seconds per image facilitates real-time analysis, making it suitable for clinical applications where timely decision-making is critical [7].
Algorithmic Advances in Motility and Kinematic Analysis
For motility assessment, AI algorithms have moved beyond simple trajectory tracking to sophisticated movement pattern classification. Modern systems employ frame rates of 60 fps to track sperm trajectories over ≥30 consecutive frames, applying stringent criteria to discard non-sperm objects [31]. The algorithms classify motility based on complex parameters: progressive motility (PR) is defined as a velocity average path (VAP) ≥25 µm/s and straightness (STR) ≥0.80; non-progressive (NP) includes motile sperm below these thresholds; and immotile (IM) sperm show no displacement >2 µm/s [31].
The table below summarizes key kinematic parameters analyzed by AI-CASA systems:
Table 1: Key Sperm Kinematic Parameters Quantified by AI-CASA Systems
| Parameter | Abbreviation | Description | Clinical Significance |
|---|---|---|---|
| Curvilinear Velocity | VCL | Total path distance per unit time | Reflects overall energy and vitality |
| Straight-Line Velocity | VSL | Net straight-line distance per unit time | Indicates progressive movement efficiency |
| Average Path Velocity | VAP | Average smoothed path velocity | Used for motility classification |
| Amplitude of Lateral Head Displacement | ALH | Mean width of head oscillation | Correlates with hyperactivation potential |
| Beat Cross Frequency | BCF | Rate of head crossing the average path | Measures flagellar beating efficiency |
| Linearity | LIN | (VSL/VCL) × 100 | Indicates trajectory straightness |
| Straightness | STR | (VSL/VAP) × 100 | Measures path consistency |
| Wobble | WOB | (VAP/VCL) × 100 | Quantifies movement oscillation |
These multidimensional kinematic analyses provide a comprehensive profile of sperm function that correlates with fertilization potential [31]. AI models integrate these parameters to generate predictive scores for sperm selection in ART procedures, significantly enhancing the objectivity of the selection process [19] [35].
Experimental Protocols and Validation Frameworks
Dataset Creation and Annotation Standards
The development of robust AI models for sperm analysis requires carefully constructed datasets that account for the substantial biological variability in human semen samples. Recent studies have established rigorous protocols for dataset creation, utilizing samples from healthy volunteers aged 18-40 years with prescribed abstinence periods of 2-7 days [7]. Samples exhibiting high viscosity, improper collection, or volume <1.4 mL are typically excluded to maintain standardization [7].
A critical advancement in this domain is the application of confocal laser scanning microscopy (LSM 800) at 40× magnification in confocal mode (LSM, Z-stack) for image acquisition [7]. This approach generates high-resolution images of 512 × 512 pixels, covering an area of 159.7 × 159.7 μm, with a Z-stack interval of 0.5 μm covering a total range of 2 μm [7]. This technical specification enables the capture of subcellular features without the need for staining, preserving sperm viability for subsequent clinical use.
For annotation, embryologists and researchers manually annotate well-focused sperm images using specialized programs like LabelImg, achieving high inter-observer reliability (correlation coefficient of 0.95 for normal sperm morphology and 1.0 for abnormal morphology) [7]. Sperm are categorized according to WHO sixth edition guidelines into multiple classes, with normal morphology requiring meeting all criteria across five frames: smooth oval head with length-to-width ratio of 1.5-2, no vacuoles, slender and regular neck, uniform tail calibre, and cytoplasmic droplets less than one-third of the sperm head [7].
Table 2: Performance Comparison of AI-CASA Versus Traditional Methods
| Assessment Method | Correlation with CASA | Correlation with CSA | Key Advantages | Limitations |
|---|---|---|---|---|
| In-house AI Model (Unstained) | r = 0.88 [7] | r = 0.76 [7] | Non-destructive; suitable for live sperm selection; high accuracy (93%) | Requires specialized imaging equipment |
| Computer-Aided Semen Analysis (CASA) | — | r = 0.57 [7] | Standardized quantification of motility parameters | Requires staining; renders sperm unusable |
| Conventional Semen Analysis (CSA) | r = 0.57 [7] | — | Established reference method | High subjectivity and inter-observer variability |
Model Training and Validation Protocols
The AI model development follows a structured transfer learning approach, typically utilizing pre-trained architectures like ResNet50, which are fine-tuned on sperm morphology datasets [7]. Training involves optimizing the model to minimize the difference between predicted and actual labels through multiple epochs (e.g., 150 epochs), with performance evaluated on separate test datasets not used during training [7]. Studies have demonstrated impressive results with this approach, with one model achieving a precision of 0.95 and recall of 0.91 for detecting abnormal sperm morphology, and 0.91 precision with 0.95 recall for normal sperm morphology [7].
Validation studies often employ prospective designs with statistically powered sample sizes. For instance, one validation study of an AI-enabled CASA system (LensHooke X1 PRO) powered for progressive motility as the primary endpoint assumed a mean increase of +6 percentage points (SD of differences, 12), with a two-sided α = 0.05 and 80% power, requiring a sample size of n=32 [31]. With 20% attrition allowance, the target enrollment was n=40, ultimately enrolling 42 patients with a median age of 31.5 years [31]. Such studies typically assess both conventional parameters (concentration, motility, morphology) and kinematic metrics (VCL, VSL, VAP, ALH, BCF, LIN, STR, WOB), controlling for false discovery rate using methods like Benjamini-Hochberg at q=0.05 [31].
Analysis of Sperm DNA Integrity Using AI
Beyond conventional parameters, AI-CASA systems are increasingly capable of assessing sperm DNA integrity, a crucial factor influencing embryonic development and pregnancy outcomes [7] [19]. While direct measurement typically requires specialized assays, AI models can predict DNA fragmentation levels by analyzing subtle morphological and motility patterns not discernible to the human eye [19]. Research indicates that normal sperm morphology correlates with intact DNA, while high DNA fragmentation adversely affects fertilization and embryonic development [7].
AI algorithms trained on large datasets can identify these correlations, enabling the non-invasive prediction of DNA integrity through routine microscopic analysis. This approach represents a significant advancement, as traditional DNA fragmentation assays are time-consuming, costly, and not routinely performed in all fertility clinics [19]. By integrating these predictive capabilities into standard semen analysis, AI-CASA systems provide a more comprehensive assessment of male fertility potential without additional laboratory procedures.
The Scientist's Toolkit: Essential Research Reagent Solutions
Implementing AI-CASA systems requires specific materials and reagents to ensure standardized and reproducible results. The following table details essential components for establishing these systems in a research or clinical setting:
Table 3: Essential Research Reagents and Materials for AI-CASA Implementation
| Item | Specification/Function | Application Notes |
|---|---|---|
| Confocal Laser Scanning Microscope | LSM 800, 40× magnification, Z-stack capability | Enables high-resolution imaging of unstained sperm; Z-stack interval 0.5 μm [7] |
| Standardized Slides | Two-chamber slide, 20 μm depth (Leja) | Ensures consistent preparation depth for reliable imaging [7] |
| Annotation Software | LabelImg program | Facilitates manual annotation with high inter-observer reliability (coefficient: 0.95-1.0) [7] |
| AI Development Framework | ResNet50 transfer learning model | Deep neural network for image classification; achieves 93% accuracy in morphology assessment [7] |
| Quality Control Standards | Calibration for every 50 samples | Maintains analytical precision; includes focus, illumination, and debris density checks [31] |
| Motility Tracking System | 60 fps frame rate, ≥30 consecutive frames | Enables accurate kinematic parameter calculation and classification [31] |
Technical Implementation and System Architecture
The successful implementation of AI-CASA systems requires careful consideration of both the computational architecture and the biological handling procedures. The system architecture typically follows a multi-stage pipeline that integrates wet laboratory procedures with computational analysis.
A critical consideration in system implementation is the handling of the "black-box" nature of complex AI algorithms. While deep learning models offer exceptional performance, their decision-making processes can be opaque [19]. Emerging approaches address this limitation through explainable AI techniques that highlight the specific features contributing to classification decisions, such as head shape abnormalities, vacuolization, or tail irregularities [4]. This transparency builds trust among embryologists and clinicians, facilitating the adoption of these systems in clinical practice.
Validation and Clinical Integration
Rigorous validation is essential before implementing AI-CASA systems in clinical environments. Recent studies demonstrate effective validation frameworks where urology residents completed structured 8-hour didactic modules on semen analysis principles followed by 10 hours of supervised hands-on sessions with AI-CASA devices [31]. Competency was verified through observed assessments requiring an intra-class correlation coefficient >0.85, with reported inter-operator variability for progressive motility at ICC = 0.89 and intra-operator repeatability at ICC = 0.92 [31].
Clinical validation studies have examined the correlation between AI-CASA findings and therapeutic outcomes. For example, in patients undergoing varicocelectomy, AI-CASA systems detected statistically significant improvements in both conventional and kinematic parameters at 3-month follow-up, demonstrating the system's sensitivity to physiological changes [31]. These improvements included enhanced sperm concentration, total motility, progressive motility, and normal morphology percentages [31].
The integration of AI-CASA systems into clinical workflows offers substantial benefits for ART procedures. By providing objective, standardized, and rapid analysis—with results available approximately one minute after complete semen liquefaction—these systems support clinical decision-making while reducing technician workload [31]. Furthermore, the ability to analyze unstained, live sperm preserves their viability for use in subsequent treatments, addressing a significant limitation of traditional morphology assessment methods [7].
Advanced CASA systems integrating AI for motility, morphology, and DNA integrity assessment represent a paradigm shift in male fertility evaluation. These systems leverage deep learning algorithms to overcome the limitations of traditional semen analysis, providing unprecedented levels of objectivity, accuracy, and efficiency. The strong correlations between AI-based assessments and established methods, coupled with the ability to analyze unstained live sperm, position these technologies as transformative tools in reproductive medicine.
Future research directions should focus on addressing current limitations, including the dependency on large, high-quality annotated datasets and challenges in model generalizability across diverse clinical settings [19] [4]. The development of more standardized, multi-center datasets and the incorporation of explainable AI techniques will be crucial for widespread adoption. Furthermore, longitudinal studies correlating AI-CASA parameters with clinical pregnancy outcomes will strengthen the evidence base for these technologies.
As AI-CASA systems continue to evolve, they hold the promise of ushering in an era of personalized, precision-based fertility care. By providing comprehensive, data-driven insights into sperm quality, these advanced systems empower clinicians to make more informed decisions, ultimately improving outcomes for couples undergoing fertility treatments.
Overcoming Implementation Challenges: Data, Standardization, and Model Optimization
The assessment of sperm morphology is a cornerstone of male fertility evaluation, yet traditional manual methods are plagued by high subjectivity, significant inter-laboratory variability, and substantial reliance on technician expertise [18] [4]. Artificial intelligence (AI) approaches, particularly deep learning, promise to revolutionize this field by enabling automation, standardization, and accelerated analysis [18] [36]. However, the performance and clinical utility of these advanced algorithms are critically dependent on the quality, scale, and standardization of the training data. The fundamental challenge facing the field is that robust AI technologies require large, diverse, and expertly annotated datasets, which are exceptionally difficult and resource-intensive to create and validate [4] [37]. This technical guide examines the core limitations plaguing sperm morphology datasets, details experimental methodologies to overcome these hurdles, and provides standardized frameworks to propel the field toward clinically reliable AI-based assessment systems.
Critical Analysis of Current Dataset Limitations
Scarcity of Standardized, High-Quality Annotated Data
The development of robust deep learning models for sperm morphology analysis requires multidimensional data extraction and analysis, which is severely constrained by the lack of standardized, high-quality annotated datasets [4]. This limitation manifests in several critical ways:
Insufficient Sample Sizes and Limited Diversity: Many existing datasets contain limited numbers of images and lack heterogeneous representation of different morphological classes. For instance, early datasets often comprised only a few hundred to a few thousand images, which is insufficient for training complex deep learning models without overfitting [18] [4]. The SMD/MSS dataset initially contained only 1,000 images, necessitating expansion to 6,035 images through data augmentation techniques [18].
Annotation Subjectivity and Expert Disagreement: Sperm morphology assessment is inherently subjective, leading to significant variability in expert classifications. Studies reveal that even experienced morphologists frequently disagree on classifications, with one study reporting only 51.5% (4,821 out of 9,365 images) achieving 100% consensus among three experts [38]. This subjectivity directly challenges the establishment of reliable ground truth labels essential for supervised learning.
Structural Complexity and Annotation Difficulties: Sperm defect assessment requires simultaneous evaluation of head, vacuoles, midpiece, and tail abnormalities, substantially increasing annotation complexity [4]. Additionally, sperm may appear intertwined in images, or only partial structures may be visible at image edges, further complicating accurate annotation and analysis.
Quantitative Analysis of Dataset Disparities
Table 1: Comparative Analysis of Sperm Morphology Datasets
| Dataset Name | Sample Size | Annotation Classes | Key Features | Reported Model Performance |
|---|---|---|---|---|
| SMD/MSS [18] | 1,000 → 6,035 (after augmentation) | 12 classes (Modified David classification) | Covers head, midpiece, tail anomalies | Accuracy: 55% - 92% |
| SCIAN-MorphoSpermGS [37] | 1,854 sperm head images | 5 classes (Normal, Tapered, Pyriform, Small, Amorphous) | Expert-classification labels from 3 referent experts | Base-line classification established |
| Ram Sperm Dataset [38] | 9,365 individual sperm images | 30-category comprehensive system | High-resolution DIC optics, 100% consensus subset (4,821 images) | Training tool improved accuracy from 53% to 90% |
| HuSHeM [36] | 216 RGB sperm head images | 4 morphological classes | Manual cropping and rotation for standardization | ViT model achieved 93.52% accuracy |
| SMIDS [36] | ~3,000 RGB images | 3 classes (Normal, Abnormal, Non-sperm) | Automatic sperm head-tail rotation-based enhancement | ViT model achieved 92.5% accuracy |
Table 2: Impact of Classification System Complexity on Assessment Accuracy
| Classification System Complexity | Number of Categories | Untrained User Accuracy | Trained User Accuracy | Application Context |
|---|---|---|---|---|
| Binary System | 2 (Normal/Abnormal) | 81.0 ± 2.5% | 98 ± 0.43% | Basic fertility screening |
| Location-Based System | 5 (Head, Midpiece, Tail defects, etc.) | 68 ± 3.59% | 97 ± 0.58% | General diagnostic assessment |
| Specialized System | 8 (Cytoplasmic droplet, Pyriform, etc.) | 64 ± 3.5% | 96 ± 0.81% | Cattle industry standard |
| Comprehensive System | 25-30 (All defects defined individually) | 53 ± 3.69% | 90 ± 1.38% | Research and detailed analysis |
Experimental Protocols for Robust Dataset Creation
Multi-Expert Consensus Framework for Ground Truth Establishment
Establishing reliable ground truth labels represents the most critical challenge in sperm morphology dataset creation. The following protocol details a rigorous multi-expert consensus approach:
Experimental Protocol 1: Ground Truth Establishment through Expert Consensus
Sample Preparation: Collect semen samples with varying morphological profiles from patients or donors with appropriate ethical approvals. For the SMD/MSS dataset, samples were obtained from 37 patients with sperm concentrations of at least 5 million/mL, excluding samples with high concentrations (>200 million/mL) to prevent image overlap [18].
Staining and Slide Preparation: Prepare smears following WHO guidelines and stain with appropriate staining kits (e.g., RAL Diagnostics staining kit) to enhance morphological features [18].
