Molecular Diagnostics vs. Nugent Score for Bacterial Vaginosis: A Critical Validation for Research and Drug Development
This article provides a comprehensive analysis of the validation of traditional Nugent scoring against modern molecular diagnostics for bacterial vaginosis (BV).
Molecular Diagnostics vs. Nugent Score for Bacterial Vaginosis: A Critical Validation for Research and Drug Development
Abstract
This article provides a comprehensive analysis of the validation of traditional Nugent scoring against modern molecular diagnostics for bacterial vaginosis (BV). Aimed at researchers, scientists, and drug development professionals, it explores the foundational principles of both methods, details their laboratory application, troubleshoots inherent limitations, and presents a critical comparison of their diagnostic performance. Synthesizing current evidence, the review highlights the superior accuracy, objectivity, and research utility of molecular techniques like qPCR and multiplex PCR, while also considering the contextual value of established microscopic methods. The discussion concludes with future directions for biomarker optimization and the integration of novel technologies in BV research and therapeutic development.
The Bedrock of BV Diagnosis: Understanding the Nugent Score and the Rise of Molecular Methods
Bacterial vaginosis (BV) represents a significant polymicrobial condition affecting women's reproductive health, associated with adverse outcomes including preterm birth and increased susceptibility to sexually transmitted infections [1] [2]. For decades, the Nugent Score has remained the gold standard microbiological method for BV diagnosis in research and clinical practice. This Gram stain-based scoring system quantitatively measures bacterial morphotypes in vaginal smears, assigning scores of 0-3 as BV negative, 4-6 as intermediate, and 7-10 as BV positive [3] [4]. Despite its established position, the Nugent Score faces challenges regarding subjectivity, interpretation of intermediate scores, and its inherent limitation as a morphological rather than etiological assessment [5] [2]. The emergence of molecular diagnostics and artificial intelligence (AI)-enhanced methodologies now prompts a critical re-evaluation of this historical gold standard within the research landscape. This review deconstructs the Nugent Score through objective comparison with modern diagnostic alternatives, providing researchers with experimental data and methodological frameworks for informed assay selection in BV research.
Methodology Deep Dive: Traditional versus Molecular Workflows
Conventional Nugent Scoring Protocol
The traditional Nugent scoring method requires experienced personnel to examine Gram-stained vaginal smears under oil immersion microscopy, evaluating the presence and quantity of three specific bacterial morphotypes: large Gram-positive rods (Lactobacillus species), small Gram-variable rods (Gardnerella vaginalis), and curved Gram-negative rods (Mobiluncus species) [4]. The final score derives from a weighted sum of these morphological assessments:
- Lactobacillus Morphotypes: Scored 0-4 (0: >30 per field; 1: 5-30; 2: 1-4; 3: <1; 4: 0)
- Gardnerella Morphotypes: Scored 0-4 (0: 0; 1: <1; 2: 1-4; 3: 5-30; 4: >30)
- Mobiluncus Morphotypes: Scored 0-2 (0: 0; 1: 1-4; 2: >5)
The composite score (0-10) determines BV status, with the intermediate category (4-6) representing a significant diagnostic challenge [2]. This manual process is time-consuming and subject to inter-observer variability, with diagnostic accuracy in clinical practice often below 50% [6].
Molecular Diagnostic Methodologies
Molecular approaches for BV diagnosis utilize nucleic acid amplification tests (NAATs) to detect and quantify specific BV-associated microorganisms. Common methodologies include:
Multiplex Real-Time PCR (qPCR): Simultaneously quantifies multiple bacterial targets using fluorescent-labeled probes, providing results within hours [5]. Key detected organisms include Gardnerella vaginalis, Fannyhessea vaginae (formerly Atopobium vaginae), Lactobacillus species (L. crispatus, L. iners, L. gasseri, L. jensenii), and BV-associated bacteria 2 (BVAB2) [5] [2] [4].
Transcription-Mediated Amplification (TMA): Used in systems like the Hologic Panther Aptima BV assay, this method targets specific RNA sequences with demonstrated high sensitivity and specificity [1].
Fluorescence In Situ Hybridization (FISH): Combines molecular detection with spatial analysis, allowing visualization of biofilm structures where Gardnerella and other bacteria like F. vaginae and Prevotella bivia form polymicrobial communities [5].
Table 1: Comparative Analytical Targets of BV Diagnostic Methods
| Method Category | Specific Method | Primary Analytical Targets | Output Format |
|---|---|---|---|
| Microscopy | Nugent Scoring | Lactobacillus, Gardnerella, Mobiluncus morphotypes | Semi-quantitative score (0-10) |
| Molecular | Multiplex qPCR | Specific DNA sequences of 4-10 BV-associated species | Quantitative bacterial loads |
| Molecular | Hologic Aptima BV | Specific RNA targets | Qualitative result (positive/negative) |
| Molecular | FISH | 16S rRNA of specific bacteria with spatial context | Visualization of biofilm structure |
| Molecular | NAATs | Organism-specific nucleic acid sequences | Qualitative/quantitative detection |
Performance Comparison: Quantitative Data Analysis
Recent studies provide robust comparative data on the performance of Nugent scoring versus molecular diagnostics:
Diagnostic Accuracy Metrics
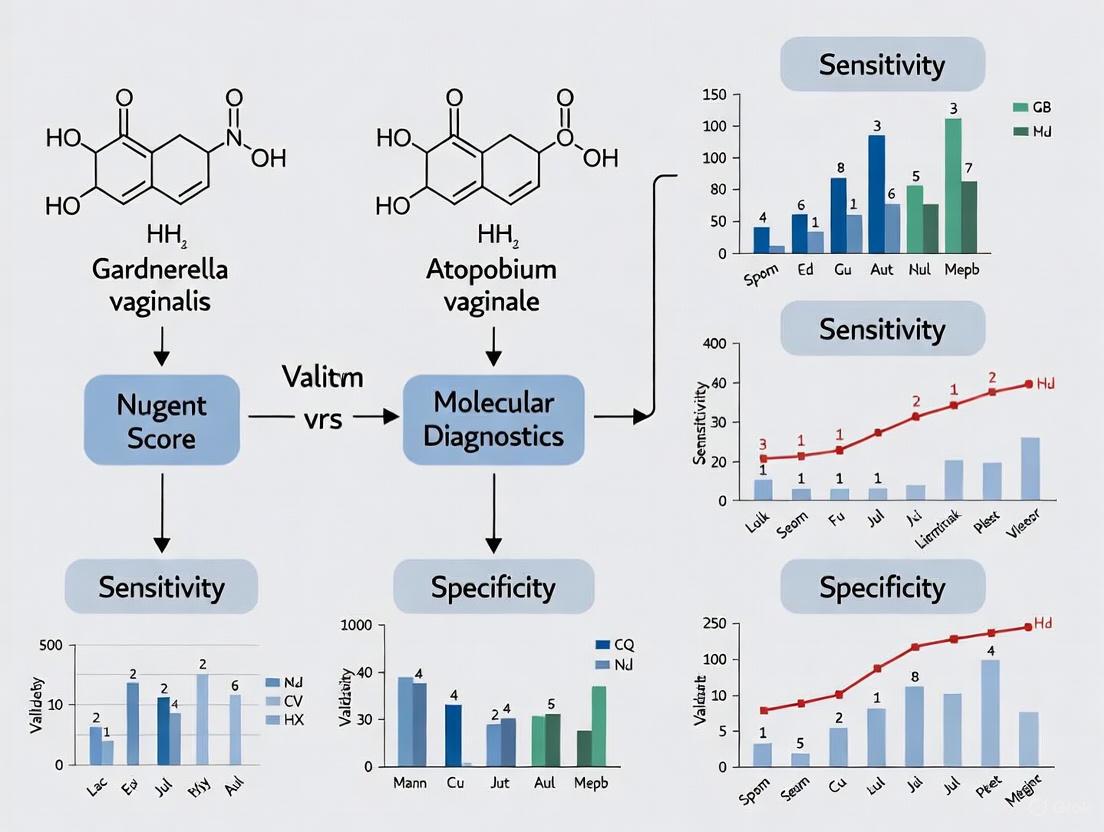
The Hologic Panther Aptima BV assay demonstrated sensitivity of 97.5% and specificity of 96.3% compared to consensus Gram stain and Nugent scoring [1]. Similar performance was observed for the Aptima CV/TV assay for vulvovaginal candidiasis and trichomoniasis [1]. In comparison, molecular tests generally show superior performance to Amsel's criteria, which have sensitivity of only 37-70% in clinical practice [4].
Research by Mala et al. highlighted the particular utility of qPCR for resolving diagnostically challenging intermediate Nugent scores (4-6), which comprised 20.8% of their clinical samples [2]. Their qPCR approach targeting Lactobacillus spp., G. vaginalis, and A. vaginae provided quantitative thresholds that objectively categorized these ambiguous cases.
Optimal Molecular Target Combinations
Investigations into the most efficient molecular marker combinations identified paired detection of Lactobacillus crispatus with Gardnerella vaginalis as particularly effective, achieving 99.29% sensitivity and 97.79% specificity [4]. This two-marker approach outperformed single-marker assays and provided a cost-effective alternative to more extensive multiplex panels targeting 4-7 organisms [4].
Table 2: Performance Comparison of BV Diagnostic Methods
| Diagnostic Method | Sensitivity (%) | Specificity (%) | Intermediate Result Rate | Key Limitations |
|---|---|---|---|---|
| Amsel's Criteria | 37-70 [4] | Not specified | Not applicable | Subjective components; requires clinical symptoms |
| Nugent Scoring | Reference standard | Reference standard | 20.8-32.7% [2] | Subjective interpretation; expertise-dependent |
| Hologic Aptima BV | 97.5 [1] | 96.3 [1] | Minimal | Does not address turn-around-time of wet mounts |
| Multiplex PCR (Various) | 90.5-96.7 [5] | 85.8-95 [5] | Resolves intermediate cases | Higher cost than microscopy |
| L. crispatus + G. vaginalis PCR | 99.29 [4] | 97.79 [4] | Resolves intermediate cases | Requires DNA extraction and PCR capability |
| Femoflor Test | 84.8-99 [5] | 93-96.2 [5] | Resolves intermediate cases | Complex result interpretation |
Emerging Innovations: AI and Machine Learning Applications
Automated Nugent Scoring Systems
AI-powered digital pathology platforms are emerging to standardize Nugent scoring by applying deep learning algorithms to Gram-stained smear images. These systems can classify images into Nugent score categories (0-3: normal; 4-6: intermediate; 7-10: BV) with consistency matching or exceeding human experts [6]. Such automation addresses key limitations of manual microscopy by reducing inter-observer variability and increasing throughput while maintaining the established Nugent framework.
Alternative AI Approaches for Vaginitis Diagnosis
Beyond replicating traditional scoring, novel AI architectures directly analyze vaginal discharge images for pathogen detection. The MobileNetV2 model achieved an F1 score >0.75 and AUC-PR >0.80 across multiple pathogen classes, with particularly strong performance in identifying Gardnerella vaginalis (both metrics >0.90) [7]. This approach demonstrates potential for rapid screening in resource-limited settings.
Addressing Diagnostic Disparities
Machine learning models applied to 16S rRNA sequencing data have revealed important ethnic variations in BV prediction performance, with models typically exhibiting lower accuracy for Black women [3]. This disparity reflects genuine biological variations in vaginal microbiome composition across ethnic groups, highlighting the need for diverse training datasets and population-specific model calibration to ensure equitable diagnostic performance [3].
Diagram 1: Comparative diagnostic pathways for BV
The Research Toolkit: Essential Reagents and Platforms
Table 3: Essential Research Reagents and Platforms for BV Diagnostics
| Category | Specific Product/Platform | Research Application | Key Features |
|---|---|---|---|
| Molecular Detection | Hologic Panther Aptima BV | Comparison studies with conventional methods | Transcription-mediated amplification; high sensitivity [1] |
| Molecular Detection | Femoflor Test | Vaginal microbiota assessment | Multiplex real-time PCR; detects 16+ organisms; provides TBM [5] |
| Molecular Detection | AmpliSens Florocenosis/BV-FRT | BV diagnosis validation | Quantitative PCR for Lactobacillus spp., G. vaginalis, A. vaginae [5] |
| AI-Based Platforms | Nugent Score AI (CarbGeM) | Automated Nugent scoring | Deep learning-based Gram-stain image classification [6] |
| AI-Based Platforms | MobileNetV2 Model | Rapid vaginitis screening | Image analysis of vaginal discharge samples [7] |
| Laboratory Reagents | Gram Stain Kit | Conventional Nugent scoring | Differentiates bacterial morphotypes for microscopy |
| Laboratory Reagents | DNA Extraction Kits | Molecular studies | Magnetic bead-based nucleic acid purification [4] |
| Target Primers/Probes | L. crispatus-G. vaginalis primer sets | Cost-effective BV diagnosis | Dual-target approach with high sensitivity/specificity [4] |
Research Implications and Future Directions
The integration of molecular diagnostics into BV research necessitates reconsideration of the Nugent score's role as a standalone gold standard. While its historical value and extensive literature correlation remain important, molecular methods offer superior objectivity, reproducibility, and ability to resolve diagnostically challenging intermediate cases [5] [2]. The clinical significance of molecularly detected organisms requires careful interpretation, particularly for Lactobacillus species differentiation and quantification of diverse anaerobic communities [1] [4].
Future research directions should focus on standardizing molecular thresholds across platforms, validating findings in diverse populations, and establishing clinical correlations for molecular intermediate profiles. The combination of AI-enhanced microscopy with targeted molecular assays presents a promising integrated approach, leveraging the strengths of both methodologies while addressing their individual limitations [6] [7]. As BV research evolves toward more personalized diagnostic frameworks, the Nugent score will likely transition from gold standard to a component in a multidimensional assessment strategy incorporating morphological, molecular, and clinical data.
Bacterial vaginosis (BV), a common dysbiosis of the vaginal microbiome, presents significant diagnostic challenges in both clinical and research settings. Traditional diagnostic methods, primarily the Nugent scoring system, have long been considered the reference standard. This microscopic technique assesses bacterial morphotypes on Gram-stained vaginal smears, providing a score from 0-10 where scores of 7-10 indicate BV [8]. While inexpensive and established, Nugent scoring suffers from several limitations: it requires trained personnel, demonstrates inter-observer variability, provides only approximate assessment of vaginal microbial composition, and offers limited insight into the complex polymicrobial nature of BV [8]. These limitations have driven the development and adoption of nucleic acid amplification tests (NAATs), which offer objective, sensitive, and comprehensive detection of BV-associated microorganisms, revolutionizing both patient care and research methodologies.
Comparative Analysis: Traditional versus Molecular Diagnostic Methods
The evolution from microscopy to molecular techniques represents a paradigm shift in BV diagnostics. The table below summarizes the key characteristics of available diagnostic methods.
Table 1: Comparison of BV Diagnostic Methods and Their Performance Characteristics
| Method | Principle | Advantages | Disadvantages | Performance |
|---|---|---|---|---|
| Nugent Score [8] | Microscopic evaluation of Gram-stained smear assessing bacterial morphotypes | Unbiased, cost-effective, established reference standard | Time-consuming, requires expert interpretation, does not detect specific bacterial taxa, "intermediate" results (4-6) are hard to interpret | Sensitivity: 89%, Specificity: 83% vs. Amsel's criteria |
| Amsel's Criteria [8] | Clinical criteria including pH, whiff test, clue cells, and discharge | Enables onsite diagnosis without specialized equipment | Subjective, abnormal discharge present in only 50% of BV cases, requires multiple components | Sensitivity: 37-70%, Specificity: 94-99% vs. Nugent score |
| Multiplex PCR [8] | Simultaneous amplification and detection of multiple bacterial DNA targets | Objective, high throughput, detects specific pathogens including fastidious bacteria, provides quantitative data | Higher cost than traditional methods, requires molecular laboratory infrastructure | Sensitivity: 91-97%, Specificity: 77-91% vs. clinical methods |
| Real-Time PCR [8] | Quantitative detection of specific bacterial DNA sequences in real time | Quantitative, reproducible, detects difficult-to-culture bacteria, results in hours | Requires specific equipment and technical expertise, higher cost per test | Detects key BVABs including Fannyhessea vaginae, Gardnerella vaginalis, and Mobiluncus spp. |
| Next-Generation Sequencing [8] | Comprehensive sequencing of 16S rRNA genes from vaginal samples | Provides complete vaginal microbiome profile, discovers novel organisms | Costly, complex data analysis, not suitable for routine clinical use | Sensitivity: 95% compared to clinical methods |
Performance Validation: NAATs Versus Reference Standards
Multiple studies have systematically evaluated the performance of various NAAT platforms against traditional reference methods, with consistent findings of superior sensitivity and variable specificity.
Table 2: Performance Characteristics of Commercial NAAT Platforms for BV Diagnosis
| Test Platform | Study Population | Reference Standard | Sensitivity (%) | Specificity (%) | Study |
|---|---|---|---|---|---|
| Allplex BV Assay | 213 symptomatic South African women | Nugent score | 94.9 | 66.7 | [9] |
| BD Max MVP | 8,878 orders (probe) vs. 10,464 (PCR) | DNA probe testing | BV detection reduced from 30% to 23% | N/A | [10] |
| Multiplex PCR with AI | 288 samples, Chinese cohort | Nugent score | 90.4 | 96.1 | [11] |
| Vaginitis Panel Prototype | 1,209 symptomatic subjects, US | Three FDA-cleared NAATs | 93.8 | 96.1 | [12] |
| BD Affirm VPIII (DNA probe) | Comparative analysis | PCR-based methods | Lower detection of VVC | Higher detection of BV | [10] |
The data reveal several important trends. First, NAATs consistently demonstrate high sensitivity (>90% across most platforms), which is crucial for minimizing false negatives in both clinical practice and research enrollment [9] [11]. Second, specificity shows greater variability between platforms and study populations, with some tests like the Allplex BV Assay showing substantially reduced specificity (66.7%) compared to Nugent scoring [9]. This may reflect the detection of microorganisms that are not assessed by the Nugent score but are nonetheless relevant to vaginal health.
Comparative studies between different molecular platforms reveal additional nuances. When one laboratory migrated from DNA probe-based testing (BD Affirm VPIII) to PCR-based testing (BD Max MVP), they observed a significant reduction in BV positivity (from 30% to 23%) but an increase in vulvovaginal candidiasis (VVC) detection (from 6.3% to 11.6%) [10]. This highlights that different molecular methods can yield substantially different prevalence estimates for vaginal conditions, with important implications for both clinical management and research findings.
Molecular Testing Methodologies: Protocols and Workflows
Sample Collection and Nucleic Acid Extraction
The analytical process for NAAT-based BV diagnosis begins with proper sample collection. For most commercial systems, this involves obtaining vaginal swabs—either clinician-collected or self-collected—from the posterior fornix or vaginal wall [13] [11]. Studies have demonstrated that self-collected vaginal swabs perform equivalently to clinician-collected specimens for molecular testing, increasing patient accessibility and convenience [13].
Following collection, nucleic acid extraction is performed using commercial kits. The process typically involves:
- Cell lysis to release nucleic acids
- Purification from proteins and other contaminants
- Elution in a suitable buffer for amplification [11]
Extracted DNA is quality-checked using spectrophotometric methods (e.g., NanoDrop) to ensure adequate concentration and purity before proceeding to amplification [11].
Target Selection and Amplification Strategies
Different NAAT platforms employ distinct target selection strategies for BV diagnosis:
- BD Max MVP utilizes real-time PCR to detect a panel of organisms including lactobacilli (L. crispatus and L. jensenii) and BV-associated bacteria (G. vaginalis, A. vaginae, Megasphaera-1, and BVAB-2), with algorithmic analysis to determine BV status [10]
- Research-use-only multiplex PCR assays may target up to 15 different organisms, including four Lactobacillus species and 11 BV-associated pathogens [11]
- Allplex BV Assay simultaneously detects individual targets using qualitative and quantitative analysis in a multiplex real-time PCR format [9]
The workflow for molecular BV testing follows a standardized pathway that can be visualized as follows:
Interpretation Algorithms
A critical advancement in molecular BV testing is the use of interpretation algorithms to convert microbial detection data into clinical diagnoses. Unlike traditional methods that rely on visual assessment, NAATs employ either:
- Fixed threshold algorithms that quantify specific bacterial ratios
- Machine learning approaches that integrate multiple microbial abundances to improve diagnostic accuracy [11]
One study developed a support vector machine (SVM) algorithm that achieved superior performance (AUC: 0.969, sensitivity: 90.4%, specificity: 96.1%) compared to Nugent scoring, particularly for intermediate cases (Nugent 4-6) [11]. Another research group utilized machine learning to develop a BV algorithm demonstrating 93.8% sensitivity and 96.1% specificity compared to three FDA-cleared NAATs [12].