Image Acquisition: Utilize high-resolution microscopy systems. The MMC CASA system with bright field mode and oil immersion 100x objective has been successfully employed [18]. Alternatively, Olympus BX53 microscopes with DIC optics at 40x magnification can capture high-resolution field of view images [38].
Multi-Expert Annotation Process: Engage multiple experienced morphologists (minimum of three) for independent classification. Each expert should classify each spermatozoon according to a standardized classification system (e.g., modified David classification with 12 classes) [18].
Consensus Determination: Establish agreement levels among experts: No Agreement (NA), Partial Agreement (PA: 2/3 experts agree), and Total Agreement (TA: 3/3 experts agree) [18]. Statistical analysis using Fisher's exact test can evaluate differences between experts, with significance set at p < 0.05 [18].
Ground Truth Compilation: Include only images with total expert agreement (TA) or implement a majority voting system for the final ground truth labels. One study achieved a robust dataset by using only the 51.5% of images (4,821 out of 9,365) that achieved 100% expert consensus [38].
Data Augmentation and Preprocessing Pipelines
To address the challenge of limited dataset sizes and class imbalance, implement comprehensive data augmentation protocols:
Experimental Protocol 2: Data Augmentation and Preprocessing
Data Cleaning: Identify and handle poor-quality images, including those with overlapping sperm, debris, or incomplete structures. Manual or automated curation ensures only viable sperm images are included [4].
Normalization/Standardization: Resize images to a standardized dimension (e.g., 80×80×1 grayscale) using linear interpolation strategy to normalize scale across samples [18].
Augmentation Techniques: Apply transformations including rotation, flipping, brightness adjustment, contrast variation, and elastic deformations to artificially expand dataset size and diversity. The SMD/MSS dataset was expanded from 1,000 to 6,035 images through such augmentation techniques [18].
Dataset Partitioning: Split the augmented dataset into training (80%), validation (10-20%), and testing (10-20%) subsets, ensuring representative distribution of all morphological classes across partitions [18].
Advanced Computational Approaches
Vision Transformers and Deep Learning Architectures
Recent advances in computational approaches, particularly vision transformers (ViTs), have demonstrated remarkable capabilities in sperm morphology analysis:
Experimental Protocol 3: Vision Transformer Implementation for Sperm Morphology
Model Selection: Evaluate various ViT variants (BEiT_Base, Swin Transformers) against traditional CNN architectures (VGG16, ResNet) through comprehensive hyperparameter optimization studies [36].
Hyperparameter Optimization: Systematically optimize learning rates, optimization algorithms, and data augmentation scales. Studies have shown that data augmentation significantly enhances ViT performance by improving generalization, particularly in limited-data scenarios [36].
Training Strategy: Implement end-to-end training that processes raw sperm images without manual pre-processing, eliminating labor-intensive steps and enabling full automation [36].
Interpretability Analysis: Utilize visualization techniques (Attention Maps, Grad-CAM) to validate the model's ability to capture discriminative morphological features, such as head shape and tail integrity [36].
Comparative studies demonstrate that transformer-based architectures consistently outperform traditional methods, with the BEiT_Base model achieving state-of-the-art accuracies of 92.5% (SMIDS) and 93.52% (HuSHeM), surpassing prior CNN-based approaches by 1.63% and 1.42%, respectively [36].
Standardized Training and Evaluation Frameworks
The development of standardized training tools has shown significant promise in improving assessment accuracy and reducing variability:
Experimental Protocol 4: Standardized Training Tool Implementation
Tool Design: Create interactive web-based interfaces that provide instant feedback to users on correct/incorrect labels for training purposes, along with proficiency assessment capabilities [38].
Adaptive Classification Systems: Structure the tool to accommodate various classification systems (2-category to 30-category systems) to ensure broad applicability across different clinical and research contexts [38].
Validation Protocol: Conduct studies with novice morphologists to assess baseline accuracy and improvement through training. One study demonstrated significant improvement in accuracy (from 53% to 90% in complex 25-category systems) and diagnostic speed (from 7.0±0.4s to 4.9±0.3s per image) after repeated training over four weeks [5].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents and Materials for Sperm Morphology Dataset Creation
| Item | Specification/Function | Application Context |
|---|---|---|
| Microscopy System | High-resolution with DIC optics (e.g., Olympus BX53), 40x-100x objectives | High-quality image acquisition [38] |
| Digital Camera | High-megapixel CMOS sensor (e.g., Olympus DP28, 8.9-megapixel) | High-resolution image capture [38] |
| Staining Kits | RAL Diagnostics staining kit or Modified Hematoxylin/Eosin procedure | Enhanced morphological visualization [18] [37] |
| CASA System | MMC CASA system for automated image acquisition and analysis | Standardized image capture and initial morphometric analysis [18] |
| Annotation Software | Web-based annotation tools for multi-expert classification | Efficient ground truth labeling [38] |
| Data Augmentation Tools | Python libraries (TensorFlow, PyTorch) with image transformation capabilities | Dataset expansion and balancing [18] |
| Computational Resources | GPU-accelerated workstations for deep learning model training | ViT and CNN model development [36] |
The limitations in dataset quality, annotation consistency, and standardization represent significant hurdles in the development of robust AI-based sperm morphology assessment systems. However, rigorous experimental protocols involving multi-expert consensus, comprehensive data augmentation, and advanced computational approaches like vision transformers provide promising pathways to overcome these challenges. The creation of standardized, high-quality datasets with validated ground truth labels, coupled with the implementation of standardized training tools, will be essential for translating AI-based sperm morphology assessment from research laboratories to clinical practice. Future efforts should focus on establishing international standards for dataset creation, promoting data sharing initiatives, and developing more sophisticated annotation tools that can further reduce subjectivity and improve consistency across institutions.
Feature engineering and selection represent fundamental processes in machine learning that significantly enhance model performance, interpretability, and computational efficiency. Within the specialized domain of sperm morphology assessment, these techniques bridge the gap between raw image data and clinically actionable diagnostic information. Traditional manual sperm morphology analysis suffers from substantial limitations, including significant inter-observer variability reaching up to 40% disagreement between expert evaluators, lengthy evaluation times (30-45 minutes per sample), and inconsistent standards across laboratories [39] [8]. These challenges have accelerated the adoption of artificial intelligence (AI) approaches, where feature engineering plays a pivotal role in transforming subjective visual assessments into quantifiable, reproducible metrics.
The evolution from conventional machine learning to deep learning-based approaches has transformed the paradigm of feature extraction in medical image analysis. Conventional computer vision techniques for sperm morphology analysis relied on manually designed features such as shape descriptors, grayscale intensity, edge detection, and contour analysis [20] [4]. These methods achieved moderate success, with one Bayesian Density Estimation-based model reporting 90% accuracy in classifying sperm heads into four morphological categories [4]. However, their fundamental limitation lay in the dependency on human expertise to identify and engineer relevant features, which constrained their ability to capture the subtle morphological variations critical for accurate fertility assessment.
Contemporary deep learning frameworks have automated the feature extraction process, enabling models to learn hierarchical representations directly from image data. The integration of feature engineering within deep learning architectures has yielded remarkable performance improvements, with one recent approach combining Convolutional Block Attention Module (CBAM) with ResNet50 architecture and achieving test accuracies of 96.08% ± 1.2% on the SMIDS dataset and 96.77% ± 0.8% on the HuSHeM dataset [39] [8]. These results demonstrate significant improvements of 8.08% and 10.41% respectively over baseline convolutional neural network performance, highlighting the critical importance of sophisticated feature processing in medical image analysis.
Traditional vs. AI-Based Feature Engineering Approaches
Conventional Feature Engineering Methodologies
Traditional computer vision approaches for sperm morphology analysis established the foundational framework for feature engineering in this domain. These methods employed carefully designed image processing pipelines to extract quantifiable characteristics from sperm images, with particular focus on morphological parameters aligned with World Health Organization (WHO) guidelines [20] [4].
Table 1: Conventional Feature Engineering Techniques in Sperm Morphology Analysis
| Feature Category | Specific Techniques | Application in Sperm Analysis | Performance Limitations |
|---|---|---|---|
| Shape Descriptors | Hu moments, Zernike moments, Fourier descriptors | Quantification of head shape abnormalities (tapered, pyriform, amorphous) | Accuracy up to 90% for head classification only [4] |
| Texture Features | Gray-level co-occurrence matrix (GLCM), Local Binary Patterns (LBP) | Analysis of acrosome integrity, vacuole presence | Limited to stained, high-resolution images [20] |
| Color Features | Color space transformations (RGB, HSV, Lab), histogram statistics | Segmentation of acrosome and nucleus in stained specimens [4] | Not applicable to unstained sperm |
| Geometric Features | Length-to-width ratios, area, perimeter, eccentricity | Assessment of head dimensions according to WHO standards [7] | Inability to capture complex structural relationships |
The technical implementation of these conventional approaches typically followed a standardized pipeline. First, preprocessing steps such as wavelet denoising and directional masking were applied to enhance image quality [8]. Next, segmentation algorithms like k-means clustering combined with histogram statistical methods isolated sperm components (head, midpiece, tail) [4]. Subsequently, feature extraction algorithms quantified morphological attributes, and finally, classifiers such as Support Vector Machines (SVM) with linear or radial basis function (RBF) kernels performed the categorization [20] [4].
A notable implementation by Chang et al. utilized Fourier descriptors and SVM to classify non-normal sperm heads but achieved only 49% accuracy, highlighting the fundamental limitations of handcrafted features in capturing the complex morphological variations in sperm cells [4]. Similarly, Mirsky et al. trained an SVM classifier on manually extracted features from over 1,400 human sperm cells, achieving 88.59% area under the receiver operating characteristic curve (AUC-ROC) but with restricted generalizability across different imaging conditions [4].
Deep Learning-Based Feature Engineering
The advent of deep learning has fundamentally transformed feature engineering from a manual, expertise-dependent process to an automated, data-driven paradigm. Convolutional Neural Networks (CNNs) automatically learn hierarchical feature representations directly from raw pixel data, capturing both low-level visual patterns (edges, textures) and high-level morphological concepts (head shape abnormalities, tail defects) [39] [8].
Advanced deep learning frameworks have incorporated attention mechanisms and structured feature engineering pipelines to further enhance performance. The CBAM-enhanced ResNet50 architecture represents a significant innovation in this domain, integrating channel and spatial attention modules to enable the network to focus on morphologically relevant regions while suppressing irrelevant background information [39] [8]. This approach demonstrates how modern feature engineering moves beyond simple feature extraction to include feature weighting and selection within the learning process.
Table 2: Performance Comparison of Feature Engineering Approaches on Benchmark Datasets
| Methodology | SMIDS Dataset Accuracy | HuSHeM Dataset Accuracy | Feature Engineering Approach | Clinical Interpretability |
|---|---|---|---|---|
| Traditional ML (SVM with handcrafted features) | ~49-87% [8] [4] | ~88% [4] | Manual feature design and selection | Moderate (features directly correspond to morphological traits) |
| Baseline CNN | 88.00% [39] | 86.36% [39] | Automated feature learning without specialization | Low (black-box representation) |
| CBAM-ResNet50 with Deep Feature Engineering | 96.08% ± 1.2% [39] | 96.77% ± 0.8% [39] | Hybrid: automated learning + structured selection | High (Grad-CAM visualization of attention maps) |
The integration of deep feature engineering (DFE) represents a sophisticated hybrid approach that combines the representational power of deep neural networks with classical feature selection techniques. This methodology extracts high-dimensional feature representations from intermediate layers of pre-trained networks, applies dimensionality reduction and feature selection techniques, and employs shallow classifiers for final prediction [8]. The optimal configuration identified in recent research (GAP + PCA + SVM RBF) demonstrates how strategic feature processing after deep learning extraction can yield substantial performance improvements [39].
Experimental Protocols and Implementation Frameworks
CBAM-Enhanced ResNet50 with Deep Feature Engineering
Architecture Specification
The integrated framework combines a ResNet50 backbone with Convolutional Block Attention Module (CBAM) and a comprehensive deep feature engineering pipeline [39] [8]. The technical implementation follows a multi-stage process:
Backbone Feature Extraction: Utilizing ResNet50 pre-trained on ImageNet as the foundational feature extractor, with weights fine-tuned on sperm morphology datasets during training.
Attention Mechanism Integration: Incorporating CBAM sequentially applies channel and spatial attention to intermediate feature maps. The channel attention module uses both max-pooling and average-pooling features followed by a multi-layer perceptron, while the spatial attention module employs similar pooling operations along the channel axis followed by a convolution layer.
Multi-Source Feature Extraction: Harvesting features from four distinct layers: CBAM attention weights, Global Average Pooling (GAP), Global Max Pooling (GMP), and pre-final fully connected layers.
Feature Selection Pipeline: Applying 10 distinct feature selection methods including Principal Component Analysis (PCA), Chi-square test, Random Forest importance, variance thresholding, and their intersections to reduce dimensionality and retain the most discriminative features.
Classification: Utilizing Support Vector Machines with RBF/Linear kernels and k-Nearest Neighbors algorithms on the processed feature set for final categorization.
Experimental Configuration and Training Protocol
The model was rigorously evaluated using 5-fold cross-validation on two benchmark datasets: SMIDS (3000 images, 3-class) and HuSHeM (216 images, 4-class) [39]. Training implemented transfer learning with initial weights from ImageNet-pre-trained ResNet50, with fine-tuning of all layers. Optimization used stochastic gradient descent (SGD) with momentum of 0.9, initial learning rate of 0.001 with cosine decay, and batch size of 32. Data augmentation techniques included random rotation (±15°), horizontal and vertical flipping, and brightness/contrast variations (±20%).
The deep feature engineering pipeline specifically extracted 2048-dimensional feature vectors from the GAP layer, which were subsequently reduced to 150 principal components using PCA, accounting for 95% of variance. SVM classifiers with RBF kernels were trained with cross-validated hyperparameter tuning for regularization parameter C and kernel coefficient γ [39].
Performance metrics included accuracy, precision, recall, F1-score, and McNemar's test for statistical significance comparing different configurations. The model achieved its superior performance of 96.08% ± 1.2% on SMIDS and 96.77% ± 0.8% on HuSHeM using the GAP + PCA + SVM RBF configuration, demonstrating statistically significant improvements (p < 0.05) over baseline approaches [39].
ResNet50 Transfer Learning for Unstained Sperm Analysis
Methodology for Live Sperm Assessment
An alternative implementation focused specifically on unstained live sperm morphology assessment utilizing ResNet50 transfer learning without CBAM enhancement [7]. This approach addressed the critical clinical need for analyzing viable sperm without staining procedures that render sperm unusable for assisted reproductive technologies.
The experimental protocol encompassed:
Dataset Preparation: Creating a novel dataset of sperm images captured with confocal laser scanning microscopy at 40× magnification in confocal mode (Z-stack with 0.5 μm interval). The dataset comprised 21,600 images with 12,683 annotated unstained sperm instances.
Annotation Protocol: Manual annotation by embryologists and researchers using LabelImg program, with inter-observer correlation coefficients of 0.95 for normal sperm morphology detection and 1.0 for abnormal morphology detection.
Classification Criteria: Categorizing each sperm image into nine datasets based on WHO criteria, including normal sperm with smooth oval head (length-to-width ratio of 1.5-2), no vacuoles, slender regular neck, uniform tail calibre, and cytoplasmic droplets less than one-third of the sperm head.
Model Training: Implementing transfer learning with ResNet50, trained on a subset of 9,000 images (4,500 normal, 4,500 abnormal) for 150 epochs with batch size of 32.
This approach achieved a test accuracy of 93%, with precision of 0.95 and recall of 0.91 for detecting abnormal sperm morphology, and precision of 0.91 and recall of 0.95 for normal sperm morphology [7]. The model's processing time was approximately 139.7 seconds for 25,000 images, enabling rapid analysis at approximately 0.0056 seconds per image.
Technical Implementation: Feature Engineering Workflows
Deep Feature Engineering Pipeline Architecture
The deep feature engineering pipeline represents a structured methodology for transforming raw image data into discriminative feature representations optimized for sperm morphology classification [39] [8]. The technical implementation involves sequential processing stages:
The feature selection phase incorporates multiple complementary approaches [39]:
- Principal Component Analysis (PCA): Linear dimensionality reduction preserving maximum variance while decorrelating features
- Chi-square Test: Filter-based selection of features with strongest statistical dependence on classification target
- Random Forest Importance: Embedded method using tree-based feature importance scores
- Variance Thresholding: Removal of low-variance features unlikely to contain discriminative information
- Intersection Methods: Combining multiple selection techniques to identify robust feature subsets
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Materials and Computational Resources for Sperm Morphology Analysis
| Resource Category | Specific Items | Technical Specification | Research Application |
|---|---|---|---|
| Imaging Hardware | Confocal Laser Scanning Microscope (LSM 800) [7] | 40× magnification, Z-stack interval 0.5 μm, frame time 633.03 ms, 512×512 pixels | High-resolution capture of unstained live sperm |
| Sample Preparation | Optixcell extender [40] | Pre-warmed at 37°C, 1:1 ratio (v/v) with semen | Sperm dilution maintaining viability |
| Staining Reagents | Diff-Quik stain [7] | Romanowsky stain variant | Conventional staining for fixed sperm morphology |
| Annotation Software | LabelImg program [7] | Python-based graphical image annotation tool | Manual bounding box annotation for dataset creation |
| Deep Learning Frameworks | TensorFlow/PyTorch with ResNet50/CBAM [39] [8] | Pre-trained on ImageNet, fine-tuned on sperm datasets | Backbone architecture for feature extraction |
| Feature Engineering Libraries | Scikit-learn [39] | PCA, SVM, feature selection implementations | Traditional ML components in hybrid pipeline |
| Evaluation Metrics | 5-fold cross-validation [39] | Accuracy, precision, recall, F1-score, McNemar's test | Robust performance assessment and statistical validation |
Discussion and Clinical Implications
The strategic implementation of feature engineering and selection methodologies has demonstrated significant impact on both technical performance and clinical utility in sperm morphology analysis. The integration of attention mechanisms with structured feature processing pipelines represents a paradigm shift from black-box deep learning toward interpretable, clinically actionable AI systems.
The CBAM-enhanced ResNet50 with deep feature engineering achieves superior performance not only through increased accuracy but also via enhanced interpretability. The attention mechanisms generate Grad-CAM visualizations that highlight morphologically relevant regions, providing embryologists with intuitive explanations for classification decisions [39] [8]. This interpretability is crucial for clinical adoption, as it aligns AI decision-making with established embryological expertise and WHO morphological criteria.
From a clinical implementation perspective, these advanced feature engineering approaches address critical limitations of conventional semen analysis. The automation of sperm morphology assessment reduces analysis time from 30-45 minutes to less than 1 minute per sample while simultaneously improving consistency and reducing inter-observer variability [39]. Furthermore, the application of these techniques to unstained sperm imaging preserves sperm viability for subsequent use in assisted reproductive technologies, creating new possibilities for real-time sperm selection during intracytoplasmic sperm injection (ICSI) procedures [7] [41].
The future evolution of feature engineering in sperm morphology analysis will likely focus on multi-modal integration, combining morphological features with motility parameters, DNA fragmentation indices, and metabolic markers to develop comprehensive sperm quality assessment systems. Additionally, continued refinement of explainable AI techniques will further enhance clinical trust and adoption, ultimately improving patient care and treatment outcomes in reproductive medicine.
The diagnostic evaluation of sperm morphology remains a cornerstone of male fertility assessment, profoundly influencing treatment pathways in assisted reproductive technologies (ART). Traditional manual morphology assessment, as outlined by the World Health Organization (WHO) guidelines, is characterized by inherent subjectivity, significant inter-observer variability (with reported kappa values as low as 0.05–0.15), and labor-intensive processes that require examining at least 200 sperm per sample, often taking 30–45 minutes per case [8] [42]. This methodological variability challenges the reliability and reproducibility of diagnostic results across laboratories, complicating clinical decision-making and the prognostic forecasting of ART success [3] [5]. Consequently, expert groups have even begun to question the prognostic value of traditional sperm morphology assessment for procedures like IUI, IVF, or ICSI [3].
Artificial intelligence (AI), particularly through machine learning (ML) and deep learning (DL), is poised to bridge this critical gap in diagnostic precision. AI-driven approaches automate sperm morphology analysis, offering objective, rapid, and highly consistent evaluations by leveraging advanced pattern recognition in image and video data [19] [42]. The transition from traditional to AI-based assessment represents a paradigm shift from subjective visual inspection to an era of data-driven, quantifiable diagnostic metrics. The core of this transition's success lies in the rigorous application of strategic algorithm selection and systematic hyperparameter tuning, which are fundamental to developing robust, generalizable, and clinically deployable AI models that enhance diagnostic accuracy beyond human capability [8].
Algorithm Selection: Architecting the Foundation for Accurate Morphology Classification
Selecting an appropriate algorithm is a foundational decision that dictates the potential performance and clinical applicability of an AI model for sperm morphology analysis. The choice is primarily governed by the nature of the available data, the complexity of the morphological classification task, and the computational constraints of the clinical environment.
Deep Learning Architectures for Image-Based Classification
Convolutional Neural Networks (CNNs) represent the dominant architectural paradigm for analyzing sperm images, given their proven efficacy in extracting hierarchical features directly from pixel data.
- ResNet50 with Attention Mechanisms: The integration of ResNet50, a deep residual network, with Convolutional Block Attention Module (CBAM) has demonstrated state-of-the-art performance. The CBAM component enables the network to focus adaptively on diagnostically salient morphological features, such as head shape or acrosome integrity, while suppressing irrelevant background information. This architecture, when combined with a deep feature engineering pipeline, has achieved exceptional test accuracies of 96.08% on the SMIDS dataset and 96.77% on the HuSHeM dataset, significantly outperforming baseline CNN models [8].
- EfficientNetV2 for Feature Extraction: The EfficientNetV2 family of models provides a strong balance between computational efficiency and representational power, making them suitable for feature extraction within ensemble frameworks. These models are often used as backbone networks to generate rich, discriminative feature sets for subsequent classification [43].
- Multi-Level Ensemble Learning: For complex classification tasks involving numerous morphological abnormality categories, a multi-level ensemble approach that combines feature-level and decision-level fusion has shown remarkable success. This strategy involves extracting features from multiple CNN architectures and fusing them to create a more robust feature vector. Classification is then performed using combinations of Support Vector Machines (SVM), Random Forest (RF), and Multi-Layer Perceptron with Attention (MLP-Attention), culminating in a final decision via soft voting. This sophisticated ensemble method achieved an accuracy of 67.70% on the challenging Hi-LabSpermMorpho dataset, which encompasses 18 distinct sperm morphology classes, substantially outperforming individual classifiers [43].
Traditional Machine Learning with Engineered Features
While deep learning excels with large image datasets, traditional machine learning algorithms remain relevant, particularly when integrated with deep feature engineering or when data is limited.
- Support Vector Machines (SVM): SVMs are powerful for binary and multi-class classification. Their performance is heavily dependent on the kernel selection and the quality of the input features. When trained on deep features extracted from pre-trained CNNs (a hybrid approach), SVMs with Radial Basis Function (RBF) kernels have demonstrated superior performance, achieving high accuracy in distinguishing normal from abnormal sperm forms [8] [43].
- Hybrid MLFFN–ACO Framework: For non-image clinical and lifestyle data, a hybrid framework combining a Multilayer Feedforward Neural Network (MLFFN) with a nature-inspired Ant Colony Optimization (ACO) algorithm has been developed. The ACO component optimizes network parameters through adaptive tuning, mimicking ant foraging behavior. This hybrid system reported a remarkable 99% classification accuracy in predicting male fertility status from a clinical dataset, highlighting the potency of bio-inspired optimization in model training [44].
Table 1: Performance Comparison of Selected Algorithms for Sperm Morphology Analysis
| Algorithm | Dataset | Key Features | Reported Accuracy | Best For |
|---|---|---|---|---|
| ResNet50 + CBAM + DFE [8] | SMIDS, HuSHeM | Attention mechanisms, deep feature engineering, PCA + SVM RBF | 96.08% - 96.77% | High-accuracy, interpretable image classification |
| Multi-Level Ensemble [43] | Hi-LabSpermMorpho (18 classes) | Feature/decision-level fusion, Multiple EfficientNetV2, Soft voting | 67.70% | Complex multi-class classification |
| Hybrid MLFFN-ACO [44] | UCI Fertility Dataset | Bio-inspired optimization, handles clinical/lifestyle data | 99.00% | Non-image clinical data analysis |
| SVM with Deep Features [8] [43] | Various | Hybrid CNN+SVM, effective on high-dimensional features | High (Specific % not listed) | Scenarios where deep features are available |
Hyperparameter Tuning: Optimizing Model Performance for Clinical Reliability
Hyperparameter tuning is the process of systematically searching for the optimal combination of model configuration settings that are not learned during training. This process is critical for maximizing a model's predictive performance and ensuring its robustness and generalizability to new, unseen clinical data.
Core Hyperparameters and Optimization Techniques
The selection of hyperparameters and the method for tuning them must align with the chosen algorithm.
- Bio-Inspired Optimization: The Ant Colony Optimization (ACO) algorithm, as implemented in the hybrid MLFFN-ACO framework, exemplifies a powerful metaheuristic approach. It automates the search for optimal network weights and biases by simulating the behavior of ants seeking paths to food sources. This method has proven highly effective, contributing to models that achieve near-perfect accuracy while requiring an ultra-low computational time of just 0.00006 seconds for prediction, demonstrating feasibility for real-time clinical use [44].
- Classical Feature Selection and Dimensionality Reduction: In deep feature engineering pipelines, techniques like Principal Component Analysis (PCA) are crucial hyperparameter-like components. PCA is used to reduce noise and the dimensionality of deep feature vectors extracted from CNNs before feeding them into a classifier like an SVM. One study demonstrated that applying PCA to ResNet50-CBAM features before SVM classification boosted accuracy by approximately 8 percentage points, from 88% to 96.08%, underscoring the profound impact of this step [8].
- Grid Search and Cross-Validation: For traditional classifiers like SVM and Random Forest, a comprehensive tuning strategy is essential. This involves:
- SVM Hyperparameters:
C(regularization parameter) andgamma(kernel coefficient) for RBF kernels.Ccontrols the trade-off between achieving a low training error and a low testing error, whilegammadefines how far the influence of a single training example reaches. - Random Forest Hyperparameters:
n_estimators(number of trees in the forest),max_depth(maximum depth of each tree), andmin_samples_split(minimum number of samples required to split a node). - Validation Method: Utilizing 5-fold cross-validation during the tuning process provides a robust estimate of model performance and helps mitigate overfitting [8] [43].
- SVM Hyperparameters:
Addressing Data-Specific Challenges
Two pervasive challenges in medical AI are class imbalance and the need for model interpretability, both of which can be addressed through targeted strategies.
- Handling Class Imbalance: Fertility datasets often have significantly more "normal" than "altered" or specific abnormality cases. Techniques such as the Proximity Search Mechanism (PSM) can be integrated to improve sensitivity to these underrepresented classes, ensuring the model does not become biased toward the majority class [44].
- Ensuring Clinical Interpretability: For AI to be adopted in clinical practice, its decisions must be interpretable. Using Grad-CAM (Gradient-weighted Class Activation Mapping) visualization with CBAM-enhanced models allows clinicians to see which parts of a sperm image (e.g., the head vacuoles or tail structure) the model focused on to make its classification, building essential trust in the AI system [8].
Experimental Protocols and Research Workflows
Translating algorithmic concepts into validated diagnostic tools requires meticulously designed experimental protocols. The following workflow outlines a standard methodology for developing and validating an AI model for sperm morphology assessment.
Diagram: AI-Based Sperm Morphology Analysis Workflow.
Detailed Protocol for an AI-Based Morphology Study
1. Sample Collection and Preparation:
- Collect semen samples from volunteers (e.g., n=30) after 2-7 days of sexual abstinence, following WHO guidelines [7].
- Ensure sample liquefaction and perform initial conventional analysis for concentration and motility using a Computer-Aided Semen Analysis (CASA) system.
2. Image Acquisition and Dataset Curation:
- Dispense a 6 µL semen droplet onto a two-chamber slide with a 20 µm depth.
- Capture images using a Confocal Laser Scanning Microscope (LSM) at 40x magnification in Z-stack mode (e.g., interval of 0.5 µm over a 2 µm range) to generate high-resolution, multi-focal plane images [7].
- Manually annotate at least 200 sperm images per sample using a tool like LabelImg. Annotation should be performed by multiple expert embryologists to establish a "ground truth" label for each sperm via consensus, a critical step for reducing label noise. Categories should align with WHO criteria and can range from simple 2-category (normal/abnormal) to more complex systems (e.g., 5, 8, or 25 categories for specific defects) [7] [5].
3. Model Training with Hyperparameter Tuning:
- Split the curated dataset into training, validation, and test sets (e.g., 70/15/15).
- For a ResNet50-CBAM model, initiate transfer learning. The tuning phase should involve:
- Integrating the CBAM attention modules to allow the network to focus on salient features.
- Extracting deep features from a pre-final layer and applying PCA for dimensionality reduction.
- Training an SVM classifier with an RBF kernel on the reduced feature set, using the validation set to optimize the
Candgammahyperparameters via grid search [8].
- Alternatively, for an ensemble method, train multiple CNN backbones (e.g., EfficientNetV2 variants), perform feature-level fusion, and apply a meta-classifier (e.g., MLP-Attention) with its own set of tuned hyperparameters [43].
4. Model Evaluation and Clinical Validation:
- Evaluate the final model on the held-out test set, reporting standard metrics: accuracy, precision, recall, F1-score, and area under the receiver operating characteristic curve (AUC-ROC).
- Validate model performance using 5-fold cross-validation to ensure robustness [8].
- Perform clinical validation by comparing the AI's morphology assessments and its correlation with clinical outcomes (e.g., fertilization rates in ICSI) against the gold standard of manual assessment by expert embryologists [7].