Research Implications and Clinical Outcomes
Impact on Clinical Research
The adoption of NAATs for BV diagnosis has profound implications for research:
- Improved diagnostic consistency across multi-center trials
- Enhanced ability to detect mixed infections that may confound treatment outcomes
- Objective endpoints for interventional studies
- Stratification capabilities based on specific microbial profiles rather than broad clinical categories
Real-world evidence demonstrates that the implementation of molecular testing significantly impacts patient management and healthcare utilization. A retrospective claims analysis found that patients receiving syndromic multiplex RT-PCR tests with next-day results had significantly lower mean total healthcare costs ($5,607) over six months compared to those receiving no testing ($6,680) [14]. The RT-PCR group also demonstrated lower outpatient service utilization and costs compared to both other PCR tests and no testing cohorts [14].
Point-of-Care Advancements
Recent developments in point-of-care (POC) molecular testing promise to further transform BV research and management. The Xpert Xpress MVP test, the first CLIA-waived, FDA-cleared POC NAAT for vaginal infections, provides results within one hour with minimal hands-on time [13]. User experience studies demonstrate that the test can be accurately performed by operators with varying educational backgrounds and clinical roles, maintaining consistent performance regardless of user qualifications [13]. This decentralization of complex testing enables rapid recruitment and classification of research participants in diverse settings.
Essential Research Reagents and Platforms
Table 3: Key Research Reagents and Platforms for Molecular BV Diagnosis
| Reagent/Platform | Function | Application in BV Research |
|---|---|---|
| BD Max MVP [10] | Automated qualitative test for detection of BV-associated organisms | Identifies BV through algorithmic analysis of lactobacilli and BV-associated bacteria |
| Allplex BV Assay [9] | Multiplex real-time PCR for simultaneous detection of BV targets | Qualitative and quantitative analysis of multiple BV-related pathogens in research settings |
| Xpert Xpress MVP [13] | POC PCR system for rapid detection of vaginal infections | CLIA-waived system for decentralized testing in clinical trials and community-based research |
| Femoflor-16 [8] | Multiplex real-time PCR test for vaginal microbiota assessment | Determines total bacterial DNA and assesses vaginal microbiota composition in research |
| Primer/Probe Sets [11] | Species-specific oligonucleotides for PCR amplification | Custom detection of specific vaginal microorganisms in mechanistic studies |
| Nucleic Acid Extraction Kits [11] | Isolation and purification of microbial DNA from vaginal swabs | Standardized preparation of samples for molecular analysis in multi-center trials |
The molecular shift in BV diagnostics represents more than just technological advancement—it constitutes a fundamental transformation in how researchers conceptualize, classify, and investigate this common condition. NAATs provide unprecedented precision in detecting the complex polymicrobial communities associated with BV, enabling more stratified research populations and more meaningful endpoints in clinical trials.
While the Nugent score remains an important historical reference method, its limitations in sensitivity, objectivity, and comprehensive microbial assessment have become increasingly apparent. Molecular methods offer enhanced reproducibility between research sites, the ability to detect fastidious organisms, and opportunities for innovative diagnostic algorithms incorporating machine learning.
As point-of-care molecular platforms become more accessible, the potential for rapid recruitment and classification of research participants across diverse settings will continue to grow. Future research will likely focus on refining diagnostic algorithms, establishing quantitative thresholds for different clinical contexts, and correlating specific microbial profiles with long-term health outcomes. The continued validation and refinement of NAATs for BV will be essential for advancing both scientific understanding and clinical management of this complex condition.
The diagnosis of bacterial vaginosis (BV) has long been anchored in the microscopic examination of bacterial morphotypes, yet recent molecular advances are fundamentally reshaping diagnostic targets. BV, a common gynecological condition affecting millions of women globally, represents a complex dysbiosis of the vaginal microbiome characterized by a reduction of lactic acid-producing lactobacilli and an overgrowth of anaerobic bacteria [15]. This shift in microbial community structure poses significant public health concerns, as BV is associated with adverse reproductive outcomes including preterm birth, pelvic inflammatory disease, and increased susceptibility to sexually transmitted infections including HIV [15] [3]. The diagnostic journey for BV reflects our evolving understanding of its polymicrobial nature—from initial observations of morphological changes to contemporary genetic analyses that reveal intricate microbial ecosystems. This comparison guide examines the validation evidence for traditional and molecular diagnostic approaches, providing researchers and drug development professionals with critical performance data and methodological insights to inform diagnostic selection and development.
Established Diagnostic Standards: Morphological Assessment
Nugent Scoring: The Historical Gold Standard
The Nugent scoring system, developed in 1991, remains the reference method for BV diagnosis in research settings. This laboratory-based technique involves Gram staining vaginal smears and microscopically evaluating bacterial morphology to generate a quantitative score from 0-10 [15] [16]. The scoring algorithm quantifies the ratio of Lactobacillus morphotypes (large Gram-positive rods) to BV-associated bacteria (small Gram-variable rods and curved Gram-negative rods) [16]. Scores of 0-3 are considered normal, 4-6 intermediate, and 7-10 confirm BV [3] [16]. While regarded as the gold standard for decades, this method suffers from several limitations: it requires experienced personnel for consistent interpretation, demonstrates inter-observer variability, and necessitates considerable time and skill to perform accurately [15] [16]. Additionally, the Nugent score was developed based on morphological characteristics that may not fully capture the complexity of vaginal microbial communities.
Amsel Criteria: Clinical Diagnosis in Practice
For clinical settings, the Amsel criteria provide a practical diagnostic framework requiring at least three of four clinical findings: homogeneous grayish-white vaginal discharge, vaginal pH >4.5, positive whiff test (amine odor upon adding potassium hydroxide), and presence of clue cells on microscopic examination [17] [15]. Though widely used, this method also demonstrates limitations in sensitivity and specificity, with some components like the whiff test being subjective and inconsistently applied in practice [17]. Some clinical settings have adapted the original criteria, such as replacing the whiff test with reported malodorous discharge, creating "Amsel-like" criteria [17].
Simplified Morphological Assessment
To address the complexity of Nugent scoring, simplified morphological classification systems have been developed. The Hay/Ison criteria categorize vaginal flora into three groups: normal (Lactobacillus morphotypes only), intermediate (reduced Lactobacillus with mixed bacterial morphotypes), and BV (mixed bacterial morphotypes with few or absent Lactobacillus) [16]. Studies comparing these methods to Nugent scoring demonstrate excellent agreement, with sensitivity ≥97.2%, specificity ≥88.1%, and kappa values ≥0.830 [16]. This simplified approach offers a practical alternative for busy clinical laboratories while maintaining diagnostic accuracy.
Molecular Diagnostics: Genetic Marker-Based Approaches
Molecular diagnostics for BV represent a paradigm shift from morphological observation to genetic marker detection, offering potentially greater objectivity, sensitivity, and specificity.
Nucleic Acid Amplification Tests (NAATs)
NAATs detect and quantify BV-associated organisms through target-specific amplification, providing improved reproducibility and consistency compared to traditional methods [1]. The Hologic Panther Aptima BV assay demonstrates sensitivity of 97.5% and specificity of 96.3% compared to consensus Gram stain and Nugent scoring [1]. Similarly, the Aptima CV/TV assay shows 100% sensitivity for both Candida vaginitis (CV) and Trichomonas vaginalis (TV), with specificities of 83.5% (compared to Gram stain) and 100% (compared to culture and TV PCR), respectively [1]. These assays utilize transcription-mediated amplification to detect specific RNA targets, providing direct detection of organisms rather than relying on morphological proxies.
Quantitative PCR (qPCR) assays targeting specific BV-associated bacteria like Fannyhessea vaginae and Gardnerella vaginalis have also been developed and validated [17]. One study evaluating such a qPCR assay found it diagnosed 31/130 (24%) patients with BV, showing 95.2% sensitivity and 100% specificity when the Nugent score was considered reference standard [17]. When the qPCR itself was used as reference, the Nugent score demonstrated 95.2% sensitivity and 95.3% specificity [17].
Advanced Molecular Approaches
Matrix-assisted laser desorption ionization–time of flight mass spectrometry (MALDI-TOF MS) represents an alternative molecular approach that analyzes protein profiles to identify microorganisms [17]. In one evaluation, MALDI-TOF MS ("VAGI-TOF") provided BV diagnoses consistent with qPCR, diagnosing 32/130 (25%) patients with BV [17]. When qPCR was reference, MALDI-TOF MS showed 90.5% sensitivity and 95.3% specificity [17]. This technology offers rapid, reproducible, and potentially lower-cost identification compared to some molecular methods.
More sophisticated genetic analyses continue to refine our understanding of BV pathogenesis. Genomic studies have revealed greater diversity within Gardnerella species than previously recognized, identifying 11 unique genospecies with differential antibiotic resistance patterns [18]. This expanded taxonomy enables development of more precise PCR tests that can distinguish metronidazole-resistant strains, potentially guiding targeted therapy [18].
Comparative Performance Data
Table 1: Comparative Performance of BV Diagnostic Methods Against Reference Standards
| Diagnostic Method | Reference Standard | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | Study |
|---|---|---|---|---|---|---|
| Amsel-like Criteria | qPCR | 90.5 | 84.1 | - | - | [17] |
| Nugent Score | qPCR | 95.2 | 95.3 | - | - | [17] |
| MALDI-TOF MS | qPCR | 90.5 | 95.3 | - | - | [17] |
| Hay's Criteria | Nugent Score | 97.2-100 | 88.1-93.4 | 80.4-93.7 | 97.1-100 | [16] |
| Hologic Aptima BV | Gram stain/Nugent | 97.5 | 96.3 | - | - | [1] |
| qPCR (F. vaginae/G. vaginalis) | Nugent Score | 87.0 | 100 | - | - | [17] |
Table 2: BV Prevalence by Diagnostic Method in Comparative Studies
| Diagnostic Method | BV Prevalence (%) | Sample Size | Study Population | Study |
|---|---|---|---|---|
| Vaginal Discharge + pH>4.5 | 42.0 | 129 | Symptomatic women | [17] |
| Amsel-like Criteria | 39.0 | 118 | Symptomatic women | [17] |
| Nugent Score | 27.0 | 84 | Symptomatic women | [17] |
| qPCR | 25.0 | 84 | Symptomatic women | [17] |
| MALDI-TOF MS | 25.0 | 84 | Symptomatic women | [17] |
| Nugent Score | 32.9 | 213 | Community-based | [16] |
| Hay's Criteria | 40.8 | 213 | Community-based | [16] |
Experimental Protocols and Methodologies
Nugent Scoring Methodology
The Nugent score protocol involves careful specimen collection and processing:
- Specimen Collection: Vaginal swabs are collected from the posterior fornix using appropriate collection swabs.
- Smear Preparation: Smears are prepared on glass slides and air-dried.
- Gram Staining: Following standardized staining protocols (e.g., crystal violet for 1 minute, Gram's iodine for 1 minute, acetone decolorization for 2-3 seconds, safranin counterstain for 1 minute).
- Microscopic Examination: Stained smears are examined under oil immersion at 1000x magnification.
- Scoring: Bacterial morphotypes are quantified per high-power field:
- Lactobacillus morphotypes (large Gram-positive rods)
- Gardnerella and Bacteroides morphotypes (small Gram-variable rods)
- Mobiluncus morphotypes (curved Gram-negative rods)
- Calculation: Composite score (0-10) is calculated based on weighted combinations of morphotype quantities [16].
Molecular Assay Protocols
qPCR Methodology for BV Diagnosis
Detailed protocol for qPCR detection of BV-associated bacteria:
- DNA Extraction:
- Automated extraction systems (e.g., EZ1 automate) with commercial kits (e.g., QIAamp Tissue Kit)
- 200 μL vaginal sample digested with G2 buffer and proteinase K at 56°C for 20 minutes
- DNA eluted in 100 μL distilled water [17]
qPCR Reaction Setup:
- 15 μL PCR mix containing:
- 10 μL Probe PCR Master Mix
- 3 μL DNAse/RNAse-free distilled water
- 0.5 μL each forward/reverse primer (50 μM)
- 0.5 μL probe (50 μM)
- 0.5 μL Uracil DNA glycosylase (UDG)
- 5 μL extracted DNA template [17]
- 15 μL PCR mix containing:
Amplification Program:
- UDG incubation: 50°C for 2 minutes
- Initial denaturation: 95°C for 5 minutes
- 39 cycles of:
- Denaturation: 95°C for 5 seconds
- Annealing/Extension: 60°C for 30 seconds [17]
Analysis: Quantification of Fannyhessea vaginae and Gardnerella vaginalis with predetermined thresholds for BV diagnosis [17].
Hologic Aptima Assay Protocol
The Hologic Panther System assays utilize transcription-mediated amplification:
- Sample Processing: Automated extraction on Panther system
- Target Amplification: Transcription-mediated amplification of specific RNA targets
- Detection: Chemiluminescent detection of amplification products
- Interpretation: Automated result interpretation based on predefined cutoffs [1]
Diagram Title: BV Diagnostic Method Workflows
Emerging Research and Diagnostic Innovations
Machine Learning in BV Diagnosis
Advanced computational approaches are being applied to BV diagnostics with promising results. Machine learning (ML) models using 16S rRNA sequencing data can predict BV with high accuracy (balanced accuracy: 0.90-0.92; AUPRC: 0.93-0.96) [3]. However, these models demonstrate performance variations across ethnic groups, with lower accuracy for Black women who more frequently have diverse vaginal microbiomes [3]. Random Forest and Logistic Regression models show the highest predictive performance, but Multi-layer Perceptron models demonstrate more comparable performance across ethnic groups [3]. Feature selection reveals unique bacterial taxa important for accurate BV prediction that vary between ethnic groups, suggesting that personalized diagnostic approaches may be necessary.
Strain-Level Differentiation and Resistance Profiling
Novel PCR tests are being developed to distinguish between different Gardnerella genospecies with differential antibiotic resistance patterns [18]. Genome sequencing of 129 Gardnerella genomes revealed 11 unique genospecies within several major clades, with two clades comprising five genospecies showing 100% resistance to metronidazole but susceptibility to clindamycin [18]. This strain-level differentiation enables development of tests that can guide targeted antibiotic therapy, potentially improving treatment outcomes and reducing recurrence.
Microbiota Analysis and BV Subtyping
Next-generation sequencing approaches have identified distinct BV subtypes with different clinical implications. One study found two distinct types of BV infection, one of which increases chlamydia risk [19]. This suggests that BV subtyping could enhance risk stratification and targeted intervention strategies. Another study comparing diagnostic methods found that the AmpliSens Florocenosis/BV-FRT PCR assay showed greatest agreement with microbiota analysis (80.6% sensitivity) compared to Amsel criteria (61.1%), Nugent score (63.9%), culture (38.9%), and the BD MAX Vaginal Panel (63.9%) [20].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for BV Diagnostic Studies
| Reagent/Equipment | Application | Specific Function | Example Products/References |
|---|---|---|---|
| Gram Stain Kits | Traditional morphology | Differentiates bacterial types based on cell wall properties | Color Gram-2 Kit [17] |
| pH Test Strips | Clinical criteria | Measures vaginal fluid pH (>4.5 suggests BV) | EcoCare pH-cotton stick [17] |
| DNA Extraction Kits | Molecular assays | Nucleic acid purification from vaginal samples | QIAamp Tissue Kit [17] |
| qPCR Master Mixes | Molecular quantification | Amplification and detection of target sequences | Eurogentec Probe PCR Master Mix [17] |
| Specific Primers/Probes | Targeted detection | Amplification of BV-associated bacteria | Fannyhessea vaginae, Gardnerella vaginalis primers [17] |
| Automated Extraction Systems | High-throughput processing | Standardized nucleic acid preparation | EZ1 automate [17] |
| MALDI-TOF MS Systems | Protein profiling | Microbial identification by mass spectrometry | VAGI-TOF platform [17] |
| 16S rRNA Sequencing Reagents | Microbiome analysis | Comprehensive microbial community profiling | [3] |
The diagnostic targets for BV have evolved significantly from bacterial morphotypes to genetic markers, reflecting advances in our understanding of vaginal microbiome complexity. While traditional methods like Nugent scoring and Amsel criteria remain important references, molecular diagnostics offer improved sensitivity, specificity, and objectivity. The research landscape continues to advance with machine learning approaches, strain-level differentiation, and BV subtyping promising even more precise diagnostic capabilities. For researchers and drug development professionals, selection of diagnostic methods must balance performance characteristics with practical considerations, while acknowledging that the diagnostic gold standard continues to evolve alongside our understanding of BV pathogenesis. Future directions point toward more personalized diagnostic approaches that account for microbial diversity across populations and enable targeted therapeutic interventions.
The Clinical and Research Landscape Driving the Need for Improved Diagnostics
Bacterial vaginosis (BV) represents a significant diagnostic challenge in clinical practice and research, affecting millions of women globally with substantial healthcare costs exceeding $1.2 billion annually in the United States alone [13] [21]. BV is characterized by a shift in the vaginal microbiome from lactobacilli dominance to a polymicrobial community, but this transition does not always correlate neatly with clinical symptoms [1] [22]. The complexity of BV pathogenesis, wherein the presence or absence of specific organisms does not necessarily indicate clinical disease, has fueled ongoing debate about optimal diagnostic approaches [1]. Historically, diagnosis has relied on either clinical criteria (Amsel's criteria) or microscopic evaluation (Nugent scoring), but recent advances in molecular technologies offer new possibilities for more accurate and precise detection. This comparison guide objectively evaluates the performance of traditional versus molecular diagnostic methods for BV, providing researchers and drug development professionals with experimental data and methodologies to inform study design and technology adoption.
Established Diagnostic Methods: Foundations and Limitations
Clinical and Microscopic Diagnostic Standards
The two traditional methods for BV diagnosis—Amsel's clinical criteria and Nugent scoring—have formed the foundation of BV diagnosis for decades but present significant limitations in both clinical and research settings.
Amsel's criteria require at least three of four clinical findings: (1) homogeneous, thin, grayish-white discharge; (2) vaginal pH >4.5; (3) positive amine ("fishy") odor when vaginal discharge is mixed with 10% KOH (whiff test); and (4) presence of clue cells on microscopic examination [23]. While simple to perform and inexpensive, this approach demonstrates variable sensitivity (37-70%) though relatively high specificity (94-99%) when compared to Nugent scoring [23]. A 2021 study comparing these methods found Amsel's criteria had a sensitivity of only 50% with specificity of 98.2% when Nugent scoring was used as the reference standard [24].
Nugent scoring, considered the historical gold standard for research, involves Gram staining of vaginal smears and microscopic evaluation of bacterial morphotypes to generate a score from 0-10 [24] [23]. A score of 7-10 indicates BV, 4-6 intermediate flora, and 0-3 normal vaginal flora. While offering greater standardization than clinical criteria, Nugent scoring has significant limitations: it requires considerable time, expertise in microscopy, and laboratory equipment [24]. Additionally, it cannot identify specific bacterial species or their potential contributions to pathogenesis, and the intermediate category (score 4-6) creates diagnostic uncertainty [1].
Table 1: Performance Characteristics of Traditional BV Diagnostic Methods
| Diagnostic Method | Sensitivity Range | Specificity Range | Key Advantages | Key Limitations |
|---|---|---|---|---|
| Amsel's Clinical Criteria | 37-70% [23] 50% [24] | 94-99% [23] 98.2% [24] | Rapid, inexpensive, clinical correlation | Subjective, variable sensitivity, requires symptoms |
| Nugent Scoring (Gram stain) | Reference standard | Reference standard | Standardized, reproducible | Time-consuming, requires expert microscopist, intermediate category |
| BVBlue (Sialidase Test) | 38-91.7% [25] | 91-100% [25] | Rapid point-of-care (10-15 minutes) | Cannot detect non-sialidase producers |
Point-of-Care Innovations: Bridging Traditional and Molecular Methods
Point-of-care tests like BVBlue represent an intermediate diagnostic approach, detecting elevated sialidase enzymes in vaginal fluid samples [25]. This chromogenic test provides results within 15 minutes and demonstrates variable sensitivity (38-91.7%) but consistently high specificity (91-100%) compared to Nugent scoring [25] [26]. The test detects sialidase activity at levels ≥7.8 U, with one unit defined as the amount of enzyme required to liberate 1 nmol of substrate/ml/min at 37°C [25]. However, a significant limitation is its inability to detect BV-associated bacteria that do not produce sialidase enzymes, potentially missing cases involving these organisms [25].
Molecular Diagnostic Advances: Performance and Applications
Nucleic Acid Amplification Tests (NAATs) for BV
Molecular diagnostics represent a paradigm shift in BV diagnosis, using nucleic acid amplification to detect specific BV-associated bacteria with superior accuracy and objectivity compared to traditional methods. Several FDA-cleared and laboratory-developed tests are now available, offering different technological approaches and target combinations.
The Aptima BV assay (Hologic) utilizes transcription-mediated amplification to detect targets including G. vaginalis, A. vaginae, and Lactobacillus species (L. crispatus, L. jensenii, L. gasseri) [1] [23]. Clinical validation studies demonstrate sensitivity of 95.0-97.3% and specificity of 85.8-89.6% using either clinician- or patient-collected vaginal swabs [23]. A 2025 study by Elvy et al. reported sensitivity of 97.5% and specificity of 96.3% for BV diagnosis compared to consensus Gram stain and Nugent score [1].
The BD Max Vaginal Panel provides algorithmic analysis of DNA detection for Lactobacillus species (L. crispatus and L. jensenii) plus G. vaginalis, A. vaginae, BVAB2, and Megasphaera type 1 [23]. This multiplex PCR assay demonstrates 90.5% sensitivity and 85.8% specificity for BV diagnosis compared to Amsel criteria and Nugent score, while also detecting Candida species and T. vaginalis [23].