Table 2: Key Research Reagent Solutions for AI-Based Sperm Morphology Analysis
| Item / Solution | Function / Application | Specification / Notes |
|---|---|---|
| Confocal Laser Scanning Microscope [7] | High-resolution image acquisition of live, unstained sperm. | Enables Z-stack imaging at 40x magnification; crucial for capturing subcellular features without staining. |
| LEJA Standard Slides [7] | Sample preparation for morphology analysis. | Two-chamber slides with 20 µm depth; standardizes preparation for consistent imaging. |
| Diff-Quik Stain [7] | Staining for conventional and CASA-based morphology assessment. | Romanowsky stain variant; used for fixed sperm in comparator methods. |
| LabelImg Program [7] | Manual annotation of sperm images for ground truth creation. | Creates bounding boxes; essential for supervised learning model training. |
| ResNet50 / EfficientNetV2 Models [8] [43] | Deep learning backbone for feature extraction and classification. | Pre-trained on ImageNet; can be fine-tuned with sperm image data. |
| Ant Colony Optimization (ACO) [44] | Bio-inspired hyperparameter and weight optimization. | Used in hybrid models for adaptive parameter tuning, improving convergence and accuracy. |
| HuSHeM / SMIDS / Hi-LabSpermMorpho Datasets [8] [43] | Benchmark public datasets for training and validation. | Provide standardized data for developing and comparing algorithm performance. |
The integration of artificial intelligence into sperm morphology assessment marks a definitive leap from subjective, variable manual methods toward precise, automated, and data-driven diagnostics. This transition is critically dependent on the foundational pillars of strategic algorithm selection—choosing the right architecture like ResNet50 with CBAM or multi-level ensembles for the task at hand—and rigorous hyperparameter tuning using techniques such as Ant Colony Optimization or PCA with grid search. These processes are not merely technical exercises; they are essential for transforming raw data and algorithmic potential into clinically reliable tools that achieve accuracies exceeding 96% in some studies [8].
The future trajectory of this field points toward more sophisticated, integrated systems. Priorities will include the development of larger, more diverse, and publicly available annotated datasets to combat overfitting and improve generalizability [4] [43]. Furthermore, the "black-box" nature of complex models will be addressed through the increased use of explainable AI (XAI) techniques, making AI decisions transparent and trustworthy for clinicians [8] [44]. As these technologies mature and undergo rigorous multicenter validation, they hold the undeniable potential to standardize male fertility diagnostics globally, personalize treatment selection, and ultimately improve success rates for couples seeking assisted reproduction.
Sperm morphology assessment is a cornerstone of male fertility evaluation, recognized as one of the three key foundational semen quality assessments alongside concentration and motility [5]. Despite its clinical importance, morphology analysis remains one of the most challenging and variable tests in andrology laboratories due to its highly subjective nature [6]. This subjectivity stems from multiple factors: differences in staining techniques, variations in the application of classification criteria, inter-laboratory procedural differences, and the inherent challenge of visual classification of complex cellular structures [6]. The problem is further compounded by the lack of standardized training protocols for morphologists, leading to significant inter-observer and intra-observer variability that compromises result reliability and clinical utility [5].
The clinical implications of this standardization crisis are substantial. Sperm morphology assessment serves as a critical tool for diagnosing male infertility and determining appropriate treatment pathways, with morphology results influencing decisions between intrauterine insemination (IUI), in vitro fertilization (IVF), and intracytoplasmic sperm injection (ICSI) [6]. Traditional assessment requires staining and high magnification (100×), which renders sperm unsuitable for further clinical use [7]. The World Health Organization has progressively revised reference values for normal sperm morphology from ≥80.5% in the first edition to ≥4% in the most recent edition, reflecting evolving understanding and persistent challenges in standardization [6].
This technical guide examines both traditional and artificial intelligence (AI)-based approaches to sperm morphology assessment, with particular focus on training methodologies, standardization tools, and their integration into clinical practice. By comparing established training protocols with emerging AI technologies, we aim to provide researchers and clinicians with a comprehensive framework for improving assessment accuracy and reliability in both human andrology and drug development contexts.
Traditional Training Approaches and Methodologies
Current Challenges in Morphologist Training
Without standardized training, sperm morphology assessment exhibits unacceptably high variability among morphologists. A recent study evaluating novice morphologists' accuracy across different classification systems revealed fundamental challenges in training effectiveness. Untrained users achieved accuracy rates of 81.0 ± 2.5% with simple 2-category systems (normal/abnormal), but performance significantly declined to 53 ± 3.69% with complex 25-category classification systems [5]. This performance degradation with system complexity highlights the cognitive load involved in morphological assessment and underscores the need for specialized training protocols.
The variability among untrained users is particularly concerning, with coefficients of variation (CV) reaching 0.28 and accuracy scores ranging dramatically from 19% to 77% among individuals with identical training backgrounds [5]. This variation persists despite established WHO guidelines that provide detailed criteria for normal sperm morphology: the sperm head should be oval-shaped, smooth, and regularly contoured, measuring 5-6μm in length and 2.5-3.5μm in width; the acrosome must occupy 40%-70% of the head area with no more than two small vacuoles occupying ≤20% of the area; the mid-piece should be slender, approximately the same length as the head, and aligned with its axis; the tail should be approximately 45μm long, uniform, and without sharp bends [6].
The Sperm Morphology Assessment Standardisation Training Tool
A groundbreaking development in traditional training methodology comes from the "Sperm Morphology Assessment Standardisation Training Tool," which applies machine learning principles of supervised learning and expert consensus labels ("ground truth") to human training [5]. This tool addresses the critical need for traceable standards in morphology assessment by providing:
- Expert Consensus Ground Truth: Images classified by multiple experts to establish validated reference standards
- Adaptable Classification Systems: Compatibility with 2-category, 5-category, 8-category, and 25-category systems
- Progressive Training Modules: Structured learning pathways from simple to complex classification tasks
- Quantitative Performance Metrics: Continuous assessment of accuracy, variability, and diagnostic speed
The training tool's effectiveness was validated through experiments with two cohorts of novice morphologists. The first cohort (n=22) demonstrated the baseline challenges, while a second cohort (n=16) exposed to visual aids and training videos achieved significantly improved first-test accuracy across all classification systems: 94.9 ± 0.66% (2-category), 92.9 ± 0.81% (5-category), 90 ± 0.91% (8-category), and 82.7 ± 1.05% (25-category) [5].
Table 1: Impact of Standardized Training on Morphologist Accuracy
| Classification System | Untrained Accuracy (%) | Trained Accuracy (%) | Final Accuracy After 4 Weeks (%) |
|---|---|---|---|
| 2-category (normal/abnormal) | 81.0 ± 2.5 | 94.9 ± 0.66 | 98 ± 0.43 |
| 5-category (by defect location) | 68 ± 3.59 | 92.9 ± 0.81 | 97 ± 0.58 |
| 8-category (cattle industry standard) | 64 ± 3.5 | 90 ± 0.91 | 96 ± 0.81 |
| 25-category (individual defects) | 53 ± 3.69 | 82.7 ± 1.05 | 90 ± 1.38 |
Experimental Protocol for Traditional Training Validation
The validation study for the training tool consisted of two structured experiments [5]:
Experiment 1: Baseline Assessment and Initial Training
- Participants: 22 novice morphologists
- Assessment across four classification systems (2-category, 5-category, 8-category, 25-category)
- Intervention: Visual aids and training videos
- Metrics: Accuracy, time per classification, variability
Experiment 2: Longitudinal Training Effectiveness
- Participants: 16 novice morphologists
- Duration: 4 weeks with repeated training sessions
- Testing schedule: 14 tests over the training period
- Metrics: Accuracy progression, diagnostic speed, inter-observer variability
The results demonstrated significant improvement in both accuracy (from 82 ± 1.05% to 90 ± 1.38%) and diagnostic speed (from 7.0 ± 0.4s to 4.9 ± 0.3s per image) over the training period [5]. This protocol provides a validated framework for laboratory training programs and highlights the potential for standardized approaches to reduce variability in morphological assessment.
AI-Based Assessment Technologies
Development and Validation of AI Models
Artificial intelligence approaches to sperm morphology assessment represent a paradigm shift from traditional subjective methods to objective, automated systems. Recent research demonstrates the development of sophisticated AI models capable of analyzing unstained live sperm using confocal laser scanning microscopy at low magnification (40×) with high resolution [7]. This approach addresses a critical limitation of traditional methods, which require staining and high magnification (100×) that renders sperm unusable for subsequent clinical procedures.
The technical architecture of these AI systems typically utilizes deep learning models, with ResNet50 transfer learning emerging as a particularly effective framework for image classification tasks [7]. These models are trained on novel datasets of sperm morphological images captured using confocal laser scanning microscopy in LSM Z-stack mode at 0.5μm intervals, covering a total range of 2μm [7]. The image acquisition protocol specifies:
- Frame time: 633.03 ms
- Image size: 512 × 512 pixels
- Field size: 159.7 × 159.7 μm
- Minimum of 200 sperm images per sample, each containing 2-3 sperm per capture
Performance Metrics and Clinical Validation
In experimental studies comparing AI assessment with traditional methods, the in-house AI model demonstrated superior correlation with computer-aided semen analysis (CASA) (r = 0.88) compared to conventional semen analysis (r = 0.76) [7]. The correlation between CASA and conventional semen analysis was notably weaker (r = 0.57), highlighting the significant variability in traditional approaches [7].
Table 2: Performance Comparison of Morphology Assessment Methods
| Assessment Method | Correlation with AI Model | Normal Morphology Detection Rate | Key Advantages | Limitations |
|---|---|---|---|---|
| In-house AI Model | Self | 93% test accuracy | Objective, works with live sperm, high throughput | Requires specialized equipment, algorithm development |
| Computer-Aided Semen Analysis (CASA) | r = 0.88 | Significantly lower than AI and conventional | Automated, reduces some subjectivity | Lower normal morphology detection |
| Conventional Semen Analysis (CSA) | r = 0.76 | Similar to AI, higher than CASA | Established methodology, widely available | Subjective, requires staining, high variability |
The AI model achieved a test accuracy of 0.93 after 150 epochs when evaluated on 900 batches of previously unseen images [7]. The training utilized a subset of 9,000 images (4,500 normal morphology, 4,500 abnormal morphology) derived from 32 pattern samples. Performance metrics showed precision of 0.95 and recall of 0.91 for detecting abnormal sperm morphology, and precision of 0.91 and recall of 0.95 for normal sperm morphology [7]. Processing efficiency was notable at approximately 139.7 seconds for 25,000 images, averaging 0.0056 seconds per image [7].
Dataset Development and Annotation Protocols
A critical component of AI model development is the creation of high-quality, annotated datasets. Current research introduces novel datasets addressing limitations in existing resources such as HSMA-DS, MHSMA, and SVIA datasets, which suffer from low resolution, limited sample size, and insufficient categories [7]. The annotation protocol involves:
- Manual Annotation: Embryologists and researchers manually annotate well-focused sperm images using bounding boxes in the LabelImg program
- Quality Control: High inter-observer reliability with correlation coefficients of 0.95 for normal sperm morphology detection and 1.0 for abnormal morphology detection
- Categorization Framework: Nine distinct datasets including normal sperm criteria (smooth oval head, length-to-width ratio of 1.5-2, no vacuoles, slender regular neck, uniform tail calibre) and abnormal categories (tapered, amorphous, pyriform, or round heads; observable vacuoles; aberrant neck or tail features)
- Validation Protocol: Normal morphology confirmation requires meeting criteria across all five captured frames [7]
Comparative Analysis: Traditional vs. AI Approaches
Methodological Workflows
The fundamental differences between traditional and AI-based approaches to sperm morphology assessment are evident in their respective workflows. The diagram below illustrates these distinct pathways:
Performance and Efficiency Metrics
When evaluating traditional versus AI-based approaches, multiple performance dimensions must be considered:
Accuracy and Reliability:
- Traditional methods: Highly variable (53%-81% accuracy untrained, 90%-98% trained) depending on classification system complexity and morphologist expertise [5]
- AI methods: Consistently high (93% accuracy) across all morphology categories, unaffected by fatigue or subjective bias [7]
Clinical Utility:
- Traditional stained methods: Render sperm unusable for subsequent procedures due to fixation and staining requirements [7]
- AI unstained methods: Preserve sperm viability for immediate use in assisted reproductive technology, potentially improving outcomes [7]
Efficiency and Throughput:
- Traditional assessment: Requires 4.9-7.0 seconds per image for trained morphologists [5]
- AI assessment: Processes images in 0.0056 seconds each, enabling high-throughput analysis [7]
Standardization Potential:
- Traditional methods: Require continuous quality control, proficiency testing, and re-training to maintain standards [6]
- AI methods: Provide consistent application of criteria once validated, though requiring ongoing algorithm monitoring [7]
Research Reagent Solutions and Essential Materials
The implementation of robust sperm morphology assessment protocols requires specific laboratory materials and reagents. The following table details essential components for both traditional and AI-based approaches:
Table 3: Essential Research Reagents and Materials for Sperm Morphology Assessment
| Item | Function | Application Context | Technical Specifications |
|---|---|---|---|
| Diff-Quik Stain | Sperm staining for morphological visualization | Traditional assessment | Triarylmethane fixative, xanthene & thiazine dyes [6] |
| Confocal Laser Scanning Microscope | High-resolution imaging of unstained live sperm | AI-based assessment | 40× magnification, LSM Z-stack mode, 0.5μm interval [7] |
| Leja Standard Two-Chamber Slides | Sample preparation with standardized depth | Both traditional and AI methods | 20μm depth, ensures consistent preparation [7] |
| Ocular Micrometer | Precise measurement of sperm dimensions | Traditional assessment | Essential for strict WHO criteria application [6] |
| LabelImg Program | Manual annotation of sperm images for AI training | AI development | Creates bounding boxes for supervised learning [7] |
| Hamilton Thorne CASA System | Automated semen analysis for comparison studies | Validation studies | IVOS II with DIMENSIONS II Morphology Software [7] |
Integration Framework and Future Directions
The convergence of traditional expertise and AI technologies presents a promising path forward for sperm morphology assessment. An integrated framework would leverage the strengths of both approaches:
Hybrid Assessment Model:
- AI systems for initial high-throughput screening and classification
- Human morphologists for complex edge cases, quality control, and system validation
- Continuous feedback loops where human expertise refines AI algorithms
Standardization Protocols:
- Implementation of the Sperm Morphology Assessment Standardisation Training Tool for all laboratory personnel
- Regular proficiency testing using AI-validated reference images
- Integration of external quality assurance programs like QuaDeGA and UK NEQAS [5]
Future Research Priorities:
- Multicenter validation studies of AI models across diverse patient populations
- Development of standardized datasets with expert consensus ground truth annotations
- Longitudinal studies comparing live birth outcomes between AI-assisted and traditional assessment
- Exploration of AI explainability to enhance morphologist training and identify novel morphological biomarkers
The integration of validated training tools with emerging AI technologies represents the most promising approach to bridging the standardization gap in sperm morphology assessment. By combining the objectivity and consistency of AI with the nuanced expertise of trained morphologists, the field can achieve new levels of reliability, efficiency, and clinical utility in male fertility assessment.
Validation and Comparative Performance: AI vs. Conventional Methods
The integration of artificial intelligence (AI) into sperm morphology assessment represents a paradigm shift in male fertility evaluation, offering a solution to the long-standing challenges of subjectivity and variability inherent in conventional methods. This whitepaper provides a technical analysis of the performance metrics—including accuracy, precision, and recall—used to validate AI models against traditional semen analysis techniques. By synthesizing findings from recent studies and detailing experimental protocols, we examine the robustness of AI algorithms in classifying sperm morphology and their potential for clinical application. The data indicate that deep learning models can achieve accuracy levels up to 93%, precision of 95%, and recall of 91% for abnormal sperm detection, outperforming both Computer-Aided Semen Analysis (CASA) and conventional semen analysis in correlation strength and reproducibility. However, the trajectory toward full clinical integration necessitates addressing critical gaps in dataset standardization, model interpretability, and multi-center validation. This analysis provides researchers and drug development professionals with a framework for evaluating AI-based sperm morphology tools within the context of assisted reproductive technology innovation.
Male infertility contributes to approximately 50% of infertility cases globally, with sperm morphology analysis representing a crucial diagnostic parameter for predicting fertilization potential [7] [42]. Traditional assessment methods, including conventional semen analysis (CSA) and computer-aided semen analysis (CASA), rely on manual evaluation by trained technicians, a process notoriously prone to subjectivity, inter-observer variability, and limited reproducibility [42] [45]. These limitations have profound implications for assisted reproductive technology (ART) outcomes, as morphology evaluation directly influences sperm selection for procedures such as intracytoplasmic sperm injection (ICSI) [7].
Artificial intelligence, particularly deep learning algorithms, has emerged as a transformative approach to automating and standardizing sperm morphology assessment. By extracting complex features directly from sperm images, AI models minimize human subjectivity and enable high-throughput analysis [19] [4]. However, the validation of these models requires rigorous evaluation using standardized performance metrics—including accuracy, precision, and recall—within robust clinical frameworks [18]. These metrics provide crucial insights into model reliability and clinical applicability, serving as benchmarks for comparison against established methods.
This technical review examines the performance metrics and clinical validation of AI-based sperm morphology assessment in direct comparison to traditional methodologies. We synthesize quantitative evidence from recent studies, detail experimental protocols for model training and validation, and analyze the implications of these findings for infertility treatment and drug development. The integration of AI into reproductive medicine represents not merely an incremental improvement but a fundamental restructuring of diagnostic paradigms, with the potential to significantly enhance ART success rates through data-driven, objective sperm selection.
Performance Metrics in AI-Based Sperm Morphology Assessment
Definition and Significance of Key Metrics
The evaluation of AI models for sperm morphology classification relies on fundamental performance metrics that quantify diagnostic accuracy and operational efficiency. Accuracy represents the proportion of correctly classified spermatozoa (both normal and abnormal) from the total analyzed, providing an overall measure of model performance. Precision indicates the model's ability to correctly identify abnormal sperm without misclassifying normal ones, crucial for minimizing false positives in clinical diagnostics. Recall (or sensitivity) measures the model's capability to detect truly abnormal spermatozoa, directly impacting false negative rates [7] [18]. The F1-score, representing the harmonic mean of precision and recall, offers a balanced metric for model comparison, especially valuable with imbalanced datasets common in sperm morphology where abnormal specimens often outnumber normal ones [18].
Beyond these classification metrics, the area under the receiver operating characteristic curve (AUC-ROC) provides a comprehensive measure of diagnostic ability across all classification thresholds, with values approaching 1.0 indicating excellent model performance [42]. Correlation coefficients (e.g., Pearson's r) quantify the agreement between AI models and established reference methods, offering critical evidence for clinical validity [7]. Processing time per image represents an additional practical metric, determining the feasibility of real-time clinical application, with advanced models now achieving analysis speeds of approximately 0.0056 seconds per image [7].
Comparative Performance: AI vs. Traditional Methods
Quantitative comparisons between AI algorithms, CASA systems, and conventional semen analysis reveal significant differences in performance metrics across studies, reflecting variations in dataset quality, model architecture, and validation protocols.
Table 1: Performance Metrics of AI Models for Sperm Morphology Assessment
| Study/Model | Accuracy | Precision | Recall | AUC-ROC | Correlation with Reference | Sample/Image Size |
|---|---|---|---|---|---|---|
| In-house AI Model (ResNet50) [7] | 0.93 | 0.95 (abnormal), 0.91 (normal) | 0.91 (abnormal), 0.95 (normal) | - | r=0.88 (with CASA), r=0.76 (with CSA) | 21,600 images |
| Deep CNN (SMD/MSS) [18] | 0.55-0.92 | - | - | - | - | 6,035 images (after augmentation) |
| SVM Classifier [4] | - | >0.90 | - | 0.8859 | - | 1,400 sperm cells |
The in-house AI model utilizing ResNet50 transfer learning demonstrated notably strong correlation with both CASA (r=0.88) and conventional semen analysis (r=0.76), outperforming the correlation between CASA and conventional methods (r=0.57) [7]. This suggests that AI models can potentially serve as a unifying standard between existing methodologies. The precision of 0.95 for abnormal sperm detection indicates a low false positive rate, essential for clinical applications where misclassification could impact treatment decisions.
Another study developing a convolutional neural network (CNN) for the SMD/MSS dataset reported a broader accuracy range (55%-92%), highlighting the significant impact of dataset composition and augmentation techniques on model performance [18]. The lower performance boundary primarily occurred in classes with limited training examples, underscoring the challenge of imbalanced morphological categories in real-world samples. Meanwhile, support vector machine (SVM) approaches, representing conventional machine learning, demonstrated strong AUC-ROC values (88.59%) but focused exclusively on sperm head classification without addressing complete sperm structures [4].
Table 2: Comparison of Sperm Morphology Assessment Methods
| Method | Key Strengths | Key Limitations | Inter-Observer Variability | Clinical Integration |
|---|---|---|---|---|
| Conventional Semen Analysis | Established guidelines, low cost | High subjectivity, requires staining | High (CV for morphology: 28.5%) [45] | Widely adopted, reference method |
| Computer-Aided Semen Analysis (CASA) | Partial automation, quantitative metrics | Limited accuracy distinguishing sperm from debris, requires staining | Moderate (reduces but doesn't eliminate human error) | Limited for morphology alone |
| AI-Based Assessment | High accuracy, objectivity, no staining required | Dependency on dataset quality and size | Low (algorithm consistency) | Emerging, requires regulatory approval |
The coefficient of variation (CV) for morphology assessment between operators in conventional semen analysis can reach 28.5%, significantly higher than for concentration (13.9%) and progressive motility (21.8%) [45]. This variability underscores the fundamental limitation that AI approaches aim to address through automated, standardized classification.
Experimental Protocols for AI Model Validation
Dataset Development and Image Processing
The foundation of robust AI model development lies in the creation of comprehensive, well-annotated datasets. Recent studies have employed meticulous protocols for sperm image acquisition and processing:
Sample Preparation and Image Acquisition: In the development of the novel confocal microscopy dataset, semen samples from 30 healthy volunteers were dispensed as 6μL droplets onto standard two-chamber slides with 20μm depth [7]. Images were captured using confocal laser scanning microscopy at 40× magnification in LSM Z-stack mode with a 0.5μm interval, covering a total range of 2μm. This approach generated high-resolution images of 512×512 pixels, capturing 2-3 sperm per image and collecting at least 200 sperm images per sample [7]. Alternatively, the SMD/MSS dataset utilized bright field mode with an oil immersion 100× objective on an MMC CASA system, capturing individual spermatozoa comprising head, midpiece, and tail structures [18].
Annotation and Ground Truth Establishment: Embryologists and researchers manually annotated well-focused sperm images using specialized programs such as LabelImg [7]. The coefficient of correlation between annotators for normal sperm morphology detection reached 0.95, while agreement on abnormal morphology reached 1.0, establishing reliable ground truth labels [7]. For the SMD/MSS dataset, three experts with extensive experience in semen analysis independently classified each spermatozoon according to the modified David classification, which includes 12 classes of morphological defects across head, midpiece, and tail compartments [18]. Statistical analysis using Fisher's exact test assessed inter-expert agreement, with discrepancies resolved through consensus.
Data Augmentation and Preprocessing: To address class imbalance and limited dataset size, augmentation techniques dramatically expanded the SMD/MSS dataset from 1,000 to 6,035 images [18]. Preprocessing steps typically include image denoising to address insufficient lighting or poor staining, normalization through resizing with linear interpolation strategies (e.g., to 80×80×1 grayscale), and data cleaning to handle missing values or inconsistencies [18].
Model Architecture and Training Protocols
Deep learning approaches for sperm morphology classification predominantly utilize convolutional neural network (CNN) architectures:
Model Selection and Training: The in-house AI model employed ResNet50 transfer learning, a deep neural network designed for image classification tasks [7]. The model was trained to minimize the difference between predicted and actual labels, with performance evaluated on a separate test dataset not used during training. Implementation typically occurs in Python environments (e.g., version 3.8) using deep learning frameworks such as TensorFlow or PyTorch [18].
Data Partitioning: Standard protocol involves partitioning the entire image dataset into training and testing subsets through random allocation, typically with 80% of data used for model training and the remaining 20% reserved for testing [18]. From the training subset, an additional portion (e.g., 20%) may be extracted for validation during hyperparameter tuning.
Performance Optimization: Training involves multiple epochs (e.g., 150), with batch processing (e.g., 900 batches of previously unseen images for testing) to evaluate learning progression [7]. The model's processing time is a critical metric, with advanced models achieving analysis speeds of approximately 0.0056 seconds per image, enabling high-throughput semen analysis [7].
The following workflow diagram illustrates the complete experimental protocol for AI model development and validation:
Clinical Validation and Correlation Studies
Validation Against Reference Standards
Robust clinical validation requires demonstrating strong correlation between AI model assessments and established reference methods across diverse patient populations:
Comparison with CASA and Conventional Semen Analysis: A fundamental study comparing an in-house AI model against CASA and conventional semen analysis demonstrated the strongest correlation between AI and CASA (r=0.88), followed by AI and conventional analysis (r=0.76) [7]. The comparatively weaker correlation between CASA and conventional analysis (r=0.57) suggests that AI models may potentially serve as a more consistent reference standard than conventional methods [7]. Both the AI model and conventional semen analysis detected normal sperm morphology at significantly higher rates than CASA, indicating potential systematic differences in how these methodologies define morphological normality.
Inter-Method Agreement Analysis: Beyond correlation coefficients, the agreement distribution between methods provides crucial insights into clinical reliability. Studies evaluating inter-expert agreement for ground truth establishment have documented three agreement scenarios: no agreement (NA) among experts, partial agreement (PA) where 2/3 experts concur on labels, and total agreement (TA) with consensus among all three experts [18]. AI model performance typically excels in morphological categories with higher expert agreement, while struggling with borderline cases that generate disagreement among human experts, reflecting the inherent complexity of sperm morphology classification.
Clinical Workflow Integration
The transition from experimental validation to clinical implementation requires addressing practical considerations:
Live Sperm Analysis without Staining: A significant advancement offered by AI models is the capability to assess unstained live sperm morphology using confocal laser scanning microscopy at low magnification [7]. This preserves sperm viability for subsequent use in ART procedures, addressing a critical limitation of conventional and CASA methods that require staining and high magnification (100×), rendering sperm unusable for further procedures [7]. The clinical implication is profound, enabling selection of high-quality sperm with normal morphology immediately before intracytoplasmic sperm injection, potentially improving fertilization rates and embryo quality.
Processing Efficiency and Throughput: AI models demonstrate remarkable processing speeds, with one study reporting approximately 139.7 seconds for 25,000 images, equating to an average prediction time of about 0.0056 seconds per image [7]. This throughput significantly exceeds manual evaluation capabilities while maintaining consistency unavailable through human assessment. Such efficiency enables comprehensive morphological analysis of larger sperm populations, potentially improving the statistical reliability of morphology assessments for clinical decision-making.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials and Reagents for AI-Based Sperm Morphology Research
| Item | Specification/Function | Application in Research |
|---|---|---|
| Confocal Laser Scanning Microscope | LSM 800, 40× magnification, Z-stack mode | High-resolution image acquisition of unstained live sperm [7] |
| CASA System | IVOS II (Hamilton Thorne) with morphometric tool | Automated sperm imaging and initial morphological measurements [18] |
| Staining Kits | RAL Diagnostics, Diff-Quik (Romanowsky variant) | Sperm staining for conventional and CASA analysis [7] [18] |
| Slide Chambers | LEJA slides (20μm depth), MAKLER chamber | Standardized depth for consistent imaging [7] [46] |
| Annotation Software | LabelImg program | Manual annotation for ground truth establishment [7] |
| Data Augmentation Tools | Python libraries (e.g., TensorFlow, PyTorch) | Dataset expansion for improved model training [18] |
| Quality Control Materials | QC beads, standardized samples | Monitoring analyzer performance and inter-operator consistency [47] |
The integration of these tools enables the development and validation of AI models for sperm morphology assessment. Confocal laser scanning microscopy, in particular, represents a significant advancement over conventional bright-field microscopy for AI applications, providing high-resolution images of unstained live sperm through optical sectioning capabilities [7]. For clinical settings where cost considerations may limit access to advanced microscopy, modified CASA systems with improved optics coupled with data augmentation techniques offer a viable alternative for model development [18].
Quality control materials, including standardized samples and QC beads, remain essential for both traditional and AI-based approaches, ensuring consistent analyzer performance and monitoring inter-operator variability, which can reach a coefficient of variation of 28.5% for morphology assessment in conventional semen analysis [47] [45]. The implementation of rigorous quality control protocols represents a fundamental requirement for any semen analysis laboratory, regardless of methodological approach.
The integration of artificial intelligence into sperm morphology assessment represents a fundamental shift in male fertility evaluation, addressing long-standing limitations of conventional methods through data-driven, objective analysis. Performance metrics from recent studies demonstrate that deep learning models can achieve accuracy levels up to 93%, with strong correlation to established reference methods (r=0.88 with CASA) while enabling analysis of unstained, live sperm—a critical advantage for ART applications [7]. These technical capabilities, combined with processing speeds of approximately 0.0056 seconds per image, position AI-based assessment as a transformative methodology for reproductive medicine [7].
Despite these promising advances, several challenges remain before widespread clinical adoption becomes feasible. The absence of standardized, high-quality annotated datasets continues to hinder model generalizability across diverse populations and clinical settings [18] [4]. The "black-box" nature of complex algorithms presents interpretability challenges in clinical contexts where diagnostic transparency is essential [19]. Furthermore, rigorous multi-center validation trials are necessary to establish universal performance benchmarks and obtain regulatory approvals for clinical use [42].
The trajectory of AI in sperm morphology assessment points toward increasingly sophisticated models capable of analyzing multiple sperm organelles and integrating morphological data with molecular markers of sperm quality. Future research should prioritize the development of standardized public datasets, explainable AI approaches for clinical interpretability, and randomized controlled trials demonstrating improved ART outcomes. As these advancements mature, AI-powered sperm analysis promises to deliver more precise, personalized fertility treatments, ultimately improving success rates for couples facing infertility challenges globally.
The diagnostic evaluation of male infertility has long relied on conventional semen analysis, which serves as a cornerstone for clinical decision-making in assisted reproductive technology (ART). Within this diagnostic paradigm, sperm morphology assessment—the evaluation of sperm size, shape, and structural integrity—represents a critical prognostic factor for fertilization success [48]. However, traditional manual morphology assessment suffers from significant inter-observer variability and subjectivity, leading to inconsistent clinical interpretations and treatment pathways [49] [5].
The emergence of artificial intelligence (AI) technologies, particularly deep learning and computer vision algorithms, promises to revolutionize this domain through automated, quantitative, and objective sperm analysis [50] [19]. This whitepaper provides a comprehensive technical comparison between AI-driven and manual sperm morphology assessment methodologies, contextualized within a broader thesis on the evolution of andrological diagnostics. Through systematic analysis of quantitative performance metrics, experimental protocols, and technical implementations, we aim to delineate the precise advantages and limitations of each approach for research and clinical applications.