Other laboratory-developed tests include the NuSwab VG (LabCorp), OneSwab BV Panel PCR with Lactobacillus Profiling by qPCR (Medical Diagnostic Laboratories), and SureSwab BV (Quest Diagnostics), which show similar performance characteristics to FDA-cleared assays but require internal validation before clinical use [23].
Table 2: Performance Characteristics of Molecular BV Diagnostic Assays
| Molecular Assay | Technology | Key Targets | Sensitivity | Specificity |
|---|---|---|---|---|
| Aptima BV (Hologic) | Transcription-mediated amplification | G. vaginalis, A. vaginae, Lactobacillus species (L. crispatus, L. jensenii, L. gasseri) | 95.0-97.5% [1] [23] | 85.8-96.3% [1] [23] |
| BD Max Vaginal Panel | Quantitative multiplex PCR | Lactobacillus species (L. crispatus, L. jensenii), G. vaginalis, A. vaginae, BVAB2, Megasphaera type 1 | 90.5% [23] | 85.8% [23] |
| Xpert Xpress MVP | PCR-based NAAT | Anaerobic bacteria associated with BV, Candida species, T. vaginalis | High PPA across all targets [13] | High NPA across all targets [13] |
Sequencing Technologies and Machine Learning Approaches
Advanced sequencing technologies provide unprecedented resolution for characterizing the vaginal microbiome in BV research, though they remain primarily research tools rather than clinical diagnostics.
16S rRNA gene sequencing (metataxonomics) enables comprehensive profiling of vaginal microbiota composition and assignment to Community State Types (CSTs) [3] [22]. CST I-IV are characterized by dominance of specific lactobacilli (L. crispatus, L. gasseri, L. iners, and L. jensenii, respectively), while CST IV represents a polymicrobial community associated with BV [3] [22]. This approach reveals significant ethnic variation in CST distribution, with Black women and women of other ethnicities more frequently having CST IV (56% and 50%, respectively) compared to White women (33%) [3].
Machine learning algorithms applied to sequencing data show promise for BV prediction but demonstrate ethnic disparities in performance [3]. Models including Random Forest, Logistic Regression, Support Vector Machine, and Multi-layer Perceptron exhibit lower balanced accuracy and higher false positive rates for Black women compared to other ethnic groups [3]. This highlights the importance of diverse training datasets and customized analytical approaches for different populations.
Multi-method comparisons reveal significant discordance between sequencing approaches. Concordance between metatranscriptomic and metataxonomic-based CST assignment can be as low as 59%, emphasizing how methodological choices influence research outcomes [22]. Each sequencing method provides complementary insights: DNA-based methods (metataxonomics and metagenomics) estimate community composition, while RNA-based metatranscriptomics captures microbial activity and viability [22].
Experimental Protocols and Methodologies
Molecular Assay Validation Protocols
The 2025 evaluation of Hologic Panther Aptima BV and CV/TV assays followed a rigorous methodology representative of molecular diagnostic validation studies [1]. The study utilized 300 vaginal swabs from symptomatic patients aged 14-60 years, with comparative reference methods including consensus Gram stain with Nugent scoring for BV, and Gram stain/culture with Aptima TV singleplex assay for CV/TV [1]. Testing was performed in a clinical reference laboratory setting serving both community and hospitalized patients. The statistical analysis included calculation of sensitivity, specificity, and positive and negative predictive values with corresponding confidence intervals, following standard procedures for diagnostic test evaluation [1].
The user experience study for the Xpert Xpress MVP test implemented a different methodological approach focused on operational characteristics [13]. This multi-site prospective study enrolled 22 users with varying backgrounds (clinicians, non-clinicians, and support staff) and educational levels. Users received only the investigational Instructions for Use and Quick Reference Instructions without additional training, simulating real-world implementation conditions [13]. Performance was assessed through positive percent agreement (PPA) and negative percent agreement (NPA) compared to reference methods, while user experience was evaluated via a 19-item questionnaire using a 5-point Likert scale [13].
Sequencing and Machine Learning Workflows
Machine learning studies for BV prediction employ sophisticated data processing and analysis pipelines [3]. The typical workflow begins with 16S rRNA sequencing data processed into operational taxonomic units (OTUs), followed by dimensionality reduction techniques such as t-distributed Stochastic Neighbor Embedding (t-SNE) for visualization [3]. Multiple ML algorithms are then trained and optimized, with hyperparameter tuning conducted for each classifier. Model performance is evaluated using metrics including balanced accuracy (BACC), area under the precision-recall curve (AUPRC), false positive rate (FPR), and false negative rate (FNR), with statistical testing to assess differences between ethnic groups [3].
Sequencing method comparisons require careful experimental design to evaluate concordance between platforms [22]. Research by et al. implemented parallel metataxonomic, metagenomic, and metatranscriptomic sequencing on the same patient samples, enabling direct comparison of microbial communities detected by each approach [22]. Statistical analyses included PERMANOVA based on UMAP distance matrices to assess microbiome variation by Amsel's criteria and Nugent score groupings, with pairwise comparisons between Nugent score categories [22].
Diagram 1: BV Diagnostic and Research Methodologies Evolution. This workflow illustrates the progression from traditional clinical methods to advanced molecular approaches and computational analytics in BV research.
The Researcher's Toolkit: Essential Reagents and Platforms
Table 3: Research Reagent Solutions for BV Diagnostic Studies
| Category | Specific Products/Assays | Research Application | Key Features |
|---|---|---|---|
| Commercial NAAT Platforms | Aptima BV (Hologic), BD Max Vaginal Panel, Xpert Xpress MVP | Clinical validation studies, diagnostic accuracy research | FDA-cleared, high sensitivity/specificity, multiplex detection |
| Laboratory-Developed Tests | NuSwab VG, OneSwab BV Panel, SureSwab BV | Research use, requires internal validation | Customizable targets, research flexibility |
| Point-of-Care Tests | BVBlue, FemExam Test Card, Affirm VP III | Rapid diagnosis, resource-limited settings | Chromogenic detection, 10-15 minute turnaround |
| Sequencing Approaches | 16S rRNA sequencing, metagenomics, metatranscriptomics | Microbiome profiling, microbial ecology studies | High resolution, species identification, functional insights |
| Computational Tools | Random Forest, Logistic Regression, SVM, MLP | Predictive modeling, biomarker discovery | Handles high-dimensional data, identifies complex patterns |
The diagnostic landscape for bacterial vaginosis is undergoing a significant transformation, moving from subjective clinical criteria and labor-intensive microscopy toward precise molecular characterization. Molecular diagnostics offer substantial advantages in sensitivity, specificity, and objectivity compared to traditional methods, though they present higher costs and operational complexity [1] [23]. The emergence of point-of-care NAATs represents a particularly promising development, combining the accuracy of molecular methods with the rapid turnaround needed for immediate treatment decisions [13].
For researchers and drug development professionals, methodological choices should align with specific research questions. Traditional methods retain value for clinical correlation studies, while molecular approaches enable deeper investigation of microbial ecology and pathogenesis [3] [22]. Future directions include addressing ethnic disparities in diagnostic performance, developing standardized analytical pipelines for sequencing data, and validating point-of-care platforms across diverse healthcare settings [3] [13]. The integration of machine learning with multi-omics data holds particular promise for advancing our understanding of BV heterogeneity and developing more targeted interventions [3].
From Bench to Biomarker: A Practical Guide to Executing Nugent and Molecular Assays
For decades, the Nugent score has served as the reference standard for diagnosing bacterial vaginosis (BV) in research settings, providing an objective microscopic method to quantify bacterial morphotypes in vaginal flora. This gram stain scoring system, developed by Robert P. Nugent in 1991, offers a reproducible approach for classifying vaginal microbiota based on the relative proportions of beneficial lactobacilli versus BV-associated pathogens. While molecular diagnostics have emerged as powerful alternatives with superior sensitivity for detecting specific pathogens, the Nugent score remains a vital tool for understanding vaginal microbiome composition and validating newer diagnostic methodologies in scientific investigations. This protocol details the standardized execution and interpretation of the Nugent score while contextualizing its performance against evolving molecular techniques in contemporary BV research.
Materials and Equipment
Essential Research Reagent Solutions
Table 1: Key Reagents and Materials for Nugent Scoring
| Item | Function/Application |
|---|---|
| Gram Stain Kit | Differentiation of bacterial morphotypes based on cell wall properties [27]. |
| Microscope with Oil Immersion Lens | Visualization and counting of bacteria at 1000x magnification [27] [28]. |
| Vaginal Swabs | Collection of vaginal fluid specimen from the posterior fornix or vaginal walls [29]. |
| Microscope Slides | Preparation of thin smear for Gram staining and microscopic examination [27]. |
| pH Test Strips (pH 4.0-7.0) | Complementary assessment of vaginal fluid pH (>4.5 suggests BV) [28] [23]. |
Step-by-Step Protocol
Sample Collection and Preparation
- Collection: Using a sterile swab, obtain a sample of vaginal fluid from the posterior fornix or vaginal walls during a speculum examination [29].
- Smear Preparation: Roll the swab across a clean glass microscope slide to create a thin, even smear.
- Fixation: Air-dry the smear completely and then heat-fix it by briefly passing the slide through a flame.
- Gram Staining: Follow standard Gram staining procedures (Crystal Violet, Iodine, Decolorizer, Safranin) according to your laboratory's validated protocol or manufacturer's instructions [27].
Microscopic Examination and Scoring
- Microscope Setup: Use a microscope with an oil immersion lens to examine at least 10-20 fields of view under 1000x magnification [27] [28].
Bacterial Enumeration: For each field, count and average the number of three specific bacterial morphotypes:
- Large Gram-positive rods (Lactobacillus morphotypes)
- Small Gram-variable rods (Gardnerella vaginalis and Bacteroides morphotypes)
- Curved Gram-variable rods (Mobiluncus spp. morphotypes)
Morphotype Scoring: Assign scores for each category based on the average number of bacteria per field, as defined in Table 2.
Table 2: Nugent Score Calculation Criteria [27] [28]
| Bacterial Morphotype | Score 0 | Score 1 | Score 2 | Score 3 | Score 4 |
|---|---|---|---|---|---|
| Lactobacillus (Large Gram-positive rods) | >30 | 5-30 | 1-4 | <1 | 0 |
| Gardnerella / Bacteroides (Small Gram-variable rods) | 0 | <1 | 1-4 | 5-30 | >30 |
| Mobiluncus (Curved Gram-variable rods) | 0 | <1 to 4 | ≥5 | - | - |
Interpretation of Results
- Final Score Calculation: Sum the scores from all three morphotype categories. The total will range from 0 to 10.
- Diagnostic Classification:
- Nugent Score 0-3: Negative for BV. Indicates a healthy, Lactobacillus-dominated microbiota [27] [23].
- Nugent Score 4-6: Intermediate. Represents a transitional microbiota [27] [23].
- Nugent Score 7-10: Positive for BV. Indicates a shift toward a polymicrobial anaerobic flora characteristic of BV [27] [23].
Performance Comparison: Nugent Score vs. Molecular Diagnostics
Molecular diagnostics for BV represent a paradigm shift from morphological assessment to nucleic acid-based detection of specific BV-associated organisms. These tests, including real-time PCR panels and NAATs, target a combination of beneficial bacteria (e.g., L. crispatus, L. jensenii) and pathogenic species (e.g., G. vaginalis, Fannyhessea vaginae, BVAB2) to determine microbial balance [29] [23] [4].
Table 3: Comparative Analysis of Nugent Score and Molecular Diagnostics for BV
| Parameter | Nugent Score | Molecular Diagnostics (e.g., PCR, NAATs) |
|---|---|---|
| Principle | Microscopic evaluation of bacterial morphotypes on Gram stain [27]. | Nucleic acid detection of specific BV-related bacteria and lactobacilli [29] [4]. |
| Key Targets | Lactobacillus, Gardnerella/Bacteroides, Mobiluncus morphotypes [27]. | Species-specific markers (e.g., L. crispatus, G. vaginalis, F. vaginae, BVAB2) [29] [4]. |
| Turnaround Time | ~30-60 minutes after specimen processing. | Several hours to days, depending on the platform [1]. |
| Subjectivity | High, requires trained, experienced personnel [27] [4]. | Low, automated or software-based interpretation [1]. |
| Sensitivity (vs. Nugent) | Reference Standard | Higher: Detects residual molecular BV missed by Nugent; one study showed 25% (5/20) of Nugent-cured cases still positive by PCR criteria [29]. |
| Specificity (vs. Nugent) | Reference Standard | High: Specificity reported from 85.8% to 98.5% for validated panels [23] [4]. |
| Advantages in Research | Established gold standard; low cost; visual assessment of flora shift. | High reproducibility; identifies specific species; enables study of microbiome dynamics beyond BV diagnosis [29] [1]. |
| Limitations in Research | Cannot identify specific bacterial species; misses "molecular BV" [29] [3]. | Higher cost; complex result interpretation; potential detection of non-pathogenic colonization [1]. |
Recent research underscores a critical limitation of the Nugent score: its inability to detect "molecular BV." A 2025 study found that after metronidazole treatment, 80% of women with classical BV (Nugent ≥7) were cured based on Nugent score (0-3), yet 25% of these "cured" cases still met the molecular BV criteria by a real-time PCR test (Flora select). This residual dysbiosis, characterized by low relative dominance of Lactobacillus and the presence of BV-associated bacteria, highlights the enhanced sensitivity of molecular methods [29].
Furthermore, molecular tests can simultaneously detect other relevant pathogens. The same study reported a 42.9% increase in the detection of Ureaplasma species post-treatment in women with initial classical BV, organisms linked to adverse pregnancy outcomes but not identified by Nugent scoring [29]. The diagnostic performance of molecular tests is robust. One study reported that a combination of L. crispatus and G. vaginalis quantification achieved a sensitivity of 99.29% and specificity of 97.79% for BV diagnosis [4].
Diagnostic Workflow and Research Context
The following diagram illustrates the procedural workflow for the Nugent score and its relationship with molecular diagnostic methods in a research setting.
The Nugent score remains a foundational, cost-effective tool for grading vaginal flora in BV research, providing a standardized framework that has enabled decades of epidemiological and clinical studies. Its utility is evident in establishing the initial link between BV and adverse health outcomes. However, the emergence of molecular diagnostics has revealed significant limitations in the Nugent score's sensitivity, particularly its failure to identify residual molecular BV after treatment and its inability to detect specific pathogens like Ureaplasma. Modern research increasingly leverages molecular tests for their superior sensitivity, specificity, objectivity, and ability to provide a more nuanced view of the vaginal microbiome. A contemporary research protocol for BV should consider the Nugent score as a valuable histological benchmark, while integrating molecular diagnostics as the advanced tool for comprehensive microbial analysis and understanding the full spectrum of vaginal dysbiosis.
The diagnosis of Bacterial Vaginosis (BV) has long relied on conventional methods like the Nugent score, a microscopic grading system of Gram-stained vaginal smears. While this has been the historical gold standard, it suffers from subjectivity, personnel-intensive requirements, and limited accuracy [30] [31]. Within the broader thesis of validating molecular diagnostics against the Nugent score for BV research, multiplex quantitative PCR (qPCR) has emerged as a powerful, objective alternative. This technology enables the precise detection and quantification of key bacterial species that define vaginal health and dysbiosis, transforming our approach to microbial analysis [32] [30]. This guide objectively compares the performance of multiplex qPCR platforms with traditional methods and details the key targets and analytical considerations for its application in BV research.
Key Bacterial Targets in the Vaginal Microbiome
Multiplex qPCR assays for BV focus on a curated panel of bacterial targets that accurately differentiate between healthy and dysbiotic states. The selection is based on decades of research into the vaginal microbiome, which has identified specific lactobacilli as protective and a consortium of anaerobic bacteria as indicative of BV [30] [31].
The primary targets can be categorized as follows:
Protective Lactobacillus Species: A healthy vaginal environment is typically dominated by specific lactobacilli that produce lactic acid, maintaining a low pH and inhibiting pathogen growth.
- Lactobacillus crispatus: Strongly associated with vaginal health and stability (CST I) [30] [31].
- Lactobacillus iners: Often found in both healthy and transitional microbiota (CST III); its dominance is considered less stable than L. crispatus [32] [30].
- Lactobacillus gasseri and Lactobacillus jensenii: Also associated with healthy states but are less commonly dominant [30].
BV-Associated Bacteria (BVAB): The increased abundance of a group of anaerobic bacteria is characteristic of BV (CST IV).
- Gardnerella vaginalis and Atopobium vaginae: Two of the most consistently detected and abundant species in BV [32] [30] [31].
- Megasphaera spp. (Type 1 and 2): Frequently identified in BV and often included in molecular assays [32] [30] [31].
- Prevotella bivia and Mycoplasma hominis: Other common pathogens that are significantly increased in BV [30].
The diagnostic power of multiplex qPCR often involves calculating a "Lactobacillus-index" (comparing the loads of L. crispatus to L. iners) or determining the dominant Community State Type (CST) based on the relative abundance of these key targets [32] [31].
Comparative Performance: Multiplex qPCR vs. Nugent Score
Extensive validation studies have demonstrated the superior performance of multiplex qPCR compared to the conventional Nugent score. The table below summarizes key quantitative performance data from recent studies.
Table 1: Diagnostic Performance of Multiplex qPCR for Bacterial Vaginosis
| Study / Assay Description | Sensitivity (%) | Specificity (%) | Positive Predictive Value (PPV) | Negative Predictive Value (NPV) | Area Under Curve (AUC) | Reference Standard |
|---|---|---|---|---|---|---|
| 5-plex qPCR (L. crispatus, L. iners, G. vaginalis, A. vaginae, Megasphaera1) | 92.0 | 96.0 | 94.0 | 95.0 | Not Reported | Nugent Score [32] |
| 15-plex BVLaB assay (4 Lactobacillus spp., 11 BVAB) with SVM model | 90.4 | 96.1 | Not Reported | Not Reported | 0.969 | Nugent Score & Clinical Exam [30] |
| 5-plex qPCR for CST I (L. crispatus-dominated) | 100.0 | 90.0 | 80.0 | 100.0 | 0.967 | 16S rDNA Sequencing [31] |
| 5-plex qPCR for molecular BV (CST IV, BVAB-dominated) | Not Reported | Not Reported | Not Reported | Not Reported | 0.950 | 16S rDNA Sequencing [31] |
The data consistently shows that multiplex qPCR exhibits high sensitivity and specificity. A pivotal advantage is its ability to accurately classify intermediate cases (Nugent score 4-6), a group that is challenging to diagnose with traditional microscopy [30]. Furthermore, the high concordance (e.g., 90.5%) with 16S rDNA sequencing, the gold standard for microbiome profiling, solidifies its position as a highly accurate molecular tool [31].
Experimental Protocols for Assay Validation
For researchers aiming to implement or validate a multiplex qPCR assay for BV, a standard workflow and rigorous protocol are essential. The following methodology, compiled from several studies, provides a reliable framework.
Sample Collection and Nucleic Acid Extraction
- Sample Collection: Vaginal secretion samples are collected using sterile swabs (e.g., multi-collect specimen collection kits) from the posterior fornix or vaginal vault [32] [30].
- Storage: Swabs should be stored in appropriate transport media and can be frozen at -80°C until processing [33] [30].
- Nucleic Acid Extraction: DNA is extracted from swabs using automated systems (e.g., Abbott M2000sp, QIAcube with QIAamp Viral RNA Kits) or manual kits (e.g., Magen HiPure Bacterial DNA kits, Upure Microbial DNA Kit) following the manufacturer's protocols [33] [32] [30]. The inclusion of a human gene target (e.g., β-globulin, β-actin) is critical as an internal control for sample adequacy and DNA quality [32] [30].
Primer and Probe Design
- Target Selection: Primers and TaqMan hydrolysis probes are designed for specific gene targets of interest (e.g., lytA for S. pneumoniae, iucD for A. baumannii in respiratory panels; 16S rRNA gene regions for vaginal taxa) [33] [32] [30].
- Bioinformatic Validation: The specificity of primer-probe sets must be checked in silico using tools like Primer-BLAST against genomic databases [32] [30].
- Empirical Validation: Assay specificity should be further tested against a panel of standard strains for the intended targets and closely related species to ensure no cross-reactivity [32].
- Multiplexing Optimization: For multiplex assays, primers are designed to yield amplicons with distinct melting temperatures (Tm), typically differing by more than 1°C, to allow for clear differentiation in melting curve analysis if using dye-based methods [33]. Fluorophores attached to probes (e.g., FAM, ROX, HEX, Cy5) must have non-overlapping emission spectra [32].
qPCR Reaction and Data Analysis
- Reaction Setup: A typical 20-25 μL reaction volume contains PCR buffer, dNTPs, Taq DNA polymerase, primer mix, and DNA template [33] [32]. The use of sensitive dyes like EvaGreen or hydrolysis probes is common.