Background and Clinical Significance
The Role of Sperm Morphology in Male Fertility Assessment
Sperm morphology assessment evaluates structural characteristics of spermatozoa, including head size and shape, midpiece integrity, and tail appearance, providing crucial information about spermatogenesis efficiency and sperm functional competence [48]. According to World Health Organization (WHO) guidelines, the reference value for normal sperm morphology using strict Tygerberg criteria is >4% normal forms, with values below this threshold associated with decreased fertilization potential in natural conception and some ART procedures [48].
Traditional manual assessment involves microscopic evaluation of stained sperm smears by trained embryologists or technicians, who classify sperm based on standardized morphological criteria [48]. This process is labor-intensive, time-consuming, and inherently subjective, with diagnostic consistency compromised by human factors including visual acuity, decision threshold variations, and classification expertise [5].
Evolution of Automated Assessment Systems
Computer-Aided Sperm Analysis (CASA) systems represented the initial transition toward automated assessment, utilizing basic image processing algorithms for sperm quantification and motility tracking [19] [49]. While offering improvements in standardization for concentration and motility parameters, conventional CASA systems demonstrated limited reliability for morphological classification due to difficulties in accurately distinguishing subtle structural defects and artifacts [49].
The integration of artificial intelligence, particularly deep convolutional neural networks (CNNs), has enabled substantial advances in automated morphology assessment through enhanced feature extraction, pattern recognition, and classification capabilities [7] [19]. Modern AI systems can now evaluate complex morphological features with human-comparable or superior accuracy while providing unprecedented throughput and consistency [7] [51].
Quantitative Performance Comparison
Analytical Accuracy and Correlation with Standard Methods
Recent studies provide direct quantitative comparisons between AI-based and manual sperm morphology assessment, demonstrating significant performance advantages for AI methodologies across multiple metrics.
Table 1: Comparison of Assessment Accuracy Between Methods
| Assessment Method | Correlation with Reference | Classification Accuracy | Processing Speed | Study Reference |
|---|---|---|---|---|
| In-house AI Model (Unstained) | r=0.88 with CASAr=0.76 with conventional analysis | 93% overall accuracyPrecision: 0.95 (abnormal), 0.91 (normal)Recall: 0.91 (abnormal), 0.95 (normal) | ~0.0056 seconds per image139.7 seconds for 25,000 images | [7] |
| Conventional Manual Assessment | r=0.57 with CASA | Variable (53-81% without training)94.9% with training (2-category) | 5-10 seconds per image (manual classification) | [7] [5] |
| Mojo AISA System | High correlation with manual (p<0.01) | Comparable to expert embryologists | 50% reduction in time vs. manual | [51] |
| CASA Systems | Variable (r=0.57-0.88 with other methods) | Underestimates normal morphology vs. manual/AI | Faster than manual but slower than AI | [7] [49] |
A 2025 experimental study directly comparing assessment methods reported that an in-house AI model demonstrated stronger correlation with computer-aided semen analysis (r=0.88) than conventional semen analysis achieved with CASA (r=0.57) [7]. Both the AI model and conventional analysis detected normal sperm morphology at significantly higher rates than CASA systems, suggesting that AI can achieve the accuracy of expert manual assessment while overcoming the subjectivity limitations of conventional methods [7].
Inter-Method Variability and Training Effects
The variability inherent in manual sperm morphology assessment presents a significant challenge for diagnostic consistency and clinical reproducibility.
Table 2: Impact of Training on Assessment Accuracy
| Classification System | Untrained Accuracy | Trained Accuracy (After Intervention) | Expert-Level Accuracy | Study Reference |
|---|---|---|---|---|
| 2-category (Normal/Abnormal) | 81.0 ± 2.5% | 94.9 ± 0.66% | 98 ± 0.43% | [5] |
| 5-category (Head, Midpiece, Tail defects) | 68 ± 3.59% | 92.9 ± 0.81% | 97 ± 0.58% | [5] |
| 8-category (Specific defect types) | 64 ± 3.5% | 90 ± 0.91% | 96 ± 0.81% | [5] |
| 25-category (Individual defects) | 53 ± 3.69% | 82.7 ± 1.05% | 90 ± 1.38% | [5] |
Research demonstrates that without standardized training, manual morphologists show high variability (coefficient of variation = 0.28) and moderate accuracy (53-81% across classification systems) [5]. However, implementation of a structured training tool utilizing machine learning principles and expert consensus labels significantly improved accuracy (to 82.7-94.9%) and reduced variation [5]. This underscores that while human performance can be enhanced through training, AI systems inherently provide consistent classification without extensive training requirements.
Experimental Protocols and Methodologies
AI Model Development and Validation
A 2025 study developed and validated an in-house AI model for unstained live sperm morphology assessment using the following experimental protocol [7]:
Sample Preparation and Imaging:
- Thirty healthy volunteers (aged 18-40) provided semen samples after 2-7 days of sexual abstinence
- Samples were divided into three aliquots for parallel assessment by AI, CASA, and conventional methods
- For AI assessment, 6μL semen droplets were placed on two-chamber slides (20μm depth)
- Sperm images were captured using confocal laser scanning microscopy at 40× magnification in LSM Z-stack mode (Z-stack interval: 0.5μm, total range: 2μm)
- Each image captured 2-3 sperm per frame at a size of 512×512 pixels (159.7×159.7μm)
Dataset Creation and Annotation:
- Researchers created a novel dataset of 21,600 sperm morphological images
- From these, 12,683 images with well-focused sperm were manually annotated using the LabelImg program
- Embryologists and researchers established annotation consistency (correlation coefficient: 0.95 for normal morphology, 1.0 for abnormal morphology)
- Each sperm image was categorized into nine distinct morphological classes based on WHO criteria
AI Model Training and Validation:
- Implemented ResNet50 transfer learning model for sperm classification
- Training utilized 9,000 images (4,500 normal, 4,500 abnormal) derived from 32 pattern samples
- Model achieved test accuracy of 93% after 150 epochs
- Precision and recall metrics demonstrated balanced performance across normal and abnormal classes
- Processing efficiency reached approximately 0.0056 seconds per image
AI Morphology Assessment Workflow: Experimental design for developing and validating an AI model for unstained live sperm morphology assessment, incorporating comparative analysis with CASA and conventional methods [7].
Traditional Manual Assessment Protocol
Conventional semen analysis for morphology assessment follows standardized WHO protocols [48]:
Sample Processing:
- Semen samples are collected by masturbation after 3-7 days of sexual abstinence
- Samples are allowed to liquefy for 30-60 minutes at 37°C before processing
- Liquefied semen is used to prepare smears on glass slides
Staining and Fixation:
- Air-dried smears are fixed and stained using Romanowsky-type stains (e.g., Diff-Quik)
- Staining differentiates sperm components: light pink acrosome, dark pink post-acrosomal region, white midpiece, and light pink tail
Microscopic Evaluation:
- Stained slides are examined under brightfield microscopy at 100× oil immersion objective
- At least 200 sperm are systematically evaluated and classified
- Classification follows strict Tygerberg criteria:
- Normal sperm: smooth oval head (length: 3.7-4.7μm, width: 2.5-3.2μm), length-to-width ratio 1.3-1.8, well-defined acrosome (40-70% of head area), no neck/midpiece/tail defects, no cytoplasmic droplets
- Abnormal sperm: defects in head (tapering, pyriform, round, amorphous, vacuolated), neck/midpiece (bent, asymmetrical, abnormally thin/thick), tail (broken, coiled, multiple)
Quality Assurance:
- Laboratories should participate in internal and external quality control programs
- Regular comparison between technicians to minimize inter-observer variability
- Adherence to standardized classification criteria across assessments
Technical Implementation of AI Systems
Architecture of AI Models for Sperm Morphology
Modern AI systems for sperm morphology assessment typically employ deep learning architectures, particularly convolutional neural networks (CNNs) optimized for image classification tasks [7] [19]. The ResNet50 architecture, utilized in recent studies, provides sufficient depth for feature extraction while mitigating vanishing gradient problems through residual connections [7].
These systems process raw sperm images through multiple hierarchical layers that automatically learn relevant morphological features without manual feature engineering. Early layers detect basic patterns (edges, contours), while deeper layers identify complex structures (acrosome shape, midpiece integrity, tail abnormalities) [19].
Training Methodologies and Validation
AI models require extensive annotated datasets for supervised learning. Training typically involves:
- Transfer learning using pre-trained networks on natural image datasets (e.g., ImageNet)
- Fine-tuning with sperm-specific morphological image data
- Data augmentation techniques (rotation, flipping, brightness adjustment) to improve generalization
- Cross-validation to prevent overfitting and ensure robustness [7]
Performance validation includes comparison with expert embryologist classifications as ground truth, calculation of standard metrics (accuracy, precision, recall, F1-score, AUC-ROC), and assessment of clinical utility through correlation with fertilization outcomes [7] [42].
AI Sperm Classification Pipeline: Technical workflow for AI-based sperm morphology classification, from image preprocessing through deep learning feature extraction to final diagnostic reporting [7] [19].
Research Reagent Solutions and Essential Materials
Table 3: Essential Research Materials for Sperm Morphology Assessment
| Material/Reagent | Specification | Application and Function | Reference |
|---|---|---|---|
| Confocal Laser Scanning Microscope | LSM 800, 40× magnification | High-resolution imaging of unstained live sperm for AI analysis | [7] |
| Chamber Slides | Leja, 20μm depth | Standardized sample presentation for microscopic evaluation | [7] |
| Romanowsky-type Stains | Diff-Quik stain | Differential staining of sperm components for manual morphology | [7] |
| Phase Contrast Optics | 100× oil immersion objective | Visualization of unstained sperm for motility and basic morphology | [5] |
| CASA System | IVOS II, Hamilton Thorne | Automated sperm concentration and motility analysis | [7] [49] |
| Sperm Morphology Staining Kit | WHO-compliant | Standardized staining for manual morphological assessment | [48] |
| Quality Control Slides | Pre-validated morphology slides | Technician training and inter-laboratory standardization | [5] |
Discussion and Future Directions
Integration of AI in Clinical Andrology
The quantitative evidence demonstrates that AI-based sperm morphology assessment offers significant advantages over manual methods in terms of standardization, throughput, and objectivity [7] [19]. The strong correlation between AI classification and expert manual assessment (r=0.76-0.88), combined with superior processing efficiency (~0.0056 seconds per image), positions AI as a transformative technology for andrology laboratories [7].
A critical advantage of AI systems is their ability to analyze unstained, live sperm, preserving sample viability for subsequent use in ART procedures [7]. This capability enables integration of morphology assessment directly into clinical workflows without compromising sample integrity, particularly valuable for intracytoplasmic sperm injection (ICSI) treatments where individual sperm selection is paramount.
Limitations and Research Gaps
Despite promising performance metrics, several challenges remain for widespread adoption of AI morphology assessment:
Data Diversity and Generalization: Most AI models are trained on limited datasets from specific populations and equipment, potentially limiting generalizability across diverse patient populations and laboratory settings [52]. Multicenter validation studies with diverse demographic representation are needed to ensure robust performance.
Regulatory and Standardization Hurdles: AI systems for clinical diagnosis require regulatory approval (CE marking, FDA clearance) and standardization across platforms [52] [42]. The absence of universally accepted validation protocols and reference standards presents barriers to clinical implementation.
Interpretability and Trust: The "black box" nature of complex deep learning models can hinder clinical adoption, as embryologists may be reluctant to trust classifications without understanding the underlying reasoning [19]. Explainable AI approaches that provide interpretable feature importance could address this limitation.
Future Research Directions
The evolving landscape of AI in sperm morphology assessment suggests several promising research directions:
- Integration of multi-parameter analysis combining morphology with motility, DNA fragmentation, and clinical metadata for comprehensive sperm quality assessment [19] [42]
- Development of real-time AI systems for live sperm selection during ICSI procedures
- Implementation of federated learning approaches to enhance model robustness while maintaining data privacy
- Longitudinal studies correlating AI-derived morphological parameters with clinical outcomes (fertilization, pregnancy, live birth rates)
- Standardization of imaging protocols and annotation criteria to facilitate multi-center research collaboration
This comprehensive analysis demonstrates that AI-based sperm morphology assessment quantitatively outperforms manual methods in accuracy, consistency, and efficiency while eliminating inter-observer variability. The robust correlation between AI classification and expert manual assessment (r=0.76-0.88), combined with dramatically faster processing speeds (~0.0056 vs. 5-10 seconds per image), establishes AI as a superior methodological approach for high-throughput andrology applications [7].
The preservation of sample viability through unstained analysis represents a significant advantage for clinical ART workflows, particularly for ICSI procedures [7]. However, the implementation of AI systems requires careful attention to validation protocols, regulatory compliance, and integration with existing laboratory practices.
As the field advances, the convergence of AI with other emerging technologies (robotics, genomics, multi-omics) promises to further transform male infertility diagnosis and treatment. Through continued refinement and validation, AI-driven morphology assessment is poised to become the new standard for objective, quantitative, and clinically predictive sperm evaluation in both research and clinical settings.
Sperm morphology assessment is a cornerstone of male fertility evaluation, providing critical diagnostic and prognostic information. For decades, this analysis has relied on conventional semen analysis (CSA) methods requiring manual microscopic examination of stained sperm slides by trained technicians—a process plagued by subjectivity, inter-laboratory variability, and time-intensive protocols [53] [5]. The emerging integration of artificial intelligence (AI) models into clinical workflows represents a paradigm shift toward automated, objective, and standardized assessment. This technical analysis examines the transformative impact of workflow integration for both traditional and AI-based sperm morphology assessment, with particular focus on efficiency gains and standardization achievements within the context of andrology laboratory operations.
The limitations of conventional morphology assessment are well-documented in scientific literature. Traditional methods require sperm to be fixed and stained before analysis, rendering them unusable for subsequent assisted reproductive technologies [7]. Furthermore, studies demonstrate significant variability among technicians, with one investigation reporting mean morphology results ranging from 7.3% to 15% normal forms when different laboratorians analyzed the same slides [53]. This high degree of subjectivity necessitates rigorous standardization protocols and continuous quality control measures, which remain challenging to implement consistently across facilities [5].
AI-based systems offer a fundamentally different approach, leveraging deep learning models trained on extensive image datasets to provide consistent, quantitative morphology assessment. Recent research demonstrates that in-house AI models can achieve test accuracy of 0.93 with precision of 0.95 and recall of 0.91 for detecting abnormal sperm morphology [7]. Crucially, these systems can assess unstained, live sperm at low magnification (40×), preserving sperm viability for subsequent clinical use while maintaining analytical precision [7]. This capability represents a significant advancement over traditional methods that require 100× magnification and staining procedures [7].
Methodology and Experimental Protocols
Traditional Sperm Morphology Assessment Protocol
The conventional workflow for sperm morphology assessment follows standardized protocols based on World Health Organization (WHO) guidelines. The process begins with semen sample collection and liquefaction, followed by preparation of smears on glass slides [53]. Critical to this methodology is the staining process, typically using Romanowsky-type stains such as Diff-Quik, which allows for clear visualization of sperm structures [7]. Stained slides are then examined under oil immersion at 100× magnification, with technicians evaluating at least 200 spermatozoa per sample across multiple microscopic fields [7].