- Thermal Cycling: Amplification is performed on a real-time PCR instrument (e.g., Roche LightCycler 480, Bio-Rad CFX96, Corbett RotorGene 6000). A standard cycling program includes:
- Data Interpretation: Results are analyzed using the instrument's software. For BV diagnosis, quantification cycle (Cq) values are used to calculate semi-quantitative loads of each target. A diagnostic outcome (e.g., BV Positive/Negative) is determined by algorithmic interpretation of these loads, such as the Lactobacillus-index or machine learning models [32] [30].
Figure 1: Multiplex qPCR Workflow for BV Diagnosis. This diagram outlines the key steps from sample collection to diagnostic result.
The Scientist's Toolkit: Essential Research Reagents and Platforms
Successful implementation of multiplex qPCR relies on a suite of reliable reagents and instruments. The following table details key solutions for building a robust research workflow.
Table 2: Research Reagent Solutions for Multiplex qPCR
| Item | Function | Examples & Notes |
|---|---|---|
| Nucleic Acid Extraction Kits | Isolate microbial DNA from complex clinical samples. | MagNA Pure 96 (Roche), QIAamp (QIAGEN), HiPure (Magen), Upure (BioKeystone). Automated systems ensure high throughput and reproducibility [32] [30] [34]. |
| qPCR Master Mixes | Provide optimized buffer, enzymes, and dNTPs for efficient amplification. | QuantiTect Multiplex (QIAGEN), kits from TaqMan or Bio-Rad. Dyes like EvaGreen offer a cost-effective alternative to probes for melting curve analysis [33] [32]. |
| Primers & Probes | Target-specific oligonucleotides for amplification and detection. | Designed in-house and validated [32] [30] or pre-optimized commercial assays (e.g., PrimePCR from Bio-Rad) to reduce optimization time [35]. |
| Internal Controls | Monitor sample collection, extraction, and amplification efficiency. | Primers/probes for human genes (β-actin, β-globulin) to confirm presence of human cells and validate DNA quality [32] [30]. |
| Real-time PCR Instruments | Perform thermal cycling and detect fluorescence signals in real-time. | Roche LightCycler 480, Bio-Rad CFX96/CFX Opus, Qiagen Rotor-Gene Q. Platform choice depends on throughput, multiplexing capacity, and detection chemistry [33] [32] [34]. |
Figure 2: Conceptual Shift from Nugent Score to Molecular Diagnosis. This diagram contrasts the limitations of the traditional method with the key advantages of multiplex qPCR.
The validation of multiplex qPCR against the Nugent score firmly establishes molecular diagnostics as a superior paradigm for BV research. The technology's objectivity, high throughput, and exceptional accuracy in identifying key bacterial targets provide researchers and drug development professionals with a powerful tool. By offering precise quantification of specific vaginal microbiota, multiplex qPCR enables a more nuanced understanding of BV pathogenesis, paves the way for the development of targeted therapies, and ultimately supports improved diagnostic and therapeutic strategies in women's health.
Bacterial vaginosis (BV) research and diagnostics are undergoing a fundamental transformation, moving from microscopy-based techniques to sophisticated molecular approaches. The traditional gold standards, particularly the Nugent score, have provided decades of service but possess inherent limitations including subjectivity, an inability to identify specific bacterial taxa beyond morphological types, and poor representation of the complex polymicrobial nature of BV [36] [8]. This article compares these established methods with emerging molecular techniques—16S rRNA gene sequencing and Fluorescence In Situ Hybridization (FISH)—framed within the critical thesis of validating the Nugent score against modern molecular diagnostics for advanced BV research. For researchers and drug development professionals, understanding the performance characteristics, experimental protocols, and applications of these tools is essential for driving the next wave of discovery in vaginal health.
Traditional vs. Molecular Diagnostic Methods: A Technical Comparison
The diagnosis of BV has historically relied on methods that assess the consequences of microbial dysbiosis rather than directly identifying the causative agents.
The Established Gold Standards
The Nugent score is a microscopic evaluation of a Gram-stained vaginal smear that quantifies bacterial morphotypes. It scores the presence of large Gram-positive rods (Lactobacillus morphotypes; scored 0-4), small Gram-variable rods (Gardnerella morphotypes; scored 0-4), and curved Gram-variable rods (Mobiluncus morphotypes; scored 0-2), resulting in a total score of 0-10. A score of 0-3 is considered normal, 4-6 intermediate, and 7-10 diagnostic of BV [36]. The Amsel criteria, a clinical diagnostic method, requires at least three of the following four signs: homogeneous thin discharge, vaginal pH >4.5, a positive amine ("whiff") test, and the presence of clue cells on wet mount microscopy [36] [8]. While these methods are cost-effective and established, their limitations are significant. The Nugent score is time-consuming, subjective, and does not identify specific bacterial species, while the Amsel criteria can be insensitive (37-70%), though highly specific (94-99%) [36].
Emerging Molecular Techniques
Molecular techniques offer a direct window into the vaginal microbiome by targeting bacterial genetic material:
- 16S rRNA Gene Sequencing: This technique utilizes the hypervariable regions of the bacterial 16S rRNA gene to identify and quantify the taxonomic composition of the vaginal microbiota. It can be applied to either the total DNA pool (reflecting the presence of bacteria) or the RNA pool (reflecting metabolically active communities) [37] [38].
- Fluorescence In Situ Hybridization (FISH): This method uses fluorescently labeled probes that bind to specific nucleic acid sequences within intact bacterial cells, allowing for microscopic visualization and quantification. A advanced variant, Peptide Nucleic Acid FISH (PNA-FISH), uses synthetic DNA mimics with a neutral backbone for higher specificity and stronger binding, proving particularly effective for detecting BV-associated pathogens like Gardnerella spp. and Fannyhessea vaginae within biofilms [39].
Table 1: Comparison of BV Diagnostic Methods
| Method | Key Principle | Sensitivity | Specificity | Key Advantages | Key Limitations |
|---|---|---|---|---|---|
| Amsel Criteria | Clinical criteria (e.g., clue cells, pH, whiff test) | 37-70% [36] | 94-99% [36] | Point-of-care potential; identifies symptomatic BV | Low sensitivity; subjective |
| Nugent Score | Gram-stain microscopy scoring | 89% (vs. Amsel) [8] | 83% (vs. Amsel) [8] | Unbiased; cost-effective; established gold standard | Subjective; time-consuming; limited taxonomic resolution |
| 16S rRNA Sequencing | High-throughput sequencing of 16S rRNA gene | ~95% (vs. clinical methods) [8] | N/A | Comprehensive community profile; discovers novel taxa | Expensive; complex data analysis; result delay |
| FISH/PNA-FISH | Fluorescent probes target specific rRNA | 84.6% (vs. Nugent) [8] | 97.6-100% (vs. Nugent) [8] | Visualizes spatial structure/biofilms; high specificity | Requires specialized equipment and expertise |
| Multiplex PCR (NAAT) | Multiplexed nucleic acid amplification | 91-97% [8] | 77-91% [8] | High throughput; automated; commercially available | Limited to pre-selected targets |
The following workflow illustrates the fundamental difference in the procedural pipeline between traditional and molecular methods, highlighting the integrated and targeted nature of molecular approaches.
Experimental Data and Performance Comparison
Molecular methods demonstrate superior performance in head-to-head validation studies against traditional methods. When 16S rRNA sequencing (V5-V7 region) was compared to Nugent scoring, it confirmed distinct microbial clusters in BV-positive women, characterized by high diversity and dominance of anaerobes like Gardnerella vaginalis, Lachnospiraceae, and Leptotrichiaceae [38]. The Gini-Simpson index of species diversity and the relative abundance of Lactobacillus species were identified as robust, consistent molecular indicators for BV [38].
The synergy of combining techniques is particularly powerful. A study comparing MICRO-CARD-FISH with 16S rDNA and 16S rRNA clone libraries for bacterioplankton demonstrated that while both techniques broadly agreed on community structure, 16S rRNA-based analysis better represented the metabolically active fraction of the community [37]. This highlights the importance of technique selection based on the research question—whether the goal is to catalog all present taxa or to identify the active contributors to the dysbiotic state.
Table 2: Quantitative Performance of Molecular Techniques in BV Research
| Technique | Targets | Key Performance Metrics | Research Applications |
|---|---|---|---|
| 16S rRNA Sequencing (V5-V7 region) | Full bacterial community; Hypervariable regions | Identified 3 distinct BV clusters; Strong correlation with Nugent score and Lactobacillus abundance [38] | Microbiome stratification; Biomarker discovery; Dynamics of recurrence |
| PNA-FISH | Fannyhessea vaginae, Gardnerella spp. | Excellent sensitivity/specificity for F. vaginae; Enables visualization of multi-species biofilms [39] | Biofilm architecture; Polymicrobial interactions; Pathogen co-localization |
| Multiplex PCR (NAAT) | G. vaginalis, F. vaginae, BVAB2, etc. | Sensitivity: 90.5-96.7%; Specificity: 85.8-95% (vs. Amsel/Nugent) [8] | High-throughput clinical screening; Targeted pathogen detection |
Detailed Experimental Protocols
To ensure experimental reproducibility, here are detailed protocols for key molecular techniques as applied in recent BV research.
16S rRNA Gene Amplicon Sequencing Protocol
This protocol is adapted from a study that sequenced the V5-V7 region to validate BV indicators [38].
- DNA Extraction: Resuspend vaginal swab samples in Agowa lysis buffer BL with zirconium beads and phenol. Lyse cells using a BeadBeater for 2 minutes. Isolate DNA from the aqueous phase using binding beads.
- Quality Assessment: Quantify the bacterial DNA load using a universal quantitative PCR (qPCR) assay for the 16S rRNA gene.
- Library Preparation: Generate a 16S rRNA gene amplicon library spanning the V5-V7 hypervariable regions using region-specific primers.
- Sequencing: Perform sequencing on a 454 GS-FLX-Titanium Sequencer (or a modern Illumina platform with an equivalent workflow).
- Bioinformatic Analysis:
- Process FASTA sequences and quality scores using a pipeline like the Mothur software platform.
- De-noise sequences using a pseudo-single linkage algorithm (e.g.,
pre.clustercommand). - Detect and remove chimeric sequences (e.g.,
chimera.uchimecommand). - Align sequences and perform taxonomic classification by comparing to a curated database (e.g., RDP database). A homology score of 1 is required for species-level identification.
PNA-FISH Probe Design and Validation Protocol
This protocol outlines the development and application of a novel PNA-FISH probe for Fannyhessea vaginae [39].
- Probe Design:
- Select high-quality 16S and 23S rRNA sequences of F. vaginae and related species from the SILVA database.
- Align sequences using the Clustal Omega tool to identify unique target regions specific to F. vaginae with one or more mismatches in non-target species.
- Select a candidate probe based on high theoretical sensitivity/specificity, GC content (40-60%), and melting temperature (>50°C). Synthesize the PNA probe with an N-terminal fluorescent label (e.g., Alexa Fluor 488).
- Hybridization:
- Prepare pure cultures or clinical samples on glass slides, fix with ethanol, and allow to air-dry.
- Cover the sample with a hybridization solution containing the PNA probe (e.g., 100 nM) and incubate in a dark, humidified chamber at an optimized temperature (e.g., 80°C for 30 minutes).
- Washing and Visualization:
- Immerse the slides in a pre-warmed washing solution to remove non-specific binding.
- Rinse the slides with cold water and air-dry in the dark.
- Mount the slides and observe under an epifluorescence microscope with appropriate filter sets.
- Multiplexing: For co-localization studies, include a second, differently labeled PNA probe (e.g., a probe targeting Gardnerella spp. labeled with Alexa Fluor 594) in the same hybridization reaction.
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of these molecular techniques requires specific, high-quality reagents.
Table 3: Essential Research Reagents for Molecular BV Diagnostics
| Reagent / Solution | Critical Function | Application Notes |
|---|---|---|
| Agowa Lysis Buffer | Efficiently breaks down bacterial cell walls for DNA release. | Critical for robust DNA yield from tough Gram-positive BVAB in vaginal samples [38]. |
| PNA Probes (e.g., vs. F. vaginae, Gardnerella) | Synthetic DNA mimics that bind to specific 16S/23S rRNA sequences with high affinity. | Neutral backbone reduces electrostatic repulsion, allowing faster, more specific hybridization than DNA probes [39]. |
| SILVA or RDP Database | Curated 16S rRNA sequence databases for taxonomic classification. | Essential for accurate bioinformatic analysis; quality and curation directly impact results [40] [38]. |
| Hypervariable Region Primers (e.g., V3-V4, V4-V5, V5-V7) | PCR primers that amplify specific variable regions of the 16S rRNA gene. | Choice of region (e.g., V4-V5 for combined Bacteria/Archaea) influences community representation [41]. |
| Fluorescent Labels (e.g., Alexa Fluor 488, 594) | Conjugated to PNA probes for microscopic detection. | Enable multiplexing; must be photostable and compatible with standard microscope filter sets [39]. |
The validation of the Nugent score against molecular diagnostics underscores a critical evolution in BV research. While the Nugent score remains a useful, cost-effective tool for defining BV status in broad terms, 16S rRNA sequencing and FISH provide a deeper, more resolutive understanding of the vaginal microbiome's composition, structure, and function. The future of BV research and advanced diagnostics lies in leveraging the strengths of these molecular tools—using 16S sequencing for comprehensive community profiling and discovery, and employing FISH to unravel the spatial polymicrobial interactions within biofilms that underpin BV pathogenesis and recurrence. For researchers and drug developers, embracing these techniques is paramount for developing novel, targeted therapeutics and personalized diagnostic strategies.
The diagnosis of bacterial vaginosis (BV) is undergoing a substantial transformation, moving from traditional microscopy-based methods toward sophisticated molecular techniques. For decades, the Nugent score—a Gram-staining scoring system that evaluates bacterial morphotypes—has served as a laboratory gold standard for BV research and diagnosis [29] [42]. While this method has high concordance with Amsel criteria when read by experienced technologists, it is labor-intensive, time-consuming, and somewhat subjective, as it relies on morphological identification of bacteria rather than specific pathogen detection [29] [4]. These limitations have catalyzed the development and adoption of molecular diagnostic panels that use nucleic acid amplification to provide precise, objective identification of BV-associated microorganisms.
This guide provides an objective comparison of three key approaches in the modern BV diagnostic landscape: the commercially available BD Max Vaginal Panel and Hologic Aptima BV assays, and Laboratory-Developed Tests (LDTs) such as the Flora select (FS) test. Framed within the broader thesis of validating molecular diagnostics against traditional Nugent scoring, we examine their performance characteristics, methodological foundations, and practical applications in research and clinical settings. Molecular diagnostics for BV utilize advanced techniques like polymerase chain reaction (PCR) to detect specific bacterial DNA sequences, offering a more accurate and reliable alternative to traditional methods [43]. The growing preference for these molecular assays in both research and clinical practice is driven by their superior accuracy, which allows for more precise patient management and potentially improved outcomes [43] [44].
Performance Comparison of Major Diagnostic Platforms
Direct comparison of performance metrics across diagnostic platforms reveals distinct strengths and applications for each technology. The table below summarizes key characteristics and performance data for the BD Max, Aptima BV, and representative LDTs.
Table 1: Performance Comparison of Commercial BV Diagnostic Panels and Laboratory-Developed Tests
| Diagnostic Platform | Technology | Key Analytes | Performance vs. Nugent/Gram Stain | Regulatory Status | Sample Workflow |
|---|---|---|---|---|---|
| BD Max Vaginal Panel | Real-time PCR | Gardnerella vaginalis, Atopobium vaginae, Lactobacillus spp., and others | Demonstrates superior performance in identifying BV-positive cases missed by Nugent scoring [43] | FDA 510(k) cleared (March 2023) [43] | High-throughput, automated testing on BD COR System [43] |
| Hologic Aptima BV | Transcription-Mediated Amplification (TMA) | RNA from BV-associated bacteria | Excellent Positive Percent Agreement (PPA) and Negative Percent Agreement (NPA) versus consensus methods [42] | FDA-cleared | Utilizes Aptima Multitest swab collection system [42] |
| Flora select (LDT) | SYBR Green-based real-time PCR | Relative abundance of Lactobacillus; Gardnerella, Prevotella, Atopobium, Streptococcus, Ureaplasma, Mycoplasma | Detected residual molecular BV in 25% of cases cured per Nugent score [29] | Laboratory-developed | Requires DNA extraction followed by PCR amplification on CFX96 system [29] |
A 2024 study highlighted that real-time PCR assays for key BV-associated species demonstrated excellent diagnostic accuracy compared to Nugent scoring, correctly identifying a significant number of BV-positive cases that Nugent scoring had classified as intermediate [43]. This enhanced detection capability is particularly valuable for research settings where precise stratification of study participants is critical. The Aptima BV assay has demonstrated particularly strong performance characteristics, with one retrospective study of 664 specimens reporting excellent positive and negative percent agreement compared to conventional laboratory-based testing methods [42].
Laboratory-developed tests like Flora select offer unique research advantages through their customizable panels. In a 2025 multicenter prospective study, the FS test demonstrated particular value in detecting "residual molecular BV" after conventional treatment, identifying that 5 (25%) of 20 cases deemed cured according to Nugent scores (0-3) still met molecular BV criteria [29]. This capacity to detect persistent dysbiosis despite microscopically confirmed resolution highlights the enhanced sensitivity of molecular methods and their potential research applications for understanding treatment failure and BV recurrence.
Detailed Experimental Protocols and Methodologies
Flora select Laboratory-Developed Test Protocol
The Flora select (FS) test employs a SYBR Green-based real-time PCR methodology to provide comprehensive vaginal microbiome assessment [29]. The detailed experimental protocol includes:
Specimen Collection and Storage: Two vaginal swabs are collected simultaneously for comparative analysis. The first swab is used for Nugent scoring via Gram staining, while the second swab is placed directly into a Copan eNAT collection tube and stored at room temperature until DNA extraction, which must be performed within 4 weeks according to the manufacturer's protocol [29].
DNA Extraction and Purification: Samples undergo pretreatment with proteinase K (≥600 U/mL) and lysozyme solution (1.5 mg/mL) for cell lysis. Genomic DNA is then extracted using the MagNA Pure 24 system (Pathogen 1000 hp 3.2 software/protocol, Roche Diagnostics GmbH) [29].
PCR Amplification and Analysis: The amplification reactions are performed on a CFX96 C1000 Touch Real-Time System (CFX Maestro, Bio-Rad Laboratories) using a total volume of 20 μL containing THUNDERBIRD Next SYBR qPCR Mix (TOYOBO Co., Ltd.), forward and reverse primer sets (0.5 μM concentration), and the extracted bacterial DNA. Each plate includes both no-template and positive controls [29].
Data Interpretation: The relative absolute abundance of Lactobacillus species is calculated by the ratio of the amount of Lactobacillus to the total amount of bacterial species. The cycle threshold (Ct) of each sample is compared with that of a standard curve made by diluting the genomic DNA of Lactobacillus crispatus. The abundance of Lactobacillus is classified into four categories based on relative dominance rates: high (≥80%), medium (50%≤, <80%), low (0.1≤, <50%), and no detection (<0.1%) [29].
Table 2: Research Reagent Solutions for Molecular BV Diagnostics
| Reagent/Equipment | Specific Function | Research Application |
|---|---|---|
| Copan eNAT Collection Tube | Stabilizes nucleic acids in vaginal swab samples | Maintains sample integrity during transport and storage for LDTs [29] |
| THUNDERBIRD Next SYBR qPCR Mix | Fluorescent DNA binding dye for real-time PCR detection | Enables quantification of bacterial loads in LDTs like Flora select [29] |
| MagNA Pure 24 System | Automated nucleic acid extraction | Standardizes DNA purification process in high-volume research settings [29] |
| Proteinase K & Lysozyme | Enzymatic cell lysis for DNA release | Facilitates breakdown of bacterial cell walls for comprehensive DNA recovery [29] |
| CFX96 Touch Real-Time PCR System | Thermal cycling with fluorescence detection | Provides platform for quantitative analysis of bacterial biomarkers in research [29] |
Nugent Score Reference Method Protocol
As the comparative reference method, the Nugent scoring protocol must be rigorously implemented:
Slide Preparation and Staining: Vaginal fluid samples are smeared onto glass slides and Gram-stained using either the Bartholomew and Mittwer methods (Muto Pure Chemicals Co., Ltd.) or the Favor method (Shimadzu Diagnostics Co.) for Gram staining [29].
Microscopic Evaluation: Stained slides are examined under oil immersion at 1000× magnification. The scoring system evaluates three bacterial morphotypes: Lactobacillus morphotypes (scored 0-4, with 0 indicating the lowest amount), small Gram-variable rods (Gardnerella vaginalis and Bacteroides morphotypes, scored 0-4, with 4 indicating the highest amount), and curved Gram-variable rods (Mobiluncus morphotypes, scored 0-2, with 2 indicating the highest amount) [29] [42].
Scoring and Interpretation: The scores for each morphotype are summed to give a total Nugent score ranging from 0-10. Scores of 0-3 are considered BV-negative, 4-6 intermediate, and ≥7 BV-positive [29] [42]. This method requires highly trained and experienced staff to ensure accurate morphological identification and scoring [4].
Molecular Targets and Diagnostic Criteria
The diagnostic accuracy of molecular panels depends on the careful selection of microbial targets that best represent the dysbiotic state characteristic of BV. Research has investigated various combinations of molecular markers to optimize diagnostic performance.
Table 3: Key Molecular Diagnostic Markers for Bacterial Vaginosis
| Microbial Target | Role in Vaginal Microbiome | Detection Rate in BV-Positive Cases | Diagnostic Utility |
|---|---|---|---|
| Lactobacillus crispatus | Primary producer of lactic acid and H₂O₂; maintains healthy vaginal environment | Higher detection rate in healthy controls [4] | Loss associated with BV; protective factor |
| Gardnerella vaginalis | Anaerobic bacterium frequently increased in BV | 95.90% sensitivity for BV diagnosis [4] | Primary BV-associated pathogen |
| Fannyhessea vaginae (formerly Atopobium vaginae) | Anaerobic bacterium strongly associated with BV | 82.79% sensitivity for BV diagnosis [4] | Secondary BV-associated pathogen |
| BVAB-2 (Oscillospiraceae bacterium strain CHIC02) | Bacterial vaginosis-associated bacterium | 72.13% sensitivity for BV diagnosis [4] | Supportive diagnostic marker |
| Ureaplasma species | Associated with vaginal dysbiosis and adverse pregnancy outcomes | Detected in 45% of Nugent-cured cases with residual molecular BV [29] | Marker for treatment-resistant dysbiosis |
Research has demonstrated that combinations of markers provide superior diagnostic performance compared to single biomarkers. A 2025 study found that combining L. crispatus with G. vaginalis achieved 99.29% sensitivity and 97.79% specificity, while L. crispatus with F. vaginae showed 98.86% sensitivity and 98.72% specificity [4]. These combinations better reflect the ecological concept of BV as an imbalance of the vaginal microbiome rather than simply the presence or absence of individual bacterial species.
The Flora select test implements this combinatorial approach through defined diagnostic criteria. The BV-FS A criterion requires relative dominance rates of Lactobacillus ≤ low (<50%), together with a positive test for Gardnerella, Prevotella, or Atopobium species. The BV-FS B criterion requires Lactobacillus at medium levels (50%≤, <80%), together with positive tests for Gardnerella plus either Prevotella or Atopobium species [29]. This nuanced approach allows for detection of dysbiosis even in cases where Lactobacillus species have not been completely displaced.
Research Applications and Clinical Validation
Molecular diagnostic panels offer significant advantages in research settings, particularly for investigating BV recurrence, treatment efficacy, and associations with adverse health outcomes. The enhanced sensitivity of molecular methods allows researchers to detect residual dysbiosis that may predict clinical recurrence.
A 2025 multicenter prospective study demonstrated this application by using the Flora select test to assess BV treatment effectiveness. The research found that while metronidazole treatment resulted in the improvement of 88.0% of classical BV cases based on Nugent score, molecular assessment revealed that a considerable proportion still harbored molecular BV detected by FS [29]. Specifically, although the Nugent scoring system indicated that 80.0% of women with classical BV were sufficiently cured as BV-negative (Nugent scores 0-3), 25% of these cured cases still met the BV-FS A/B criteria for molecular BV [29]. This residual dysbiosis, undetectable by conventional microscopy, may represent a biological basis for the high recurrence rates observed in BV.
Furthermore, molecular panels can detect important pathogens that are missed by traditional methods. The FS test identified Ureaplasma species in 45% of cases considered cured by Nugent criteria, and positive rates of Ureaplasma species in women with classical BV increased by 42.9% following metronidazole treatment [29]. This suggests that certain opportunistic pathogens may proliferate following antibiotic therapy, potentially contributing to recurrence or other complications.
The research utility of these molecular panels extends to understanding BV's role in adverse obstetric outcomes. Preterm birth is a major cause of perinatal mortality and morbidity, and its risk is increased by BV during pregnancy [29]. Molecular diagnostics offer the potential to identify high-risk women more accurately than traditional methods, with one randomized clinical trial demonstrating that real-time PCR tests and treatment for BV based on the quantification of Gardnerella and Atopobium significantly reduced preterm birth rates [29].
Visualizing Molecular BV Testing Workflows
The transition from traditional to molecular diagnostics represents a fundamental shift in laboratory approach. The following diagram illustrates the key decision points and methodological pathways in molecular BV diagnosis.
Molecular BV Diagnostic Decision Pathway
The relationship between diagnostic methods and their corresponding targets can be visualized through their complementary roles in BV assessment, as shown in the following diagram.
BV Diagnostic Methods and Their Applications
The validation of molecular diagnostic panels against traditional Nugent scoring represents a significant advancement in BV research methodology. While the Nugent score has provided decades of valuable service as a standardized microscopy-based approach, molecular panels offer enhanced sensitivity, objectivity, and the ability to detect clinically significant dysbiosis that would otherwise be missed. The BD Max Vaginal Panel, Hologic Aptima BV, and laboratory-developed tests like Flora select each bring distinct strengths to research settings, from regulatory clearance and standardization to customizability and comprehensive microbiome assessment.
The research implications of this technological transition are substantial. Molecular panels enable more precise stratification of research participants, detection of residual dysbiosis following treatment, and identification of specific microbial patterns associated with adverse outcomes like preterm birth. As these technologies continue to evolve and become more accessible, they promise to deepen our understanding of BV pathogenesis, recurrence, and optimal treatment strategies. For researchers designing studies involving BV diagnosis, molecular panels now offer a robust alternative to traditional methods, particularly when investigating treatment efficacy, microbial dynamics, or subtle microbiome alterations associated with clinical outcomes.
Navigating Diagnostic Challenges: Limitations and Pitfalls of Current BV Tests
Bacterial vaginosis (BV) represents one of the most prevalent vaginal conditions affecting women of reproductive age worldwide, yet its diagnosis remains challenging due to the complex nature of vaginal microbiota. The Nugent scoring system, developed in 1991, has served as the reference standard for BV diagnosis in research settings for decades. This laboratory-based method assesses Gram-stained vaginal smears through microscopic evaluation of bacterial morphotypes, generating a score from 0 to 10. While scores 0-3 indicate healthy vaginal flora and scores 7-10 confirm BV, the intermediate range of 4-6 represents a diagnostic gray zone with significant clinical ambiguity. This "intermediate flora" category neither qualifies as normal vaginal flora nor fulfills the criteria for full BV, creating substantial challenges for both clinical management and research consistency [45]. The fundamental conundrum lies in the fact that this intermediate state may represent a transitional phase between health and disease, a distinct pathological condition, or simply an artifact of imperfect diagnostic categorization [45]. Understanding this ambiguity is crucial for researchers and clinicians aiming to improve BV diagnostics and develop targeted interventions.
The Nugent Score System: Components and Limitations
Technical Framework of Nugent Scoring
The Nugent score is calculated by assessing three bacterial morphotypes on Gram-stained vaginal smears under oil immersion microscopy. Lactobacillus species (large Gram-positive rods) are scored from 0-4, with higher scores indicating decreasing abundance. Gardnerella and Bacteroides morphotypes (small Gram-variable or Gram-negative rods) are scored from 0-4, with higher scores indicating increasing abundance. Curved Gram-variable or Gram-negative rods (Mobiluncus species) are scored from 0-2 based on their presence. The sum of these three components yields the final Nugent score: 0-3 (normal), 4-6 (intermediate), and 7-10 (BV) [45] [4]. This method provides a semi-quantitative assessment of the shift from Lactobacillus-dominated flora to polymicrobial communities characteristic of BV.
Reliability and Reproducibility Concerns
The Nugent score's accuracy is highly dependent on the technical skill and experience of laboratory personnel, leading to significant inter-observer variability. A 2018 study evaluating interobserver reliability found that three independent microbiologists gave concordant results in only 64.03% of smears, with complete disagreement occurring in 4.51% of cases [46]. The agreement between different observer pairs ranged from 59.9% to 66.7%, reflecting only fair to good reliability [46]. This variability stems from challenges in consistently identifying bacterial morphotypes, differences in bacterial density across microscopic fields, and variations in microscope optics [46]. The intermediate scores demonstrated the lowest agreement rates among observers, highlighting the particular subjectivity in interpreting this ambiguous category [46]. These limitations have prompted investigations into automated scoring systems, including deep learning approaches that show promise for standardizing Nugent score interpretation [47].
Table 1: Interobserver Reliability of Nugent Scoring in Vaginal Smear Interpretation
| Assessment Type | Percentage of Smears | Kappa Value Range | Reliability Interpretation |
|---|---|---|---|
| Complete agreement between 3 observers | 64.03% | N/A | N/A |
| Partial agreement (2/3 observers) | 31.63% | 0.4-0.75 | Good to fair agreement |
| Complete disagreement | 4.51% | N/A | N/A |
| Observer pair agreement | 59.9%-66.7% | 0.4-0.75 | Good to fair agreement |
The Intermediate Flora Category: Clinical Ambiguity and Research Challenges
Diagnostic Uncertainty and Clinical Management
The intermediate Nugent score (4-6) represents perhaps the most significant limitation of the scoring system, creating substantial uncertainty in both clinical and research settings. This category functions as a "garbage can" for samples that defy clear classification rather than representing a true transitional state between normal flora and full BV [45]. Contrary to what might be expected, this intermediate group has been associated with potentially more serious complications than classic BV in some studies, including mid-trimester pregnancy loss [45]. The clinical management of women with intermediate scores remains controversial, as they may not respond to standard BV treatments like metronidazole, suggesting different underlying pathology [45]. This ambiguity complicates treatment decisions, research participant selection, and epidemiological studies aiming to define true BV prevalence and associated health risks.
Research Implications and Cohort Definition Challenges
In research settings, the intermediate category creates significant methodological challenges. Studies investigating BV-associated risks must decide whether to exclude, include, or regroup intermediate cases, with each approach potentially yielding different conclusions. The 2024 deep learning study on Nugent score automation highlighted this challenge, noting that a score of 4 may indicate the absence of vaginal flora rather than alteration, suggesting that the traditional three-category system may oversimplify complex microbial states [47]. This has led some researchers to propose four-category classification systems to better capture the biological diversity within the intermediate range [47]. The inability to consistently classify this gray zone represents a fundamental constraint on advancing BV research and developing targeted interventions for different manifestations of vaginal dysbiosis.
Molecular Diagnostics: A New Paradigm for BV Assessment
Molecular Targets and Diagnostic Approaches
Molecular diagnostics for BV have emerged as powerful alternatives to microscopy-based methods, offering improved objectivity, sensitivity, and specificity. These approaches typically utilize quantitative polymerase chain reaction (qPCR) to detect and quantify specific BV-associated bacteria. The most promising molecular markers include Lactobacillus crispatus (protective species), Gardnerella vaginalis, Fannyhessea vaginae (formerly Atopobium vaginae), and BVAB-2 (Oscillospiraceae bacterium strain CHIC02) [4]. Additional markers such as Megasphaera-1 and Megasphaera-2 have been investigated but demonstrate lower detection rates in BV-positive cases (35.25% and 19.67% respectively), limiting their diagnostic utility [4]. The combination of L. crispatus with G. vaginalis has shown exceptional diagnostic performance, with 99.29% sensitivity and 97.79% specificity, outperforming single-marker approaches and traditional Nugent scoring [4].
Comparative Performance: Molecular Methods vs. Nugent Scoring
Recent studies have directly compared the performance of molecular assays with conventional Nugent scoring. The Hologic Panther Aptima BV assay demonstrated 97.5% sensitivity and 96.3% specificity compared to consensus Gram stain and Nugent scoring [1]. For candidiasis and trichomoniasis, the Aptima CV/TV assays showed 100% sensitivity for both conditions, with specificity of 83.5% compared to Gram stain and 96.3% compared to culture for candidiasis, and 100% specificity for trichomoniasis [1]. These molecular assays eliminate the subjectivity of microscopic interpretation and address the problematic intermediate category by providing definitive positive/negative results based on predefined microbial thresholds. However, they introduce new challenges related to cost, technical requirements, and interpretation of multi-organism detection in the context of clinical symptoms.
Table 2: Performance Comparison of BV Diagnostic Methods
| Diagnostic Method | Sensitivity | Specificity | Advantages | Limitations |
|---|---|---|---|---|
| Nugent Score | N/A | N/A | Low cost, minimal equipment | Subjective, intermediate category ambiguity |
| Amsel's Criteria | 37-70% | N/A | Bedside assessment, immediate results | Limited sensitivity, subjective components |
| L. crispatus + G. vaginalis qPCR | 99.29% | 97.79% | High accuracy, objective quantification | Requires specialized equipment, higher cost |
| Hologic Panther Aptima BV | 97.5% | 96.3% | High sensitivity/specificity, automated | Platform-specific, moderate cost |
| Deep Learning Model (1000x) | 89% (4-category) | 89% (4-category) | Reduces observer variability | Requires validation, specialized implementation |
Comparative Methodologies: Experimental Protocols and Workflows
Nugent Score Laboratory Protocol
The standard Nugent scoring protocol begins with collection of vaginal swabs from the posterior fornix. Smears are prepared by rolling the swab on a glass slide, followed by air drying and Gram staining. The stained slides are examined under oil immersion microscopy (1000x magnification) [4]. Technicians systematically assess multiple fields to evaluate the proportions of three bacterial morphotypes: Lactobacillus (large Gram-positive rods), Gardnerella/Bacteroides (small Gram-variable or Gram-negative rods), and Mobiluncus (curved rods) [45]. Each morphotype is assigned a score based on its relative abundance, and the composite Nugent score is calculated. Quality control requires experienced personnel and regular validation between observers to minimize variability. This method typically takes 15-20 minutes per sample when performed by experienced technologists [4].
Molecular Detection Protocol
Molecular diagnosis begins with similar vaginal swab collection, but specimens are immediately stored at -80°C until DNA extraction can be performed [4]. DNA extraction utilizes commercial kits with magnetic bead-based technology, followed by purity and concentration measurement using spectrophotometry [4]. Quantitative PCR assays target specific bacterial genes, with primer sets designed for optimal detection of key BV-associated organisms. The reaction mixture typically includes DNA template, specific primers, probe (if using TaqMan chemistry), and PCR master mix. Amplification conditions follow standard qPCR parameters with annealing temperatures optimized for each primer set. Results are interpreted based on cycle threshold (Ct) values compared to established cut-offs for each target organism, with algorithms combining multiple targets to determine BV status [4].
Diagram Title: Comparative Workflows for BV Diagnostic Methods
Emerging Technologies and Future Directions
Deep Learning and Automated Image Analysis
Recent advances in artificial intelligence offer promising solutions to address the subjectivity of Nugent scoring. A 2024 study developed convolutional neural network (CNN) models to classify vaginal smear images into Nugent categories, achieving 89% accuracy at 1000x magnification in four-group classification (normal, no flora, altered flora, BV) [47]. After optimization with additional images, the advanced model reached 94% accuracy, outperforming the average technician accuracy of 92% [47]. These models demonstrated particularly strong agreement for normal vaginal flora (92%) and BV (100%), with lower but still substantial agreement for altered vaginal flora (91%) [47]. This technology has potential to standardize BV diagnosis, reduce inter-observer variability, and facilitate consistent implementation in resource-limited settings where experienced microscopists are scarce.
Sequencing Technologies and Community State Typing
High-throughput sequencing approaches provide unprecedented resolution for characterizing vaginal microbiota, revealing significant limitations in current BV diagnostic categories. Metataxonomic (16S rRNA gene sequencing), metagenomic (whole genome sequencing), and metatranscriptomic (RNA sequencing) analyses have identified five major community state types (CSTs): CST I (L. crispatus-dominated), CST II (L. gasseri-dominated), CST III (L. iners-dominated), CST V (L. jensenii-dominated), and CST IV (polymicrobial) [22]. However, significant discordance exists between molecular CST assignments and clinical diagnoses, with women characterized as BV-negative by Amsel's criteria and Nugent scoring often assigned to BV-associated CSTs (IIIB and IV) [22]. Furthermore, concordance between different sequencing methods can be as low as 59% for CST assignment between metatranscriptomic and metataxonomic approaches [22], highlighting the methodological challenges in defining BV through microbial composition alone.
The Sexual Transmission Paradigm and Diagnostic Implications
A landmark 2025 randomized controlled trial published in the New England Journal of Medicine demonstrated that treating male partners of women with BV significantly reduced recurrence rates (35% vs. 63% in controls) [48] [49] [50]. This finding fundamentally shifts the conceptualization of BV from a simple dysbiosis to a sexually associated condition, with profound implications for diagnostic approaches. The study utilized a combination of oral metronidazole and topical clindamycin for male partners, highlighting the potential need to expand diagnostic testing beyond women to include sexual partners [50]. This paradigm shift underscores the limitations of current diagnostic methods that focus exclusively on women's vaginal microbiota and may necessitate development of new testing approaches for extra-vaginal sites and male partners.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagents and Materials for BV Diagnostic Studies
| Reagent/Material | Application | Specifications | Research Function |
|---|---|---|---|
| Gram Stain Kit | Nugent scoring | Standardized staining solutions | Differentiates bacterial morphotypes on vaginal smears |
| DNA Extraction Kit | Molecular diagnostics | Magnetic bead-based (e.g., Shengxiang S1006) | Nucleic acid purification from vaginal swabs |
| qPCR Master Mix | Molecular diagnostics | Probe-based chemistry | Amplification of target bacterial genes |
| Species-Specific Primers/Probes | Molecular diagnostics | Targets: L. crispatus, G. vaginalis, F. vaginae | Quantification of specific BV-associated bacteria |
| Digital Slide Scanner | Deep learning applications | 400x/1000x magnification capability | Converts glass slides to digital images for analysis |
| Vaginal Swabs | Sample collection | Nylon fiber tips | Standardized specimen collection from posterior fornix |
The intermediate flora category in Nugent scoring represents a significant limitation in BV diagnosis, creating ambiguity in both clinical management and research applications. While the Nugent score remains valuable for its low cost and accessibility, its subjectivity and problematic intermediate category drive the need for more objective diagnostic methods. Molecular approaches using biomarker combinations like L. crispatus and G. vaginalis demonstrate superior accuracy and eliminate the ambiguous intermediate category, but at higher cost and with greater technical requirements. Emerging technologies including deep learning for automated Nugent scoring and advanced sequencing for community state typing offer promising avenues for standardized, accurate BV assessment. The recent recognition of BV as a sexually associated condition further underscores the need for evolved diagnostic paradigms that extend beyond traditional microscopy of vaginal samples. Future research should focus on developing integrated diagnostic approaches that combine the accessibility of traditional methods with the precision of molecular techniques, while expanding testing strategies to include sexual partners in cases of recurrence.
Operator Dependency and Subjectivity in Microscopic Methods
Bacterial vaginosis (BV) is a common vaginal dysbiosis associated with significant adverse health outcomes, including increased risk for preterm birth and acquisition of sexually transmitted infections [51]. Accurate diagnosis is therefore critical for both clinical management and public health research. For decades, the Nugent score, a microscopic method based on the Gram staining of vaginal fluid, has been considered the gold standard for BV diagnosis in research settings [51] [52]. This guide provides an objective comparison between this established microscopic method and emerging molecular diagnostics, focusing specifically on the limitations introduced by operator dependency and subjectivity. The analysis is framed within the broader thesis of validating robust and reproducible diagnostic protocols for BV research, a matter of paramount importance to scientists and drug development professionals seeking to ensure data integrity and translatability.
Comparative Analysis of Methodological Principles
The fundamental difference between these diagnostic approaches lies in their basic principle: the Nugent score relies on visual interpretation of cellular morphology, whereas molecular methods quantify specific bacterial nucleic acids.
- Nugent Score (Microscopic Method): This is a semi-quantitative scoring system where a stained vaginal smear is examined under a microscope. The technologist morphologically identifies and quantifies three bacterial morphotypes: large Gram-positive rods (Lactobacillus), small Gram-variable rods (Gardnerella vaginalis), and curved Gram-negative rods (Mobiluncus) [52]. A score of 0-10 is generated, with scores 0-3 classified as BV negative, 4-6 as intermediate, and 7-10 as BV positive [51] [52]. The diagnosis hinges entirely on the technician's ability to correctly identify and count these bacterial forms.
- Molecular Diagnostics (e.g., Aptima BV Assay, molBV): These are nucleic acid amplification tests (NAATs) that detect and quantify organisms associated with normal vaginal flora (e.g., Lactobacillus crispatus, L. gasseri, L. jensenii) and those associated with BV (e.g., Gardnerella vaginalis, Fannyhessea vaginae) [52]. The molBV pipeline, for instance, uses 16S rRNA gene sequencing and a classification algorithm to generate a Nugent-like score of 0-10, but based on molecular data rather than visual interpretation [51]. The Aptima BV assay uses a proprietary algorithm to analyze the relative detection of key bacteria and returns a qualitative positive or negative result, eliminating the intermediate category [52].
Quantitative Performance Comparison
The following tables summarize key experimental data comparing the operator dependency and overall performance of microscopic versus molecular methods.