The assessment criteria for traditional morphology focus on specific structural characteristics. Normal sperm are identified by a smooth oval head with length-to-width ratio of 1.5–2, no vacuoles, a slender regular neck, and a uniform tail without cytoplasmic droplets exceeding one-third of the head size [7]. Abnormalities are categorized by location (head, midpiece, tail) with classification systems ranging from simple 2-category (normal/abnormal) to complex 25-category systems that specify individual defect types [5]. Studies indicate that technician accuracy decreases significantly as classification systems become more complex, with untrained users achieving only 53% accuracy with 25-category systems compared to 81% with simple 2-category classification [5].
Table 1: Traditional Morphology Assessment Method Details
| Protocol Aspect | Specification | Impact on Standardization |
|---|---|---|
| Staining Method | Diff-Quik (Romanowsky variant) | Potential for staining-induced morphological alterations [53] |
| Magnification | 100× oil immersion | Standardized across laboratories but requires high expertise |
| Sperm Counted | Minimum 200 per sample | Follows WHO guidelines but time-intensive |
| Classification System | 2 to 25 categories | Accuracy decreases with system complexity (81% to 53%) [5] |
| Technician Training | Variable; requires experienced morphologists | High inter-technician variability (CV=0.28) without standardized training [5] |
AI-Based Morphology Assessment Protocol
The development and implementation of AI models for sperm morphology assessment follows a structured computational workflow. Recent research utilized confocal laser scanning microscopy at 40× magnification in confocal mode (LSM, Z-stack) to capture high-resolution images of unstained live sperm [7]. The Z-stack interval was set at 0.5 μm covering a total range of 2 μm, producing images of 512×512 pixels with a size of 159.7×159.7 μm per slide [7]. This imaging protocol generated at least 200 sperm images per sample, with each capture containing 2-3 sperm.
The annotation process involved manual labeling by embryologists and researchers using the LabelImg program, with a high coefficient of correlation between annotators (0.95 for normal sperm morphology detection and 1.0 for abnormal morphology detection) [7]. The resulting dataset contained 21,600 images with 12,683 annotated as unstained sperm [7]. For model development, researchers selected a ResNet50 transfer learning model, a deep neural network designed for image classification tasks [7]. The model was trained on a subset of 9,000 images (4,500 normal and 4,500 abnormal sperm morphology) and tested on 900 batches of previously unseen images [7].
The AI model achieved a test accuracy of 0.93 after 150 epochs, with precision of 0.95 and recall of 0.91 for detecting abnormal sperm morphology, and precision of 0.91 and recall of 0.95 for normal sperm morphology [7]. Processing time was approximately 139.7 seconds for 25,000 images, equating to an average prediction time of about 0.0056 seconds per image [7]. This represents a significant efficiency improvement over traditional manual assessment.
Diagram 1: AI Model Development and Deployment Workflow
Quantitative Comparison of Assessment Methods
Performance Metrics and Correlation Analysis
Comparative studies between assessment methodologies reveal significant differences in performance characteristics. Recent research demonstrates that in-house AI models show the strongest correlation with computer-aided semen analysis (CASA) at r=0.88, followed by conventional semen analysis at r=0.76 [7]. The correlation between CASA and conventional semen analysis was notably weaker at r=0.57 [7]. Both the in-house AI and conventional semen analysis methods detected normal sperm morphology at significantly higher rates than CASA, suggesting potential methodological differences in classification criteria [7].
The integration of standardized training tools significantly improves performance for both human morphologists and AI systems. Research utilizing a Sperm Morphology Assessment Standardisation Training Tool demonstrated that novice morphologists achieved initial accuracy of 81.0±2.5% with 2-category classification systems, which improved to 94.9±0.66% after training with visual aids and video instruction [5]. With repeated training over four weeks, final accuracy rates reached 98±0.43% for 2-category systems and 90±1.38% for complex 25-category systems [5]. Diagnostic speed also improved significantly from 7.0±0.4 seconds to 4.9±0.3 seconds per image classification [5].
Table 2: Performance Comparison of Assessment Methods
| Performance Metric | Traditional CSA | Computer-Aided (CASA) | AI-Based Assessment |
|---|---|---|---|
| Correlation with AI | r=0.76 [7] | r=0.88 [7] | Self-benchmark |
| Correlation with CSA | Self-benchmark | r=0.57 [7] | r=0.76 [7] |
| Normal Morphology Detection Rate | Significantly higher than CASA [7] | Lower than CSA and AI [7] | Significantly higher than CASA [7] |
| Assessment Speed | ~7.0s/image (novice) [5] | Variable | ~0.0056s/image [7] |
| Training Improvement | 82% to 90% accuracy with training [5] | Not specified | 93% test accuracy [7] |
| Multi-Category Accuracy (25 categories) | 53% (untrained) to 90% (trained) [5] | Not specified | 91% precision for abnormal detection [7] |
Impact on Clinical Workflow Efficiency
The integration of AI models into clinical workflows generates substantial efficiency improvements across multiple parameters. The automated nature of AI assessment eliminates the time-intensive manual examination process, reducing the analytical time requirement from seconds per sperm to milliseconds per image [7] [5]. Furthermore, AI systems can operate continuously without fatigue, enabling high-throughput analysis that significantly expands laboratory capacity.
The pre-analytical phase also benefits from AI integration through the elimination of staining procedures. By utilizing unstained live sperm, laboratories reduce material costs associated with staining reagents and eliminate the 30-60 minute staining protocol from the workflow [7]. This modification also preserves sperm viability for subsequent therapeutic use in assisted reproductive technologies, creating a seamless transition from diagnostic assessment to clinical application [7].
Standardization Achievements Through Workflow Integration
Reduction in Technical Variability
The implementation of AI systems directly addresses the critical challenge of inter-technician variability that has traditionally plagued sperm morphology assessment. Studies demonstrate that untrained users exhibit high variation in morphological classification (CV=0.28) with accuracy scores ranging from 19% to 77% when using the same samples [5]. This variability persists despite adherence to WHO standardized methodologies, highlighting the inherent subjectivity of human-based assessment.
AI models provide consistent classification criteria across all analyses, effectively eliminating the interpersonal variation that compromises result reliability in multi-technician laboratories. The ResNet50 transfer learning model demonstrated precision of 0.95 and recall of 0.91 for detecting abnormal sperm morphology across all test samples, performance metrics that remain stable regardless of sample volume or processing duration [7]. This consistency establishes a new standard for reproducibility in sperm morphology assessment, particularly valuable for longitudinal studies and multi-center clinical trials requiring standardized outcome measures.
Quality Control and Training Standardization
AI integration facilitates enhanced quality control protocols through the creation of standardized classification benchmarks. The development of training tools based on machine learning principles, utilizing expert consensus labels ("ground truth"), has demonstrated significant improvements in morphologist accuracy [5]. These tools function similarly to the supervised learning approaches used in AI training, providing consistent reference standards that can be deployed across multiple laboratory sites.
The application of a Sperm Morphology Assessment Standardisation Training Tool demonstrated that structured training protocols could reduce variation and improve accuracy across all classification system complexities [5]. This approach addresses the observed phenomenon that accuracy decreases as classification systems become more complex, with the 25-category system showing the lowest initial accuracy (53±3.69%) but still achieving substantial improvement after training (90±1.38%) [5]. Such tools provide a mechanism for continuous quality improvement that complements AI systems in mixed workflow environments.
Diagram 2: Standardization Challenges and Solutions in Morphology Assessment
Implementation Considerations and Research Reagent Solutions
Essential Research Materials and Reagents
Successful integration of AI-based morphology assessment requires specific research reagents and laboratory materials that differ substantially from traditional methodology. The following table details essential components for implementing AI-driven sperm morphology assessment protocols.
Table 3: Research Reagent Solutions for AI-Based Sperm Morphology Assessment
| Reagent/Material | Specification | Function in Workflow |
|---|---|---|
| Imaging Chamber | Standard two-chamber slide with 20μm depth (Leja) | Standardized sample presentation for imaging [7] |
| Microscopy System | Confocal laser scanning microscope (e.g., LSM 800) | High-resolution image acquisition without staining [7] |
| Annotation Software | LabelImg program | Manual annotation for training dataset creation [7] |
| AI Development Framework | ResNet50 transfer learning model | Deep neural network for image classification [7] |
| Synthetic Data Tool | AndroGen open-source software | Generating customized synthetic images for model training [54] |
| Analysis Software | FlowJo, Cytobank | Multiparametric data analysis and dimensional reduction [55] |
| Training Tool | Sperm Morphology Assessment Standardisation Training Tool | Standardizing morphologist training using machine learning principles [5] |
Integration Pathways for Clinical Laboratories
The transition from traditional to AI-enhanced workflows requires strategic implementation planning. Laboratories can pursue multiple integration pathways depending on existing infrastructure and clinical volume. One approach involves parallel operation of both traditional and AI systems during a validation period, allowing for comparative analysis and staff training. Alternatively, laboratories may opt for a phased implementation, beginning with AI assessment for specific indications such as intracytoplasmic sperm injection (ICSI) cases before expanding to comprehensive diagnostic services.
Critical to successful integration is the establishment of validation protocols that verify AI system performance against established laboratory standards. This process should include correlation studies between AI results and manual morphology assessment across a representative sample range, with particular attention to borderline cases and uncommon morphological variants. Ongoing quality assurance must include regular review of false positive and false negative classifications to identify potential algorithmic biases or image acquisition artifacts.
The integration of AI-based systems into sperm morphology assessment workflows represents a significant advancement in andrology laboratory practice, addressing longstanding challenges in both efficiency and standardization. Quantitative evidence demonstrates that AI models can achieve correlation coefficients of 0.88 with computer-assisted systems while maintaining accuracy rates of 93% with processing speeds of 0.0056 seconds per image [7]. These performance characteristics enable laboratories to expand testing capacity while reducing technical variability associated with human assessment.
The standardization benefits extend beyond analytical consistency to encompass training and quality control processes. Research shows that standardized training tools based on machine learning principles can improve morphologist accuracy from 53% to 90% even with complex 25-category classification systems [5]. When combined with AI systems that provide consistent classification criteria regardless of operator experience or workload, laboratories can achieve unprecedented levels of reproducibility in sperm morphology assessment.
Future developments in AI-based morphology assessment will likely focus on multidimensional analysis incorporating additional sperm parameters such as DNA fragmentation, mitochondrial function, and molecular markers [55] [50]. The integration of radiomics approaches that extract quantitative features from medical images using data characterization algorithms may further enhance predictive value for clinical outcomes [50]. As these technologies mature, the workflow integration of comprehensive AI-based sperm assessment systems will increasingly become the standard of care in advanced andrology laboratories, ultimately improving diagnostic accuracy and therapeutic outcomes in male infertility treatment.
Sperm morphology analysis, the microscopic evaluation of sperm size, shape, and structural integrity, has long been a cornerstone of male fertility assessment. Traditional manual assessment, performed by trained technicians according to World Health Organization (WHO) guidelines, classifies sperm based on strict criteria for head, neck, midpiece, and tail abnormalities [12]. However, this method suffers from significant limitations, including high inter-observer variability (with studies reporting up to 40% disagreement between expert evaluators), lengthy evaluation times (30-45 minutes per sample), and inconsistent standards across laboratories [8] [56]. These limitations have prompted the development of artificial intelligence (AI)-based approaches that leverage deep learning and computer vision to automate sperm classification with greater speed, objectivity, and consistency [4] [8].
The clinical context for this technological transition is evolving. Recent guidelines, such as those from the French BLEFCO Group, have questioned the prognostic value of traditional morphology assessment for predicting assisted reproductive technology (ART) success, instead recommending its simplified use primarily for detecting specific monomorphic abnormalities like globozoospermia [3]. Concurrently, the advent of intracytoplasmic sperm injection (ICSI) has reduced emphasis on conventional semen parameters, as the technique requires only few sperm and bypasses many natural selection barriers [56]. This whitepaper examines the trends, barriers, and future considerations shaping the adoption of AI-based sperm morphology analysis within this complex clinical and research landscape.
Current Adoption Trends in AI-Based Sperm Analysis
The adoption of AI-based sperm morphology analysis is progressing along two parallel tracks: research validation and initial clinical implementation. In research settings, deep learning models have demonstrated exceptional performance, with recent studies reporting classification accuracies exceeding 96% for distinguishing normal from abnormal sperm forms [8]. These systems can reduce analysis time from 30-45 minutes to under one minute per sample, representing a significant efficiency improvement [8]. The research focus has shifted from conventional machine learning approaches, which relied on handcrafted features, to deep learning architectures that automatically learn discriminative features from raw image data [4].
Table 1: Performance Comparison of Sperm Morphology Assessment Methods
| Method | Accuracy Range | Processing Time | Key Advantages | Major Limitations |
|---|---|---|---|---|
| Traditional Manual Assessment | N/A (High variability) | 30-45 minutes | Low initial equipment cost; Well-established in guidelines | Subjective (up to 40% inter-observer variability); Labor-intensive |
| Conventional Machine Learning | 49%-90% [4] | 5-10 minutes | Reduced subjectivity compared to manual; Automated feature extraction | Limited to pre-defined features; Lower accuracy for complex abnormalities |
| Deep Learning Approaches | 87%-96.77% [8] | <1 minute [8] | High accuracy; Minimal human intervention; Continuous learning potential | High computational requirements; Extensive training data needed |
In clinical environments, adoption remains cautious but growing. AI systems are increasingly integrated with computer-assisted sperm analysis (CASA) platforms, enhancing their morphology assessment capabilities beyond traditional motility and concentration parameters [57] [58]. Emerging point-of-care applications, such as portable AI-driven microscopes for veterinary use, demonstrate the potential for decentralized testing in resource-limited settings [58]. However, comprehensive clinical adoption in human fertility clinics remains limited by regulatory, validation, and standardization barriers.
Technical and Economic Barriers to Adoption
Data Quality and Availability Constraints
A fundamental barrier to robust AI system development is the lack of standardized, high-quality annotated datasets [4]. Medical institutions historically have not systematically archived sperm morphology images, resulting in limited data availability [4]. When images are available, they often suffer from quality issues such as sperm overlapping, partial structure visibility, or staining inconsistencies [4]. Annotation complexity presents another significant challenge, as each sperm requires simultaneous evaluation of head, vacuoles, midpiece, and tail abnormalities according to strict criteria [38] [4]. Creating datasets with sufficient size, diversity, and annotation consistency for training generalized models remains labor-intensive and requires rare expertise in both embryology and data annotation.
Development and Implementation Costs
The economic considerations for AI system adoption involve substantial upfront investment versus potential long-term efficiencies. Development costs include not only the AI algorithm creation but also the integration with high-quality microscopy imaging systems. For instance, research-grade systems utilize differential interference contrast (DIC) optics with high numerical apertures and high-resolution cameras [38], representing an investment of tens to hundreds of thousands of dollars. However, emerging solutions are addressing cost barriers through innovative approaches. Recent research has demonstrated the feasibility of low-cost, portable AI-driven microscopes that integrate custom-built microscopes with Raspberry Pi platforms and microfluidic chips, offering a more accessible alternative to traditional CASA systems [58].