Table 1: Operator Dependency and Reproducibility
| Metric | Nugent Score (Microscopic) | Molecular Methods (e.g., Aptima, molBV) |
|---|---|---|
| Inter-observer Variability | High; 20% of samples were discordant on duplicate examination [52] | Low; automated systems provide objective, reproducible results [52] |
| Diagnostic Classification | Subjective, based on morphological identification and counting [52] | Objective, based on algorithmic analysis of quantitative data [51] [52] |
| Result Categories | Three: Negative, Intermediate, Positive [52] | Two: Negative, Positive (no intermediate) [52] |
Table 2: Overall Diagnostic Performance from Clinical Evaluations
| Performance Measure | Nugent Score | Molecular Methods (vs. Nugent as Reference) |
|---|---|---|
| Sensitivity | Considered the reference, but with inherent variability [52] | 97.5% (Aptima BV assay) [52] |
| Specificity | Considered the reference, but with inherent variability [52] | 96.3% (Aptima BV assay) [52] |
| Generalizability | Limited by technical skill and protocol differences | High; molBV shown to be independent of the 16S rRNA region and generalizable across populations [51] |
Detailed Experimental Protocols for Method Comparison
A rigorous method comparison study is essential for validating a new diagnostic against an existing one. The following protocols detail the key experiments cited in the performance tables.
Protocol for Assessing Nugent Score Reproducibility
This protocol is derived from a 2024 clinical laboratory evaluation [52].
- Objective: To determine the inter-observer variability and subjectivity of the Nugent score microscopic method.
- Materials: Vaginal swab samples, glass slides, Gram stain reagents, microscope.
- Method:
- Gram stains are prepared from vaginal swabs and stained, ideally using an automated staining device.
- Each slide is read independently by two trained laboratory personnel, who are blinded to any previous results.
- Each reader applies the Nugent score, resulting in a categorical diagnosis (Negative, Intermediate, Positive).
- Any discordant categorical results between the first and second reading are resolved by a third independent reader.
- The final consensus result is recorded when at least two of the three categorical scores match.
- Data Analysis: The percentage of samples with discordant results between the first two readers is calculated. A high discordance rate (e.g., the reported 20% [52]) directly evidences operator dependency.
Protocol for a Method Comparison Study
This protocol follows guidelines for method comparison experiments [53] [54] and is exemplified in the Aptima BV assay evaluation [52].
- Objective: To estimate the systematic error (bias) and performance (sensitivity, specificity) of a new molecular method against the comparative Nugent score method.
- Study Design: A minimum of 40 patient samples should be used, though 100 or more are preferable to identify unexpected errors [53] [54]. Samples must be selected to be representative and analyzed within their stability period, ideally within 2 hours of collection [53] [54].
- Materials: Paired vaginal swabs from each participant (one for the Nugent score, one for the molecular test), Gram stain materials, molecular testing platform (e.g., Hologic Panther system).
- Method:
- For each patient sample, perform diagnostic testing using both the standard Nugent score (following the consensus protocol above) and the new molecular method (e.g., Aptima BV or molBV) [52].
- The order of analysis should be randomized to avoid carry-over effects or systematic bias [53].
- Testing should be performed over several days (at least 5) to mimic real-world conditions [53] [54].
- Data Analysis:
- 2x2 Table: Create a contingency table to calculate sensitivity, specificity, positive predictive value, and negative predictive value of the molecular method using the Nugent consensus result as the reference [52].
- Statistical Analysis: Correlation coefficients alone are insufficient to assess agreement [53]. Use statistical methods like Passing-Bablok or Deming regression, which are more appropriate for method comparison than simple t-tests or correlation analysis [53].
Visualizing Diagnostic Workflows and Logical Relationships
The diagrams below illustrate the procedural flow and decision points for each diagnostic method, highlighting where subjectivity is introduced.
Nugent Score Microscopic Workflow
Molecular Diagnostic Workflow
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for BV Diagnostic Research
| Item | Function in Research | Application Context |
|---|---|---|
| Gram Stain Kit | Stains bacterial cells on a microscope slide to differentiate them by cell wall morphology (Gram-positive vs. Gram-negative). | Essential for performing the Nugent score. Components include crystal violet, iodine, decolorizer, and counterstain [52]. |
| Aptima Multitest Swab | Specimen collection swab designed to stabilize nucleic acids (DNA/RNA) for molecular testing. | Used with the Hologic Panther system for Aptima BV, CV, and TV assays [52]. |
| Nucleic Acid Extraction Kits | To isolate and purify bacterial DNA or RNA from vaginal swab samples, removing inhibitors. | Critical first step for all molecular methods, including 16S rRNA sequencing and commercial NAATs [51] [52]. |
| 16S rRNA Gene Primers | Short, specific DNA sequences that bind to and allow amplification of the 16S ribosomal RNA gene for identification and profiling of bacteria. | Used in research-grade molecular pipelines like molBV to characterize the vaginal microbiome [51]. |
| Sabouraud Dextrose Agar (SAB) | A culture medium that supports the growth of fungi, particularly yeasts like Candida. | Used as a reference method for vulvovaginal candidiasis in parallel BV studies [52]. |
| Real-time PCR or TMA Reagents | Chemicals and enzymes required for nucleic acid amplification and real-time detection/quantification of specific bacterial targets. | Core component of commercial NAATs (e.g., Aptima BV uses TMA) and laboratory-developed tests [52]. |
Bacterial vaginosis (BV) represents a significant diagnostic challenge in clinical microbiology and women's health research. The condition, characterized by a shift from Lactobacillus-dominated vaginal flora to a polymicrobial anaerobic community, affects millions of women globally and is associated with serious reproductive health complications including increased risk of preterm birth, sexually transmitted infections, and pelvic inflammatory disease [55]. For researchers and drug development professionals, the fundamental dilemma lies in navigating the complex landscape of BV diagnostic methods, each with distinct strengths, limitations, and implications for research outcomes.
The historical gold standard for BV diagnosis in research settings has been the Nugent score, a Gram stain scoring system that quantifies the ratio of Lactobacillus morphotypes to BV-associated bacteria including Gardnerella, Bacteroides, and Mobiluncus species [56]. A Nugent score of 0-3 is considered normal, 4-6 intermediate (often termed "altered flora"), and 7-10 diagnostic of BV [23]. This system, while widely adopted, demonstrates significant limitations—approximately 10% of vaginal swabs yield indeterminate "altered flora" results, leaving researchers without clear categorization [56]. This diagnostic gray area has propelled the development and validation of molecular diagnostics that target specific BV-associated organisms through DNA-based detection methods.
The validation of Nugent score versus molecular diagnostics for BV research forms a critical nexus in women's health research, influencing everything from basic microbiome studies to therapeutic intervention trials. This comparison guide objectively analyzes the performance characteristics of these diagnostic approaches, providing researchers with experimental data and methodologies to inform study design and interpretation of discordant results.
Comparative Performance of BV Diagnostic Methods
Traditional Diagnostic Methods: Nugent Score and Microscopy
The Nugent scoring system has served as the reference standard for BV diagnosis in research settings since its development in 1991 [56]. This laboratory-based method involves Gram-staining vaginal smears and microscopically examining bacterial morphotypes to generate a numerical score from 0-10. The methodology is labor-intensive and requires trained personnel for accurate interpretation, introducing subjectivity as a potential confounding variable [56]. Additionally, some BV-associated bacteria exhibit Gram-variable morphotypes, further complicating accurate scoring [56].
Clinical diagnosis via Amsel's criteria provides a more rapid, point-of-care alternative requiring at least three of four clinical findings: homogeneous thin discharge, clue cells on microscopy, vaginal pH >4.5, and a positive whiff test [23]. However, this method demonstrates variable sensitivity (37%-70%) and high specificity (94%-99%) when compared to Nugent scoring [23]. The Amsel criteria's dependence on clinical signs rather than microbiological characterization limits its utility for precise research applications where quantitative endpoints are required.
A significant limitation of both traditional methods is their inability to fully characterize the complex polymicrobial nature of BV. Microscopic methods cannot differentiate between specific bacterial species with distinct pathogenic potentials, metabolic profiles, or antibiotic resistance patterns. This limitation becomes particularly problematic for samples falling into the "altered flora" or intermediate Nugent category (scores 4-6), which represent a diagnostic challenge in both clinical and research settings [56].
Molecular Diagnostic Platforms
Molecular diagnostics for BV have emerged as powerful research tools that target specific bacterial nucleic acids to characterize vaginal microbiota. These platforms generally utilize polymerase chain reaction (PCR) technology to detect and quantify BV-associated organisms including Gardnerella vaginalis, Atopobium vaginae, Bacterial Vaginosis-Associated Bacterium 2 (BVAB-2), and Megasphaera type 1, alongside protective Lactobacillus species (L. crispatus, L. jensenii, and L. gasseri) [57] [23].
Several FDA-cleared and laboratory-developed tests are now available for research applications:
- BD Max Vaginal Panel: Utilizes an algorithmic analysis of molecular DNA detection of Lactobacillus species (L. crispatus and L. jensenii) plus G. vaginalis, A. vaginae, BVAB2, and Megasphaera type 1 [23].
- Aptima BV: Detects G. vaginalis, A. vaginae, and Lactobacillus species including L. crispatus, L. jensenii, and L. gasseri [23].
- Research PCR assays: Target specific BV-associated organisms with high sensitivity, such as quantitative PCR assays detecting G. vaginalis or A. vaginae exceeding 10^9 copies/mL or 10^8 copies/mL, respectively [58].
These molecular approaches provide several advantages for research settings, including objective binary outcomes (positive/negative), the ability to detect fastidious organisms that may not be visible on microscopy, and opportunities for archival analysis of stored samples. Additionally, some platforms can simultaneously detect other vaginal pathogens including Candida species and Trichomonas vaginalis, providing a more comprehensive diagnostic profile [57].
Head-to-Head Performance Comparison
Table 1: Diagnostic Performance of BV Testing Modalities Compared to Reference Standards
| Diagnostic Method | Sensitivity (%) | Specificity (%) | Positive Predictive Value (%) | Negative Predictive Value (%) | Overall Agreement with Reference (%) |
|---|---|---|---|---|---|
| Nugent Score (vs. Molecular) | 76.0 | 76.0 | - | - | 76.0 [56] |
| Molecular Test (vs. Nugent) | 90.5-100 | 85.8-93.0 | 73.0 | 100.0 | 94.5 [57] [58] |
| Amsel Criteria (vs. Nugent) | 37.0-70.0 | 94.0-99.0 | - | - | - [23] |
| BVBlue (vs. Nugent) | 38.0-91.7 | 91.0-100.0 | - | - | - [25] |
Table 2: Analytical Characteristics of BV Diagnostic Methods in Research Settings
| Method | Sample Processing Time | Technical Expertise Required | Ability to Detect Mixed Infections | Quantification Capability | Cost Considerations |
|---|---|---|---|---|---|
| Nugent Score | 30-60 minutes | High (microscopy expertise) | Limited | Semi-quantitative (score-based) | Low |
| Amsel Criteria | 15-20 minutes | Moderate (clinical assessment) | Limited | No | Low |
| Molecular Panels | 3-24 hours | Moderate (PCR technical skills) | Excellent (multiplex detection) | Quantitative (organism load) | High |
| BVBlue | 10-15 minutes | Low | Poor (sialidase-negative BV not detected) | No | Moderate |
Molecular methods demonstrate superior performance characteristics for BV diagnosis in research settings, with significantly higher sensitivity and negative predictive value compared to traditional methods [57]. One comprehensive study comparing microscopy with Nugent scoring against a targeted quantitative PCR panel with an interpretive algorithm found 76% overall agreement between the methods [56]. Notably, molecular testing successfully categorized samples designated as "altered flora" by Nugent scoring, with five of nine such samples categorized as BV positive and four as BV negative by qPCR [56].
The diagnostic accuracy advantage of molecular methods is further supported by receiver operating characteristic (ROC) analysis. One study comparing Nugent-BV and Donders criteria (for BV and aerobic vaginitis) against molecular BV identified by 16S rRNA sequencing found that Nugent-BV more accurately aligned with molecular-BV (AUC: 0.88, 95% CI: 0.84-0.91) compared to Donders-AV/BV (AUC: 0.84; CI: 0.80-0.87) [59]. This suggests that while molecular methods outperform traditional approaches, Nugent scoring remains a reasonably accurate method for BV detection in research settings.
Resolution Strategies for Discordant Results
Algorithmic Approach to Diagnostic Discordance
Diagram Title: Diagnostic Discordance Resolution Algorithm
Discordant results between diagnostic methods present both challenges and opportunities for BV researchers. The algorithm above provides a systematic approach for resolving such discordance, particularly for samples with indeterminate Nugent scores (4-6) that require further characterization. Implementation of this structured pathway ensures consistent handling of ambiguous cases across research studies.
When Nugent and molecular results conflict, evidence suggests molecular methods may provide more biologically relevant categorization. In one study, comprehensive qPCR analysis of Nugent "altered flora" samples revealed alternative etiologies summarized as 38% BV, 10% aerobic vaginitis (AV), 5% Candida, 2% sexually transmitted infections, 10% mixed infection, and 35% negative for all targets [56]. This detailed stratification enables researchers to create more homogeneous patient subgroups for analysis, potentially reducing confounding in therapeutic trials and mechanistic studies.
Advanced Resolution Techniques
For research requiring high-resolution characterization of discordant samples, several advanced techniques provide deeper insights:
16S rRNA Microbiome Sequencing: This powerful approach sequences the hypervariable regions of the bacterial 16S rRNA gene to provide comprehensive, culture-independent characterization of vaginal microbial communities. The method confirms bacterial qPCR results and identifies differentiating patterns between AV, BV, and Lactobacillus-dominated vaginal microbiomes [56]. For discordant samples, 16S sequencing can resolve uncertainties by revealing complex community structures not captured by targeted methods.
Machine Learning Applications: Advanced computational methods applied to microbiome sequencing data show promise for improving BV diagnosis while highlighting important methodological considerations. Recent research indicates differential predictive performance across ethnic groups, with models exhibiting lower accuracy for Black women [3]. This highlights the importance of ensuring diverse representation in training datasets and algorithm development. Variation in significant bacterial taxa for predicting BV by ethnicity further supports the need for population-specific analytical approaches in global research [3].
Strain-Level Characterization: Emerging research reveals that what was previously classified as Gardnerella vaginalis actually comprises multiple distinct species with different clinical implications. Drexel researchers have developed a PCR-based test that distinguishes among 11 unique Gardnerella genospecies, with two clades composed of five genospecies showing 100% resistance to metronidazole [60]. This strain-level resolution provides mechanistic insights into treatment failure and recurrence, offering valuable stratification for interventional trials.
Experimental Protocols for Method Comparison
Protocol 1: Nugent Scoring Methodology
Sample Collection and Preparation:
- Collect vaginal specimens using nylon-flocked Copan ESwabs placed into Amie's transport medium
- Transport samples to laboratory within 4-6 hours post-collection
- Prepare vaginal smears by rolling swab onto clean glass slide
- Fix slides with methanol and Gram stain using standard protocol [56]
Microscopic Examination and Scoring:
- Examine slide under oil immersion (1000x magnification)
- Count bacterial morphotypes in multiple fields:
- Large Gram-positive rods (Lactobacillus morphotypes)
- Small Gram-variable rods (Gardnerella morphotypes)
- Curved Gram-variable rods (Mobiluncus morphotypes)
- Calculate Nugent score:
- Lactobacillus: 0 (≥30 per field), 1 (5-30 per field), 2 (1-4 per field), 3 (<1 per field), 4 (0 per field)
- Gardnerella: 0 (0 per field), 1 (<1 per field), 2 (1-4 per field), 3 (5-30 per field), 4 (≥30 per field)
- Mobiluncus: 0 (0 per field), 1 (<1 per field), 2 (≥1 per field)
- Sum scores for final Nugent score: 0-3 (negative), 4-6 (intermediate), 7-10 (BV positive) [56]
Quality Assurance:
- Certified laboratory technologists should perform reading and scoring
- Regular proficiency testing and inter-rater reliability assessments
- Blinded duplicate readings for a subset of samples to assess consistency
Protocol 2: Molecular Testing Using qPCR
DNA Extraction:
- Centrifuge swab transport medium (13,000 × g for 5 minutes)
- Treat bacterial pellets with enzymatic lysis buffer containing lysozyme (20 mg/ml)
- Incubate for 30 minutes at 37°C
- Add proteinase K and buffer AL, incubate for 30 minutes at 56°C
- Use QIAamp spin column protocol (QIAamp DNA Mini kit, Qiagen) for DNA purification
- Elute DNA in 100 μL molecular-grade water [56]
qPCR Assay Setup:
- Utilize commercial PCR/fluorogenic probe-based tests (e.g., BD MAX Vaginal Panel)
- Reaction mix includes primers and probes for:
- BV markers: Lactobacillus spp. (L. crispatus and L. jensenii), G. vaginalis, A. vaginae, BVAB-2, Megasphaera 1
- Candida group: C. albicans, C. tropicalis, C. parapsilosis, C. dubliniensis
- Trichomonas vaginalis
- Set up reactions according to manufacturer specifications
- Include positive and negative controls in each run [57]
Amplification and Analysis:
- Perform real-time PCR using appropriate cycling conditions (typically 2-3 hours)
- Use proprietary algorithm to determine BV positive/negative status based on presence and concentration of each marker
- For research applications, quantitative values (copy numbers) can be recorded for analytical depth [57]
Protocol 3: 16S rRNA Sequencing for Discordant Resolution
Library Preparation:
- Amplify V3-V4 hypervariable region of 16S rRNA gene using universal primers
- Attach Illumina sequencing adapters and dual indices via PCR
- Clean amplified libraries using magnetic bead-based purification
- Quantify libraries using fluorometric methods [56]
Sequencing and Bioinformatics:
- Pool normalized libraries and sequence on Illumina platform (e.g., MiSeq)
- Process raw sequences using QIIME2 or similar pipeline:
- Demultiplex sequences
- Denoise with DADA2 to obtain amplicon sequence variants (ASVs)
- Assign taxonomy using reference database (e.g., Silva, Greengenes)
- Analyze community composition and diversity metrics [56] [3]
Table 3: Research Reagent Solutions for BV Diagnostic Studies
| Research Reagent | Manufacturer/Supplier | Function in Experimental Protocol |
|---|---|---|
| Copan ESwabs | Copan Diagnostics | Standardized vaginal specimen collection and transport |
| Amie's Transport Medium | Copan Diagnostics | Preserves microbial viability during transport |
| QIAamp DNA Mini Kit | Qiagen | Nucleic acid extraction from vaginal samples |
| BD MAX Vaginal Panel | Becton, Dickinson | Multiplex PCR detection of BV, Candida, Trichomonas |
| Illumina MiSeq Reagent Kits | Illumina | 16S rRNA gene sequencing for microbiome analysis |
| Lysozyme | Sigma-Aldrich | Enzymatic cell lysis for DNA extraction |
| Proteinase K | Qiagen | Protein degradation for improved DNA yield |
Research Implications and Future Directions
Interpretation of Discordant Results
The systematic evaluation of discordant diagnostic results reveals several important considerations for BV researchers:
Biological Significance of "Altered Flora": The intermediate Nugent category (scores 4-6) represents a biologically heterogeneous group that may include transitional states, mild BV, or alternative vaginal dysbiotic conditions such as aerobic vaginitis [56]. Molecular characterization enables resolution of this heterogeneity, creating opportunities to study the natural history of vaginal microbiome transitions and identify biomarkers of progression.
Ethnic and Population Considerations: Machine learning analyses have demonstrated that BV-associated bacterial taxa vary significantly across ethnic groups [3]. This population-level variation in vaginal microbiome composition complicates diagnostic accuracy and necessitates careful consideration of participant demographics in research studies. Models trained on diverse populations and validated within specific ethnic groups will strengthen research generalizability.
Therapeutic Implications: Diagnostic discordance has direct relevance for therapeutic development. The discovery that specific Gardnerella genospecies demonstrate 100% resistance to metronidazole but remain susceptible to clindamycin [60] provides a mechanistic explanation for treatment failure and highlights the potential for precision medicine approaches to BV therapy.
Emerging Technologies and Approaches
Diagram Title: Future BV Diagnostic Research Framework
The evolving landscape of BV diagnostics and therapeutics points to several promising research directions:
Precision Microbiology: Moving beyond species-level characterization to strain-level resolution will enable researchers to address fundamental questions about BV pathogenesis, transmission, and recurrence. The development of PCR assays that distinguish metronidazole-resistant Gardnerella genospecies represents an important step toward personalized BV management [60].
Partner Intervention Strategies: The recent ACOG recommendation supporting concurrent sexual partner therapy for recurrent BV [61] marks a paradigm shift in BV management. This approach, based on new data demonstrating reduced recurrence when partners receive combination oral and topical antimicrobials, opens new avenues for research into BV transmission dynamics and couple-based interventions.
Novel Therapeutic Approaches: High recurrence rates following antibiotic therapy (50-80% within 6-12 months) [55] have stimulated investigation of alternative approaches including vaginal microbiome transplantation, probiotics, phage therapy, and biofilm disruption. The limited efficacy of current antibiotics underscores the need for continued therapeutic innovation informed by sophisticated diagnostic approaches.
The resolution of discordant BV diagnostic results represents both a methodological challenge and scientific opportunity for researchers. The comparison presented in this guide demonstrates that molecular diagnostics offer superior sensitivity and objectivity compared to traditional Nugent scoring, particularly for resolving indeterminate cases. However, Nugent scoring remains a valuable method for many research applications, especially when complemented by molecular approaches for ambiguous samples.
The optimal diagnostic strategy depends on research objectives, resources, and population characteristics. For large epidemiological studies, Nugent scoring may provide sufficient resolution at manageable cost. For therapeutic trials and mechanistic studies, molecular methods reduce misclassification and enable precise stratification. Emerging technologies including strain-level characterization and machine learning algorithms promise to further enhance diagnostic precision while highlighting the importance of diverse, representative populations in research datasets.
As the field advances, integration of multiple diagnostic approaches within a comprehensive research framework will maximize insights into BV pathogenesis, transmission, and therapeutic response. This comparative guide provides researchers with the experimental protocols and analytical frameworks necessary to navigate diagnostic discordance and advance the field of BV research.
Bacterial vaginosis (BV) represents a significant diagnostic challenge in clinical microbiology and women's health research. The condition, characterized by a shift from a Lactobacillus-dominated vaginal microbiome to a polymicrobial anaerobic state, affects millions of women globally and is associated with serious health complications including increased susceptibility to sexually transmitted infections and adverse pregnancy outcomes [56]. For researchers and drug development professionals, selecting appropriate diagnostic methods involves navigating a complex trade-off between analytical accuracy and practical accessibility. This comparison guide provides an objective evaluation of the established Nugent scoring system against emerging molecular diagnostics, examining their respective performance characteristics, methodological requirements, and implementation considerations to inform research design and development decisions.
Methodological Frameworks: A Comparative Analysis of Diagnostic Approaches
Nugent Scoring: The Microscopic Gold Standard
The Nugent scoring system, developed in the early 1990s, remains the reference standard for BV diagnosis in research settings. This method employs light microscopy to examine Gram-stained vaginal smears, quantifying bacterial morphotypes to generate a numerical score from 0-10 [56]. The protocol involves specific examination of three bacterial forms: large Gram-positive rods (Lactobacillus), small Gram-variable rods (Gardnerella vaginalis), and curved Gram-variable rods (Mobiluncus). Researchers calculate the score by subtracting the sum of Lactobacillus and Mobiluncus morphotype counts from the Gardnerella count, with scores of 7-10 indicating BV, 4-6 representing intermediate/"altered flora," and 0-3 considered normal [24]. The method's standardization allows for reasonable reproducibility across laboratory settings, though it requires experienced microscopists to correctly identify and enumerate bacterial morphotypes.
Amsel's Clinical Criteria: The Bedside Alternative
Often used alongside Nugent scoring in research protocols, Amsel's criteria offer a clinical diagnostic approach requiring at least three of four findings: homogeneous grayish-white discharge, vaginal pH >4.5, positive amine odor with 10% KOH addition, and presence of clue cells on microscopic examination [24]. While accessible and inexpensive, this method demonstrates variable sensitivity compared to laboratory-based standards, with one study reporting 50% sensitivity against Nugent scoring, though with high specificity (98.2%) [24]. The clinical nature of Amsel's criteria makes them particularly relevant for researchers validating point-of-care diagnostic platforms or conducting clinical outcome studies.
Molecular Diagnostics: Precision through Amplification
Molecular approaches to BV diagnosis represent a paradigm shift from morphological to genomic identification. Quantitative PCR (qPCR) methods target specific bacterial DNA sequences, including 16S rRNA genes, to identify and quantify BV-associated organisms such as Gardnerella vaginalis, Atopobium, Prevotella, and Megasphaera species [56]. Next-generation sequencing of the 16S rRNA gene provides even more comprehensive microbiome profiling, enabling researchers to characterize complex microbial communities without prior taxonomic assumptions. Commercial automated molecular platforms like the Hologic Panther Aptima BV and CV/TV assays have emerged as alternatives to conventional testing, though questions remain about their clinical significance relative to cost implications [62].
Performance Comparison: Quantitative Data Analysis
Table 1: Diagnostic Performance Metrics Across BV Testing Methodologies
| Diagnostic Method | Sensitivity | Specificity | Positive Predictive Value | Negative Predictive Value | Technical Complexity |
|---|---|---|---|---|---|
| Nugent Scoring | 76% (vs. qPCR) [56] | 76% (vs. qPCR) [56] | 87.5% (vs. Amsel's) [24] | 88.8% (vs. Amsel's) [24] | Moderate |
| Amsel's Criteria | 50% (vs. Nugent) [24] | 98.2% (vs. Nugent) [24] | 87.5% (vs. Nugent) [24] | 88.8% (vs. Nugent) [24] | Low |
| qPCR with Algorithm | Reference | Reference | Not reported | Not reported | High |
| Microscopy for Candida | 94% (vs. qPCR) [56] | 94% (vs. qPCR) [56] | Not reported | Not reported | Moderate |
Table 2: Diagnostic Categorization Across Methods in a Comparative Study (n=93)
| Diagnostic Category | qPCR with Algorithm | Nugent Scoring | Discrepancy Notes |
|---|---|---|---|
| BV Positive | 38% | 24% | 5/9 "altered flora" by Nugent were BV+ by qPCR |
| Aerobic Vaginitis | 10% | Not detected | Identified alternative etiology for Nugent-negative cases |
| Candida Infection | 5% | Similar to microscopy | 94% agreement between methods |
| STI Pathogens | 2% | Not detected | Additional diagnostic information |
| Mixed Infections | 10% | Not differentiated | Multiple positive targets identified |
| Negative for All Targets | 35% | 47% | Comprehensive ruling out of infection |
Diagnostic Workflows: Technical Pathways
Diagram 1: Comparative diagnostic pathways for bacterial vaginosis testing
Research Reagent Solutions: Essential Materials for BV Diagnostics
Table 3: Key Research Reagents and Platforms for BV Diagnostic Studies
| Reagent/Platform | Application | Research Utility |
|---|---|---|
| Copan ESwabs with Amie's Transport Medium | Sample collection and preservation | Maintains microbial viability for both culture and molecular studies; enables standardized specimen transport [56] |
| Gram Stain Reagents | Nugent scoring microscopy | Differentiates bacterial morphotypes for quantitative scoring of Lactobacillus, Gardnerella, and Mobiluncus [24] |
| QIAamp DNA Mini Kit | Nucleic acid extraction | Provides high-quality DNA for downstream qPCR and sequencing applications; includes enzymatic lysis for Gram-positive bacteria [56] |
| 16S rRNA Primers | Microbiome profiling | Enables amplification of conserved bacterial regions for community analysis via next-generation sequencing [56] |
| Species-Specific qPCR Assays | Targeted pathogen detection | Quantifies specific BV-associated organisms (Gardnerella, Atopobium, etc.) with high sensitivity [56] |
| APTIMA T. vaginalis Assay | STI co-testing | Detects trichomoniasis infections that may co-occur with BV using NAAT technology [56] |
| Hologic Panther Platform | Automated molecular testing | Standardized high-throughput testing for multi-pathogen detection in large-scale studies [56] |
Cost-Benefit Considerations for Research Implementation
Analytical Performance Versus Accessibility
The fundamental trade-off between diagnostic accuracy and practical accessibility presents researchers with methodological decisions that significantly impact study validity, generalizability, and cost. Molecular methods like qPCR with interpretive algorithms demonstrate superior diagnostic precision, identifying BV in 38% of samples compared to 24% by Nugent scoring in one study [56]. More importantly, comprehensive qPCR panels identified alternative diagnoses in samples categorized as "altered flora" by Nugent scoring, including aerobic vaginitis (10%), STIs (2%), and mixed infections (10%) [56]. This expanded diagnostic capability comes with substantial cost implications, including equipment investment, reagent expenses, and technical expertise requirements.
Resource Allocation in Study Design
Research settings with limited budgets must carefully consider whether the incremental diagnostic precision of molecular methods justifies their additional costs. The high specificity of Amsel's criteria (98.2%) suggests potential utility as a screening tool in resource-constrained settings, though its low sensitivity (50%) risks missing true positive cases [24]. The "altered flora" categorization in Nugent scoring (approximately 10% of cases) represents a particular challenge, leaving researchers and clinicians without clear guidance [56]. Molecular methods successfully reclassify these indeterminate cases but require significant infrastructure. This balance mirrors broader diagnostic trade-offs observed in clinical decision support systems, where increasing disease coverage typically reduces accuracy—approximately 1% drop in top-3 accuracy for every 10 diseases added to coverage [63].
Research Context Considerations
The optimal diagnostic approach varies significantly by research context. Epidemiological studies requiring high throughput may prioritize standardized microscopy, while therapeutic intervention trials may benefit from molecular methods' precision. Drug development targeting specific BV-associated pathogens necessitates qPCR or sequencing for patient stratification and treatment response assessment. Studies examining microbiome dynamics or novel therapeutic mechanisms would benefit most from 16S rRNA sequencing, despite its higher cost and computational requirements [56]. Research in resource-limited settings might employ a tiered approach, using Amsel's criteria for screening with reflexive molecular testing for indeterminate cases [24].
The validation of BV diagnostic methodologies requires careful consideration of research objectives, resource constraints, and intended applications. While Nugent scoring provides a standardized, cost-effective approach suitable for many research settings, molecular diagnostics offer unprecedented resolution for characterizing vaginal dysbiosis and detecting mixed infections. The research question should drive method selection: therapeutic mechanism studies warrant investment in molecular precision, while large-scale epidemiological surveillance may prioritize accessibility and standardization. As molecular technologies become more accessible and computational tools more sophisticated, integrated approaches that leverage both microscopic and molecular data will likely provide the most comprehensive insights into bacterial vaginosis pathogenesis and treatment.
Head-to-Head Validation: Diagnostic Accuracy, Sensitivity, and Specificity
Bacterial vaginosis (BV) represents a common gynecological condition characterized by a dysbiosis of the vaginal microbiome, where healthy Lactobacillus-dominant flora is replaced by anaerobic bacteria including Gardnerella vaginalis, Fannyhessea vaginae (formerly Atopobium vaginae), and others [52] [23]. This imbalance has significant health implications, associating with increased risk of sexually transmitted infections, adverse pregnancy outcomes, and pelvic inflammatory disease [23]. Accurate diagnosis is thus critical for both clinical management and public health initiatives.
The diagnostic landscape for BV has traditionally been dominated by the Nugent score, a Gram stain-based scoring system that quantifies the ratio of bacterial morphotypes to determine vaginal flora status [52]. However, molecular methods utilizing nucleic acid amplification tests (NAATs) have emerged as promising alternatives, offering potential improvements in objectivity, throughput, and accuracy [52] [64]. This meta-analysis comprehensively evaluates the performance characteristics of both diagnostic approaches within the context of BV research, providing researchers and drug development professionals with critical insights for methodological selection.
Comparative Diagnostic Performance Data
Extensive evaluation across multiple studies reveals distinct performance patterns for Nugent scoring versus molecular assays. The table below synthesizes key performance metrics from recent comparative studies.
Table 1: Diagnostic Performance of Nugent Score versus Molecular Assays for Bacterial Vaginosis
| Diagnostic Method | Sensitivity (%) | Specificity (%) | Reference Standard | Study (Citation) |
|---|---|---|---|---|
| Nugent Score | 63.9 | >92.4 | Microbiota Analysis | [20] |
| Amsel Criteria | 61.1 | >92.4 | Microbiota Analysis | [20] |
| Hologic Aptima BV Assay | 95.0 - 97.5 | 85.8 - 96.3 | Nugent Score & Gram Stain | [52] [23] |
| BD MAX Vaginal Panel | 90.5 - 96.2 | 85.8 - 96.1 | Nugent Score & Composite Reference | [23] [65] |
| AmpliSens Florocenosis/BV PCR | 80.6 | >92.4 | Microbiota Analysis | [20] |
| Vaginal Panel (Vircell) qPCR | 93.1 | 88.8 | Nugent Score | [64] |
Molecular assays consistently demonstrate superior sensitivity compared to traditional methods, with the Hologic Aptima BV assay achieving sensitivity rates as high as 97.5% in evaluations against Gram stain and Nugent score [52]. The BD MAX Vaginal Panel also shows high sensitivity (96.2%) and specificity (96.1%) in comparative studies [65]. This enhanced detection capability translates to more reliable identification of BV-positive cases in research cohorts.
Analysis of Discordant Results and Indeterminate Categories
A significant finding across multiple studies concerns the handling of intermediate or indeterminate results. The Nugent score categorizes samples as negative (0-3), indeterminate (4-6), or positive (7-10) for BV, creating a "grey zone" that complicates definitive diagnosis [52]. One evaluation noted that Gram stain interpretation was prone to error, with 20% of samples showing discordant results on duplicate examination [52].
In contrast, most molecular assays provide dichotomous outcomes (positive/negative), eliminating indeterminate categories. Research indicates that patients with BV-indeterminate Nugent results were less likely to receive treatment than those with positive results, suggesting that molecular methods could lead to more consistent clinical management [52]. Furthermore, molecular testing identified additional BV cases classified as normal by Nugent scoring, demonstrating enhanced detection capability [66].
Detailed Experimental Protocols
Nugent Score Methodology
The Nugent score remains the reference standard for BV diagnosis in research settings. The protocol involves microscopic evaluation of Gram-stained vaginal smears with semi-quantitative assessment of three bacterial morphotypes [52]:
- Sample Collection: Vaginal swabs are collected from the posterior fornix using standardized collection systems (e.g., Amies agar gel swabs).
- Slide Preparation: Smears are prepared on glass slides and heat-fixed before undergoing Gram staining. Automated staining systems (e.g., Aerospray automated staining device) may be employed for consistency.
- Microscopic Examination: Stained slides are examined under oil immersion (1000x magnification). The scorer quantifies:
- Large Gram-positive rods (Lactobacillus morphotypes): Scored from 0 (≥30 per field) to 4 (none seen)
- Small Gram-variable rods (Gardella vaginalis morphotypes): Scored from 0 (none seen) to 4 (≥30 per field)
- Curved Gram-variable rods (Mobiluncus morphotypes): Scored from 0 (none seen) to 2 (5-30 per field)
- Scoring and Interpretation: The individual category scores are summed to generate a total Nugent score:
- 0-3: Negative for BV (normal vaginal flora)
- 4-6: Intermediate/indeterminate
- 7-10: Positive for BV
To ensure research reliability, slides should be read independently by two trained personnel, with a third reader resolving discordant categorical results [52]. This method requires substantial expertise and suffers from subjectivity in morphological interpretation.
Molecular Assay Protocols
Molecular methods employ NAATs to detect and quantify BV-associated organisms. The following protocol represents a generalized approach for commercial PCR-based BV assays:
- Sample Collection: Vaginal swabs are collected using manufacturer-specific collection kits (e.g., Aptima Multitest swab, BD MAX UVE Specimen Collection Kit) to preserve nucleic acids.
- Nucleic Acid Extraction: Automated extraction systems (e.g., COBAS AmpliPrep) purify total nucleic acids from specimens according to established protocols [66]. Eluted DNA is quantified for quality control.
- Target Amplification and Detection: Multiplex real-time PCR reactions are performed using commercial master mixes targeting key organisms:
- BV-Associated Bacteria: G. vaginalis, F. vaginae, Megasphaera-1, BVAB-2
- Healthy Flora Bacteria: L. crispatus, L. jensenii, L. gasseri
- Result Interpretation: Proprietary algorithms analyze the quantitative ratios of bacterial targets to generate a final diagnosis. For example:
- The Hologic Aptima BV assay uses transcription-mediated amplification (TMA) to detect Lactobacillus species (L. gasseri, L. crispatus, L. jensenii) versus BV-associated bacteria (F. vaginae and G. vaginalis) [52].
- The AmpliSens assay calculates ratio coefficients (RC) based on log differences between bacterial concentrations to determine BV status [66].
These systems provide automated, objective results with minimal operator interpretation bias, making them suitable for high-throughput research environments.
Diagram 1: Comparative diagnostic workflows for BV testing
Key Research Reagent Solutions
Table 2: Essential Research Materials for BV Diagnostic Studies
| Reagent / Kit Name | Manufacturer | Primary Function in Research | Key Components / Targets |
|---|---|---|---|
| Aptima BV Assay | Hologic | NAAT for BV diagnosis using TMA technology | Detects L. crispatus, L. jensenii, L. gasseri, G. vaginalis, F. vaginae |
| BD MAX Vaginal Panel | Becton Dickinson | Multiplex real-time PCR for vaginitis diagnosis | Detects BV targets (Lactobacillus, G. vaginalis, A. vaginae), Candida species, T. vaginalis |
| AmpliSens Florocenosis/BV PCR | ATRiDA (InterLabService) | Quantitative PCR for BV diagnosis | Detects Lactobacillus spp., G. vaginalis, A. vaginae, total bacterial DNA |
| Vaginal Panel Realtime PCR | Vircell, SL | Multiplex qPCR for vaginitis pathogens | Detects G. vaginalis, Lactobacillus spp., F. vaginae, Candida species, T. vaginalis |
| Affirm VPIII | Becton Dickinson | DNA probe hybridization for vaginitis | Detects G. vaginalis, Candida spp., T. vaginalis |
| BVBlue | Sekisui Diagnostics | Point-of-care sialidase activity test | Detects sialidase enzymes produced by BV-associated bacteria |
| Nugent Score Components | N/A | Reference standard method | Gram stain reagents, microscope, standardized swab collection systems |
Discussion and Research Implications
Performance and Methodological Considerations
The accumulated evidence demonstrates that molecular assays offer significant advantages in sensitivity over traditional Nugent scoring, with more modest gains in specificity [52] [65]. This enhanced detection capability stems from the quantitative nature of PCR-based methods, which can identify specific bacterial targets at lower concentrations than visually discernible thresholds in microscopy.
The diagnostic approach using a combination of L. crispatus with G. vaginalis has shown particularly promising performance, with one study reporting sensitivity of 99.29% and specificity of 97.79% [4]. This two-target approach effectively captures the essential ecological shift in BV while maintaining cost-effectiveness for large-scale studies.
Molecular methods also eliminate the problematic indeterminate category inherent to Nugent scoring, providing definitive binary outcomes that simplify data analysis in research settings [52]. Furthermore, automated platforms reduce technical variability and operator dependency, enhancing reproducibility across multiple research sites [52] [64].
Limitations and Future Directions
Despite their advantages, molecular assays present limitations for research applications. Current FDA-cleared NAATs are approved for symptomatic women only, and their performance characteristics in asymptomatic research populations remain less defined [23]. Additionally, the higher cost of molecular testing may be prohibitive for large epidemiological studies with limited budgets.
Future research should focus on standardizing molecular thresholds across platforms and validating their use in diverse asymptomatic populations. The integration of novel biomarkers, such as sialidase detection in the BVBlue system [67], may provide complementary approaches for specific research questions. As molecular technologies continue to evolve and become more cost-effective, they are poised to become the new reference standard for BV diagnosis in research contexts, potentially replacing the century-old Gram stain method that has defined the field to date.
The diagnosis of bacterial vaginosis (BV), a common gynecological condition linked to serious adverse health outcomes, has long relied on microscopic methods such as the Nugent score [8]. However, the evolving understanding of BV as a polymicrobial condition and the documented limitations of traditional techniques have accelerated the adoption of molecular diagnostic platforms, including quantitative polymerase chain reaction (qPCR) [8]. This guide provides an objective, data-driven comparison of the Nugent score and qPCR tests for BV diagnosis within clinical cohorts, framing the discussion within the broader thesis of validating molecular diagnostics for BV research. We summarize performance metrics from recent clinical studies, detail experimental protocols, and visualize the workflows to aid researchers, scientists, and drug development professionals in evaluating these technologies.
Performance Comparison in Clinical Cohorts
Recent prospective studies directly comparing qPCR and the Nugent score within the same cohort reveal key differences in their diagnostic capabilities. The table below summarizes quantitative performance data and findings from clinical studies.
Table 1: Direct Performance Comparison of qPCR and Nugent Score in Clinical Studies
| Study (Year) | Cohort Size & Description | qPCR Platform / Method | Key Comparative Findings | Reported Performance Metrics |
|---|---|---|---|---|
| Flora Select (2024) [68] | 556 pregnant women (8-12 weeks gestation) | Flora Select real-time PCR test | • qPCR detected BV-associated bacteria (Gardnerella, Prevotella, Atopobium) more effectively than conventional culture.• Ureaplasma was detected in 40.4% of women with Nugent ≥7 and in 45% of those "cured" post-treatment (Nugent 0-3). | • Gardnerella was detected in 100% of women with Nugent ≥7 [68]. |
| Flora Select (2025) [29] | 25 pregnant women with BV (Nugent ≥7) pre- and post-treatment | Flora Select real-time PCR test | • After metronidazole, 88% (22/25) were cured per Nugent score, but 65.2% (15/23) still met molecular BV criteria via qPCR.• qPCR identified residual molecular BV and Ureaplasma in Nugent-cured cases. | • 25% (5/20) of cases cured by Nugent score still met qPCR BV criteria [29]. |
| Vaginal Panel PCR Kit (2024) [64] | 1,011 vaginal swab specimens | Vaginal Panel Real-Time PCR kit (Vircell, SL) | The qPCR test showed high concordance with the Nugent score for BV diagnosis. | • Sensitivity: 93.1%• Specificity: 88.8%• Kappa Value: 0.82 [64] |
These studies consistently demonstrate that while the Nugent score and qPCR show good agreement, qPCR offers additional insights. It is more sensitive in detecting specific BV-associated pathogens and can identify a state of "molecular BV" that persists after treatment even when the Nugent score normalizes [68] [29]. Furthermore, qPCR can simultaneously detect other miscarriage/preterm birth-associated bacteria like Ureaplasma and Mycoplasma, which the Nugent score does not [68].