Table 2: Cost-Benefit Analysis of AI-Based Sperm Analysis Systems
| Cost Component | Research-Grade System | Portable AI System [58] | Traditional CASA |
|---|---|---|---|
| Microscope Hardware | High-end DIC microscope ($50,000-$150,000) | Custom-built inverted microscope | Research-grade microscope |
| Imaging Sensor | High-resolution CMOS camera (~$10,000) | Raspberry Pi camera module | Integrated camera system |
| Computing Platform | High-performance GPU workstations (~$10,000) | Raspberry Pi 4 | Dedicated computer |
| Per-Sample Cost | Low | Very low | Moderate |
| Throughput | High | Moderate | High |
Validation and Regulatory Hurdles
The transition from research validation to clinical implementation requires rigorous demonstration of analytical validity and clinical utility. Currently, AI systems for sperm morphology analysis exist in a regulatory gray area, with no clearly defined approval pathways from agencies like the FDA or EMA. Analytical validation must demonstrate that the AI system can accurately and reliably identify sperm abnormalities across diverse patient populations and laboratory conditions [4] [8]. Clinical validation requires proof that AI-derived morphology parameters meaningfully predict clinical outcomes such as fertilization rates, pregnancy, or live birth [12] [56]. The French BLEFCO guidelines' skepticism about the clinical relevance of traditional morphology assessment raises questions about what endpoints should be used for validating AI systems [3].
Standardization and Training Requirements
Protocol Standardization Challenges
The lack of standardized protocols for sample preparation, imaging, and analysis contributes to significant inter-laboratory variability in morphology assessment [59] [12]. Variations in staining methods (Diff-Quik, Papanicolaou, etc.), microscope optics (brightfield, phase contrast, DIC), and magnification (1000x oil immersion recommended) introduce pre-analytical variables that challenge AI model generalization [59]. The Australian standardization program (UQSMSP) demonstrates the value of centralized protocols, including specific equipment requirements, standardized counting methodologies, and regular proficiency testing [59]. AI systems must demonstrate robustness across these methodological variations to achieve widespread adoption.
Personnel Training and Competency Assessment
Traditional morphology assessment requires extensive technician training, typically involving months of supervised practice and ongoing quality control [38]. A novel approach to addressing training challenges is the development of standardized sperm morphology assessment tools that provide instant feedback on classification accuracy [38]. These systems use expert-validated "ground truth" images to train and assess technician competency, potentially reducing the time required to achieve proficiency. For AI systems, the training requirement shifts from morphological classification expertise to system operation, quality control, and results interpretation. This transition may eventually reduce dependency on rare technical expertise, but initially requires dual competency in both traditional morphology and AI system management.
Experimental Protocols for AI-Based Morphology Analysis
Protocol 1: Deep Learning Model Development for Sperm Classification
This protocol outlines the methodology for developing a deep learning model for sperm morphology classification, based on recent research [8].
Materials and Reagents:
- Annotated sperm image dataset (SMIDS or HuSHeM)
- Python 3.8+ with TensorFlow 2.4+ or PyTorch 1.8+
- High-performance computing environment with GPU (NVIDIA RTX 3080+ recommended)
- Data augmentation libraries (Albumentations, Imgaug)
Procedure:
- Data Preprocessing: Resize all images to uniform dimensions (typically 224×224 or 299×299 pixels). Apply normalization using ImageNet statistics (mean=[0.485, 0.456, 0.406], std=[0.229, 0.224, 0.225]).
- Data Augmentation: Implement real-time augmentation during training including random rotation (±15°), horizontal/vertical flipping, brightness/contrast adjustment (±10%), and Gaussian noise injection.
- Model Architecture: Implement a hybrid architecture combining ResNet50 backbone with Convolutional Block Attention Module (CBAM). Replace final fully connected layer with task-specific classification head.
- Transfer Learning: Initialize model with pre-trained weights (ImageNet). Fine-tune all layers rather than only the classifier to adapt features to sperm morphology domain.
- Training Configuration: Use Adam optimizer with initial learning rate 1e-4, reduced by factor 10 when validation loss plateaus. Train for 100 epochs with batch size 32.
- Deep Feature Engineering: Extract features from multiple network layers (CBAM, Global Average Pooling, Global Max Pooling). Apply feature selection methods (PCA, Random Forest importance, variance thresholding). Train SVM with RBF kernel on selected features.
- Validation: Perform 5-fold cross-validation, reporting mean accuracy and standard deviation across folds. Use McNemar's test for statistical significance.
Protocol 2: Development of a Low-Cost AI-Driven Microscope
This protocol details the methodology for creating a portable, affordable AI-based sperm analysis system, adapted from recent research [58].
Materials and Reagents:
- Raspberry Pi 4 Model B (4GB RAM)
- Raspberry Pi High Quality Camera module
- Custom-built inverted microscope frame
- 3D-printed microfluidic chip fabrication supplies
- AI models (EfficientDet-Lite or custom CNN)
- Bull semen samples preserved in buffered formal saline
Procedure:
- Microscope Assembly: Construct inverted microscope using LED illumination, 40x magnification objective lens, and mechanical stage. Integrate Raspberry Pi camera module for image capture.
- Microfluidic Chip Fabrication: Design microfluidic channel dimensions (15,000 μm width × 10,700 μm length × 20 μm height). Fabricate using UV photolithography with AZ P4620 photoresist on glass substrate.
- Sample Preparation: Load approximately 10 μL semen sample into microfluidic chip. Ensure even distribution without bubbles for consistent imaging.
- Image Acquisition: Capture multiple fields of view across the microfluidic channel. Maintain consistent lighting conditions and focus throughout acquisition.
- AI Model Deployment: Convert trained model to TensorFlow Lite format for edge deployment. Optimize model weights through quantization to reduce computational requirements.
- Inference Pipeline: Implement sperm detection using trained model. Classify individual sperm for motility parameters (progressive, non-progressive, immotile) and concentration.
- Validation: Compare results with conventional CASA system using correlation analysis and Bland-Altman plots across key parameters (motility, concentration).
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents and Materials for AI-Based Sperm Morphology Analysis
| Item | Specification | Research Application |
|---|---|---|
| Microscope System | DIC optics with 1000x magnification, oil immersion [59] | High-resolution imaging for ground truth annotation |
| Staining Reagents | Diff-Quik, Papanicolaou, or SpermBlue stains | Sample preparation for morphological assessment |
| Annotation Software | Web-based annotation tools with expert consensus [38] | Creation of validated training datasets |
| Deep Learning Framework | TensorFlow, PyTorch, or Keras | Model development and training |
| Data Augmentation Library | Albumentations or Imgaug | Dataset expansion and model regularization |
| Microfluidic Chips | PDMS-based with 20μm channel height [58] | Sample preparation standardization for portable systems |
| Edge Computing Device | Raspberry Pi 4 or NVIDIA Jetson Nano | Deployment of portable AI analysis systems |
Visualization of AI-Based Sperm Analysis Workflow
The following diagram illustrates the complete workflow for AI-based sperm morphology analysis, from sample preparation to clinical reporting:
AI-Based Sperm Morphology Analysis Workflow
The adoption of AI-based sperm morphology analysis represents a paradigm shift in male fertility assessment, offering solutions to long-standing challenges of subjectivity, variability, and inefficiency in traditional methods. Current evidence demonstrates that deep learning approaches can achieve expert-level classification accuracy while reducing analysis time from 45 minutes to under one minute [8]. However, significant barriers remain, including data standardization, regulatory approval, clinical validation, and implementation costs.
Future development should focus on several key areas: (1) creating large, diverse, and publicly available datasets with expert-validated annotations; (2) demonstrating clinical utility through prospective studies linking AI-derived morphology parameters to reproductive outcomes; (3) developing standardized protocols and reference materials for quality assurance; and (4) creating cost-effective, accessible systems suitable for resource-limited settings [4] [58] [59].
The research community is actively addressing these challenges, with recent advancements in attention mechanisms, feature engineering, and edge computing showing particular promise [8]. As these technologies mature and validation evidence accumulates, AI-based sperm morphology analysis is poised to transition from research curiosity to clinical standard, ultimately enhancing diagnostic accuracy, treatment personalization, and patient outcomes in reproductive medicine.
Conclusion
The integration of AI into sperm morphology assessment represents a transformative advancement, offering a solution to the long-standing challenges of subjectivity and variability inherent in traditional methods. Research demonstrates that AI models, particularly deep learning architectures enhanced with attention mechanisms, can achieve diagnostic accuracy exceeding 96%, significantly outperforming manual analysis. The clinical adoption of these technologies is steadily growing, with over 50% of fertility specialists now reporting AI usage. Future directions must focus on developing large, standardized, multi-center datasets, improving model interpretability, and conducting robust clinical trials to validate AI's impact on live birth rates. For the biomedical research community, the priority lies in creating reproducible, transparent algorithms that can be seamlessly integrated into existing diagnostic workflows, ultimately paving the way for personalized, data-driven fertility treatments and enhanced drug development processes in reproductive medicine.
References
- 1. WHO laboratory manual for the examination and ... [https://www.who.int/publications/i/item/9789240030787]
- 2. Morphological Parameters of 29994 sperm in a fertile male ... [https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2025.1546290/full]
- 3. An Expert Review From the French BLEFCO Group [https://pubmed.ncbi.nlm.nih.gov/41185394/]
- 4. Deep learning-based approach for sperm morphology analysis [https://bmcurol.biomedcentral.com/articles/10.1186/s12894-025-01946-w]
- 5. Use of a sperm morphology assessment standardisation ... [https://www.nature.com/articles/s41598-025-07515-3]
- 6. Sperm Morphology Assessment in the Era of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9253798/]
- 7. Artificial intelligence model for the assessment of unstained ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12060770/]
- 8. Deep feature engineering for accurate sperm morphology ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0330914]
- 9. Interobserver Variability in Semen Analysis - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC10620459/]
- 10. An assessment tool for computer-assisted semen analysis ... [https://www.nature.com/articles/s41598-022-20943-9]
- 11. OpenCASA: A new open-source and scalable tool for sperm ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6355034/]
- 12. Sperm morphology: Evaluating its clinical relevance in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11194684/]
- 13. Sperm morphology value in assisted reproduction [https://pmc.ncbi.nlm.nih.gov/articles/PMC11624537/]
- 14. Use of a sperm morphology assessment standardisation ... [https://pubmed.ncbi.nlm.nih.gov/40595188/]
- 15. On the Origins of the Semen Analysis - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC5799927/]
- 16. Evolution of the WHO “Semen” processing manual from ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8842884/]
- 17. Semen analysis [https://en.wikipedia.org/wiki/Semen_analysis]
- 18. Deep-learning based model for sperm morphology ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12599357/]
- 19. Revolutionizing Sperm Analysis with AI: A Review of ... [https://www.mdpi.com/2079-3197/13/6/132]
- 20. Deep learning-based approach for sperm morphology analysis [https://pmc.ncbi.nlm.nih.gov/articles/PMC12538767/]
- 21. Deep learning for the classification of human sperm [https://www.sciencedirect.com/science/article/abs/pii/S0010482519302112]
- 22. Artificial intelligence based classification and prediction of ... [https://www.nature.com/articles/s41598-025-93718-7]
- 23. Convolutional Neural Networks in 2025: Architecture & ... [https://vinova.sg/convolutional-neural-networks-architecture-applications/]
- 24. GM-CBAM-ResNet: A Lightweight Deep Learning Network ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11942712/]
- 25. Tea disease identification based on ECA attention ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1489655/full]
- 26. Dense skip-attention for convolutional networks [https://www.nature.com/articles/s41598-025-09346-8]
- 27. Deep-learning based model for sperm morphology ... [https://pubmed.ncbi.nlm.nih.gov/41203404/]
- 28. The Differences between Manual and AI Semen Analysis [https://www.andrologycenter.in/blog/the-differences-between-manual-and-ai-semen-analysis/]
- 29. HKUMed develops world-first AI-powered fertilization ... [https://www.eurekalert.org/news-releases/1096651]
- 30. Multidimensional morphological analysis of live sperm ... [https://www.sciencedirect.com/science/article/pii/S2001037024000497]
- 31. Validation of AI-based semen analysis by urologists in training [https://pmc.ncbi.nlm.nih.gov/articles/PMC12634335/]
- 32. Artificial intelligence (AI) approaches to male infertility in IVF [https://pmc.ncbi.nlm.nih.gov/articles/PMC11971770/]
- 33. Artificial intelligence in assisted reproductive technology [https://www.sciencedirect.com/science/article/pii/S1472648325000628]
- 34. Current Updates on Involvement of Artificial Intelligence ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10890589/]
- 35. Artificial intelligence for sperm selection—a systematic review [https://www.sciencedirect.com/science/article/pii/S0015028223005332]
- 36. Unveiling the capabilities of vision transformers in sperm ... morphology [https://peerj.com/articles/cs-3173/]
- 37. Gold-standard for computer-assisted morphological sperm ... [https://www.sciencedirect.com/science/article/abs/pii/S0010482517300525]
- 38. Development of a sperm morphology assessment ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12036963/]
- 39. Deep feature engineering for accurate sperm morphology ... [https://pubmed.ncbi.nlm.nih.gov/40929291/]
- 40. Design and Implementation of a Deep Learning System to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12567593/]
- 41. Improving Infertility Outcomes Through Artificial Intelligence [https://advances.massgeneral.org/obgyn/article.aspx?id=1289]
- 42. Artificial intelligence (AI) approaches to male infertility in IVF: a mapping review | European Journal of Medical Research | Full Text [https://eurjmedres.biomedcentral.com/articles/10.1186/s40001-025-02479-6]
- 43. Advanced Multi-Level Ensemble Learning Approaches for Comprehensive Sperm Morphology Assessment [https://www.mdpi.com/2075-4418/15/12/1564]
- 44. Enhancing the precision of male fertility diagnostics ... [https://www.nature.com/articles/s41598-025-25052-x]
- 45. Development of a Validation Protocol for ... [https://www.mlj.goums.ac.ir/browse.php?a_id=1464&slc_lang=en&sid=1&ftxt=1&html=1]
- 46. The Motility Ratio method as a novel approach to qualify ... [https://www.nature.com/articles/s41598-024-79500-1]
- 47. Standardized Laboratory Procedures, Quality Control and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8761242/]
- 48. Semen Analysis - StatPearls - NCBI Bookshelf - NIH [https://www.ncbi.nlm.nih.gov/books/NBK564369/]
- 49. Comparison of two automated sperm analyzers using 2 ... [https://www.sciencedirect.com/science/article/abs/pii/S246878472100026X]
- 50. Artificial Intelligence in Andrology: From Semen Analysis to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10782130/]
- 51. Revolutionizing semen analysis: introducing Mojo AISA, ... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2023.1203708/full]
- 52. Artificial Intelligence in Reproductive Medicine: Transforming ... [https://jivfww.scholasticahq.com/article/137620-artificial-intelligence-in-reproductive-medicine-transforming-assisted-reproductive-technologies]
- 53. Developing techniques for determining sperm morphology ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3455498/]
- 54. AndroGen: Open-source synthetic data generation for ... [https://pubmed.ncbi.nlm.nih.gov/41172583/]
- 55. Multidimensional data analysis algorithms facilitate and ... [https://www.sciencedirect.com/science/article/pii/S0093691X25001190]
- 56. Still counting sperm? Why novel, truly informative ... [https://link.springer.com/article/10.1007/s12020-025-04453-y]
- 57. Semen evaluation: methodological advancements in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8255896/]
- 58. A low-cost AI-driven microscope for bull sperm analysis [https://www.sciencedirect.com/science/article/abs/pii/S0924424725006879]
- 59. The Role of Sperm Morphology Standards in ... [https://www.frontiersin.org/journals/veterinary-science/articles/10.3389/fvets.2021.672058/full]