Methodologies and Analytical Workflows
A critical differentiator between these diagnostic approaches lies in their underlying methodologies. The following diagrams and descriptions outline the core workflows for each technique.
Nugent Score Workflow
The Nugent score is a laboratory-based microscopic method that involves Gram-staining vaginal smears and scoring bacterial morphotypes based on their appearance [8].
Diagram 1: Nugent Score Workflow
The Nugent score is calculated by assessing three bacterial morphotypes under oil immersion microscopy (1000x magnification) [8] [69]:
- Large Gram-positive rods (Lactobacillus morphotypes): Scored from 0 (≥30 per field) to 4 (0 per field)
- Small Gram-variable rods (Gardnerella vaginalis and Bacteroides morphotypes): Scored from 0 (0 per field) to 4 (≥30 per field)
- Curved Gram-variable rods (Mobiluncus morphotypes): Scored from 0 (0 per field) to 2 (≥5 per field)
The final score (0-10) determines the diagnosis: 0-3 (BV negative), 4-6 (intermediate), and 7-10 (BV positive) [8]. A key limitation is inter-observer variability, as accuracy depends heavily on the technician's skill and experience [47] [46]. One study reported only 64.03% complete agreement between three independent microbiologists [46].
qPCR Diagnostic Workflow
qPCR tests for BV use DNA extraction and amplification to quantitatively detect and identify specific bacterial species present in the vaginal microbiome [8].
Diagram 2: qPCR Diagnostic Workflow
The qPCR process involves [29]:
- DNA Extraction: Bacterial DNA is extracted from vaginal swab samples using commercial kits on automated systems (e.g., MagNA Pure 24).
- Amplification & Detection: The extracted DNA is amplified using a real-time PCR system (e.g., CFX96 Touch, Bio-Rad) with SYBR Green or fluorescent-labeled probes and specific primers targeting relevant bacteria.
- Analysis & Interpretation: The test quantifies the total bacterial load, determines the relative abundance of Lactobacillus species, and detects specific BV-associated pathogens (e.g., Gardnerella, Prevotella, Atopobium) as well as other bacteria like Ureaplasma and Mycoplasma [68] [29]. Diagnostic criteria are based on established thresholds for bacterial loads and ratios [29].
Analysis of Key Molecular Targets
The superior specificity of qPCR stems from its ability to target precise genomic sequences of key bacteria. Research into optimal molecular markers indicates that combining a protective Lactobacillus species with a key pathogen offers a highly accurate and cost-effective diagnostic strategy.
Table 2: Key Molecular Diagnostic Targets for BV
| Bacterial Target | Role in Vaginal Microbiome | Utility in BV Diagnosis |
|---|---|---|
| Lactobacillus crispatus | Dominant healthy flora; produces lactic acid and H₂O₂ to maintain low pH and inhibit pathogens [4]. | A marker for a healthy vaginal environment. Its depletion is a key indicator of dysbiosis. |
| Gardnerella vaginalis | Facultative anaerobe; a key pathogen in BV; produces adhesins and sialidase, disrupts epithelial barriers, and forms biofilms [4] [69]. | The most significant biomarker for BV. Its overgrowth is strongly associated with clinical BV. |
| Fannyhessea vaginae (formerly Atopobium vaginae) | Anaerobic bacterium highly associated with BV, often co-occurring with G. vaginalis in biofilms [4] [64]. | Improves diagnostic accuracy when combined with G. vaginalis, especially for recurrent BV. |
A 2025 study found that using a single bacterium for diagnosis was suboptimal, but combining L. crispatus and G. vaginalis achieved a sensitivity of 99.29% and specificity of 97.79%, representing a highly effective and cost-efficient two-marker approach [4].
Advanced Technologies and Research Directions
The field of BV diagnostics is rapidly evolving with several advanced technologies emerging from research settings.
Machine Learning and Deep Learning
To address the subjectivity of the Nugent score, deep learning models have been developed to automate the classification of Gram-stained vaginal smears. One study achieved 94% accuracy in a four-group classification, outperforming the average accuracy of laboratory technicians (92%) [47]. However, researchers must be cautious of potential biases; one analysis showed that machine learning models for BV can exhibit differential predictive performance across ethnicities, with lower accuracy for Black women, potentially due to natural variations in healthy vaginal microbiomes [3]. Mitigation strategies include using paired-ethnicity training datasets and tailored feature selection [3].
Isothermal Amplification and CRISPR-Based Assays
For resource-limited settings, novel molecular techniques are being developed. One study established a rapid assay for Gardnerella vaginalis using Recombinase-Aided Amplification (RAA) combined with CRISPR-Cas12a [69]. This method:
- Is performed at a constant temperature (39°C), eliminating the need for expensive thermal cyclers.
- Can provide results in 15-20 minutes after DNA extraction.
- Has high sensitivity and specificity, making it suitable for point-of-care testing [69].
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Kits for BV Diagnostic Research
| Research Tool | Specific Examples | Primary Function in BV Research |
|---|---|---|
| DNA Extraction Kits | Multi-type Sample DNA/RNA Extraction-Purification Kit (magnetic beads method) [4]; MagNA Pure 24 system (Roche) [29] | Isolates high-purity bacterial genomic DNA from complex vaginal swab samples for downstream molecular analysis. |
| qPCR Master Mixes | THUNDERBIRD Next SYBR qPCR Mix (TOYOBO) [29] | Provides enzymes, buffers, and fluorescent dyes for efficient and specific amplification of target DNA in real-time PCR. |
| Commercial qPCR Kits | Flora Select (Varinos Inc.) [68] [29]; Vaginal Panel Real-Time PCR Kit (Vircell, SL) [64] | Multiplex assays for simultaneous detection and quantification of multiple BV-related bacteria and lactobacilli. |
| Gram Staining Kits | Bartholomew and Mittwer methods (Muto Pure Chemicals); Favor method (Shimadzu Diagnostics) [29] | Enables microscopic differentiation of bacterial morphotypes for Nugent scoring. |
| Isothermal Amplification Kits | RAA amplification kit (Hangzhou ZC Bio-Sci&Tech) [69] | Enables rapid amplification of target DNA at a constant temperature for point-of-care diagnostic development. |
| CRISPR Reagents | LbCas12a protein (Tolo Biotech) [69] | Provides the enzyme and buffer system for highly specific detection of amplified DNA sequences in CRISPR-based assays. |
Direct comparisons in clinical cohorts solidify the complementary roles of the Nugent score and qPCR in BV research. The Nugent score remains a cost-effective, standardized method but is constrained by subjectivity and an inability to identify specific pathogens. In contrast, qPCR offers superior sensitivity, specificity, and objectivity, providing a quantitative molecular profile of the vaginal microbiome. This includes detecting persistent dysbiosis post-treatment and key pathogens like Ureaplasma, which are invisible to microscopy [68] [29]. The choice between them hinges on research objectives: the Nugent score is sufficient for large-scale phenotyping, while qPCR is indispensable for studies exploring the molecular ecology of BV, treatment efficacy beyond clinical cure, and associations with specific pathogens. Emerging technologies like deep learning and CRISPR-based assays promise to further enhance diagnostic accuracy and accessibility, shaping the future of BV research and clinical management.
An objective comparison for research and development professionals
The Diagnostic Shift: From Microscopy to Molecules
The diagnosis of vaginal conditions is undergoing a fundamental transformation, moving from subjective, morphology-based microscopic methods toward precise, nucleic acid-based molecular techniques. For decades, the Nugent score—a microscopic evaluation of a Gram-stained vaginal smear—has been the gold standard in bacterial vaginosis (BV) research, classifying results as BV-negative (0-3), intermediate (4-6), or BV-positive (7-10) [8]. However, this method has critical limitations: it only assesses bacterial morphotypes, lacks species-level resolution, and its "intermediate" category is notoriously difficult to interpret clinically [8] [22]. Molecular diagnostics now challenge this paradigm by offering unprecedented resolution into the vaginal microbiome, enabling the specific identification of alternative etiologies that traditional methods miss.
This guide provides an objective comparison of these technologies, framing them within the ongoing validation of Nugent scores versus molecular diagnostics for BV research. We present performance data, experimental protocols, and essential research tools to inform your developmental work.
Molecular Testing Platforms: A Performance Comparison
Molecular diagnostics for vaginal health encompass several technology platforms, each with distinct strengths and applications in research and development.
| Technology Platform | Key Detectable Targets | Advantages | Limitations | Reported Performance (vs. Reference Methods) |
|---|---|---|---|---|
| Multiplex Real-Time PCR (qPCR) [8] [11] | • Specific BV-associated bacteria (e.g., G. vaginalis, A. vaginae)• Candida species and groups• Trichomonas vaginalis• Key Lactobacillus species | • High sensitivity and specificity• Quantitative results• Automated, commercially available platforms• Relatively fast turnaround (~3 hours) | • Limited to pre-defined targets• Does not assess microbial viability or activity | BV: Sensitivity: 90.5-96.7%; Specificity: 85.8-95% [8]CV: Sensitivity: 96.3%; Specificity: 91.2% (vs. culture) [1]TV: Sensitivity & Specificity: ~100% [1] [57] |
| Next-Generation Sequencing (NGS) [8] [22] | • Broad, unbiased profiling of vaginal microbiome• Community State Type (CST) classification (I-V) | • Hypothesis-free, comprehensive discovery• Reveals complex microbial community structures | • Higher cost and complexity• Data interpretation requires bioinformatics expertise | • High concordance with clinical diagnosis [8]• Reveals significant discordance with CST assignment from other methods [22] |
| Metatranscriptomics [22] | • Active microbial community (via RNA)• Gene expression and functional pathways | • Distinguishes active from dormant community members• Provides functional insights into the microbiome | • Even higher cost than NGS• Technically challenging sample preparation | • Concordance with metataxonomic CST assignment can be as low as 59% [22] |
Key Insights from Comparative Studies
Beyond Single-Target Detection: The power of multiplex PCR lies in its algorithmic approach. For BV, diagnosis is not based on a single pathogen but on a proprietary algorithm that weighs the presence and concentration of a panel of markers, including protective lactobacilli and BV-associated bacteria [57] [12]. One study developing a syndromic panel using machine learning reported 93.8% sensitivity and 96.1% specificity for BV [12].
Revealing Diagnostic Discordance: A 2025 study highlighted that common sequencing methods (metataxonomics, metagenomics, and metatranscriptomics) can lead to substantially different molecular diagnoses of BV, with concordance between methods as low as 59% for Community State Type (CST) assignment [22]. This underscores that the choice of molecular method itself can significantly impact research outcomes and diagnostic conclusions.
Pinpointing Alternative Etiologies: Molecular testing excels at differentiating mixed infections. For example, a test may identify specific Candida species (e.g., C. glabrata, C. krusei), which is crucial as some species exhibit innate resistance to first-line antifungals like fluconazole [1] [57]. Furthermore, by quantifying the absence of lactobacilli and the overgrowth of anaerobes, these tests can objectively indicate Aerobic Vaginitis (AV) and other dysbiotic states that a Nugent score might misclassify or miss entirely [8] [11].
Experimental Protocols for Validation
For researchers validating new assays or conducting comparative studies, understanding the underlying methodologies is critical. Below are outlines of common experimental workflows.
Detailed Methodology for a Multiplex PCR Study
The following protocol is synthesized from recent studies that compare molecular tests to reference standards like the Nugent score [57] [11].
1. Sample Collection and Ethics: The study should obtain approval from an institutional review board (IRB). Symptomatic participants are recruited, and vaginal swabs are collected. A key step is using a predetermined, rotating order of collection to avoid bias from swab sequence [57]. Multiple swabs are needed for the index test (the molecular assay) and the reference methods (Nugent, culture, etc.).
2. Reference Method Execution:
- Nugent Scoring: A vaginal swab is used to create a smear on a glass slide, which is Gram-stained. A trained microscopist examines the slide, scoring it based on the presence of three bacterial morphotypes: large Gram-positive rods (Lactobacillus), small Gram-variable rods (Gardnerella), and curved Gram-variable rods (Mobiluncus). The scores are summed to generate a final Nugent score of 0-10 [57] [8].
- Culture: For Candida and Trichomonas vaginalis, culture remains a common reference standard. Swabs are inoculated onto appropriate culture media (e.g., Sabouraud dextrose agar for Candida, InPouch TV system for T. vaginalis) and incubated. For Candida, subsequent bidirectional sequencing of the ITS2 gene from purified isolates provides species-level confirmation [57].
3. Index Molecular Testing: The swab for the molecular test is stored in a specific buffer and transported to the laboratory. Automated systems (e.g., BD MAX, Hologic Panther) are often used. The process involves:
- Nucleic Acid Extraction: Automated extraction of DNA and/or RNA from the sample.
- Amplification and Detection: The test uses real-time PCR with specific primers and fluorogenic probes to detect DNA targets for a panel of organisms. For BV, this includes quantitating Lactobacillus species (e.g., L. crispatus, L. jensenii) and BV-associated bacteria (e.g., G. vaginalis, A. vaginae, BVAB2). For CV/TV, targets include Candida group, C. glabrata, C. krusei, and T. vaginalis [57] [11].
- Result Interpretation: For syndromic panels, a proprietary algorithm interprets the quantitative data to generate a positive or negative result for BV, CV, and TV.
4. Data Analysis: Sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) are calculated for the molecular test compared to the reference methods. Statistical differences can be determined using logistic modeling, and agreement can be assessed using Cohen's kappa coefficient [57].
The Scientist's Toolkit: Essential Research Reagents & Materials
For laboratories embarking on research in this field, the following reagents and materials are fundamental.
| Item | Function / Application in Research |
|---|---|
| BD MAX Vaginal Panel [57] [49] | A commercially available, FDA-cleared multiplex PCR test for BV, VVC, and Trichomoniasis. Useful as a benchmark in comparative assay studies. |
| Hologic Panther Aptima BV & CV/TV Assays [1] | A commercial NAAT system that utilizes transcription-mediated amplification (TMA). Used in studies evaluating performance against traditional methods. |
| OSOM BVBLUE Test [49] | A rapid, CLIA-waived point-of-care test that detects sialidase enzyme activity associated with BV. Useful for studies on rapid diagnostics and treatment initiation. |
| Nugent Score Components (Gram stain reagents, microscope) [8] | The essential materials for the reference standard method. Required for any study seeking to validate a new molecular assay against the classical benchmark. |
| InPouch TV Culture System [57] | The culture system used as a reference standard for the detection and growth of Trichomonas vaginalis. |
| Specific Primers & TaqMan Probes [11] | Custom-designed oligonucleotides for in-house qPCR development. They target specific genes of interest (e.g., 16S rRNA, ITS2) for quantifying vaginal microbiota. |
| Machine Learning Algorithms (SVM, RF, DT) [11] | Not a physical reagent, but a critical modern tool. Used to develop sophisticated diagnostic models from complex multiplex PCR data, improving predictive performance. |
The evidence demonstrates that molecular testing provides a clear advantage over traditional microscopy by delivering objective, reproducible, and highly granular data on vaginal microbiota. For the research community, these technologies are indispensable for understanding the complex etiologies of AV, STIs, and candidiasis, and for developing the next generation of targeted therapies and personalized treatment approaches. The future of vaginal health research lies in integrating these molecular insights to move beyond the limitations of the Nugent score.
Accurate patient stratification is a foundational element of successful clinical trials in women's health, directly influencing the reliability of efficacy endpoints and the speed of therapeutic development. For conditions like bacterial vaginosis (BV), a common gynecological disorder associated with significant reproductive morbidity and increased susceptibility to sexually transmitted infections, diagnostic methodology becomes particularly crucial [1] [56]. BV affects approximately 29% of women globally, with recurrence rates reaching 50% within six months despite antibiotic therapy, highlighting the pressing need for more effective treatments [70]. The traditional reliance on microscopy-based Nugent scoring for BV diagnosis, while established, presents significant limitations for clinical trial design, including subjectivity, technical expertise requirements, and inability to resolve intermediate cases [71] [2] [56]. Molecular diagnostics now offer transformative potential for precision patient stratification, enabling clinical trials to target specific microbial profiles, reduce misclassification, and generate more definitive endpoints for evaluating novel therapeutics [1] [4] [56].
Comparative Analysis of BV Diagnostic Modalities
Performance Characteristics of Current Diagnostic Methods
Table 1: Comparative performance of BV diagnostic methods for clinical trial stratification
| Diagnostic Method | Sensitivity (%) | Specificity (%) | Throughput | Objective Quantification | Intermediate Case Resolution |
|---|---|---|---|---|---|
| Nugent Scoring | 76.0 [56] | 76.0 [56] | Low | No | No [2] [56] |
| Amsel's Criteria | 37-70 [4] | Variable | Low | No | No |
| qPCR (L. crispatus + G. vaginalis) | 99.3 [4] | 97.8 [4] | High | Yes | Yes [2] |
| Deep Learning (Nugent Automation) | 89-94 [71] | 89-94 [71] | Medium | Partial | Limited |
| Multiplex PCR (Commercial Panels) | 90.0-97.5 [1] [57] | 85.0-96.3 [1] [57] | High | Yes | Yes |
Resolution of Diagnostic Ambiguity: The Intermediate Nugent Challenge
A critical advantage of molecular diagnostics for patient stratification is their ability to resolve diagnostically ambiguous cases. Approximately 10-20% of vaginal samples yield intermediate Nugent scores (4-6), creating uncertainty in patient eligibility and endpoint determination for clinical trials [2] [56]. Molecular methods effectively decipher these indeterminate cases through quantitative assessment of key bacterial targets. Research demonstrates that quantitative PCR (qPCR) targeting Lactobacillus species, Gardnerella vaginalis, and Atopobium vaginae can accurately categorize intermediate Nugent cases as BV-positive or BV-negative based on objective thresholds [2]. This capability significantly reduces misclassification in clinical trial populations, enhancing the statistical power to detect true treatment effects.
Molecular Assay Configurations for Precision Stratification
Optimized Bacterial Target Combinations
Table 2: Key molecular targets for BV diagnostic panels in clinical research
| Target Organism | Function in Vaginal Microbiome | Detection Method | Clinical Utility |
|---|---|---|---|
| Lactobacillus crispatus | Protective; produces lactic acid and H₂O₂ | qPCR [4] [2] | Marker of vaginal health |
| Gardnerella vaginalis | BV-associated anaerobe; biofilm former | qPCR [4] [2] | Primary pathogen marker |
| Fannyhessea vaginae | BV-associated anaerobe (formerly Atopobium) | qPCR [4] | High specificity for BV |
| BVAB-2 | BV-associated bacterium | PCR [4] | Supplemental marker |
| Megasphaera-1 | BV-associated bacterium | PCR [4] | Supplemental marker |
Research indicates that combinatorial approaches using two key targets provide optimal diagnostic performance while maintaining cost-effectiveness for large-scale trial screening. The combination of L. crispatus with G. vaginalis demonstrates exceptional sensitivity (99.3%) and specificity (97.8%), effectively capturing the essential dynamics of vaginal dysbiosis [4]. Similarly, pairing L. crispatus with F. vaginae achieves 98.9% sensitivity and 98.7% specificity [4]. These streamlined panels offer significant advantages over complex multi-target panels that increase reagent costs and analytical complexity without substantial diagnostic improvement.
Advanced Algorithmic Approaches for Population-Specific Stratification
Machine learning algorithms applied to 16S rRNA sequencing data represent the cutting edge of BV diagnostics for clinical trial stratification. However, recent evidence indicates that predictive performance varies significantly across ethnic groups, with models typically exhibiting lower accuracy for Black women [3]. This disparity reflects genuine biological variations in vaginal microbiome composition across populations, with Black and Hispanic women more frequently exhibiting diverse vaginal microbiota even in healthy states [3]. For clinical trials targeting global populations, these findings underscore the necessity of implementing ethnicity-stratified analytical approaches or developing population-specific algorithms to ensure equitable diagnostic performance across all participant subgroups [3].
Experimental Protocols for Diagnostic Validation
Reference Standardized qPCR Protocol for BV Assessment
Sample Collection and Processing: Vaginal samples are collected using sterile nylon-flocked swabs from the mid-vaginal region and immediately placed in Amies transport medium [56]. Specimens should be transported to the laboratory within 4-6 hours and stored at -80°C if batch processing [56].
DNA Extraction: Bacterial pellets are pretreated with enzymatic lysis buffer containing lysozyme (20 mg/ml) and incubated at 37°C for 30 minutes [56]. Proteinase K and buffer AL are then added followed by incubation at 56°C for 30 minutes [56]. DNA purification is completed using commercial silica-membrane columns with elution in molecular-grade water [56].
Quantitative PCR Amplification:
- Reaction volume: 20-25µL containing master mix, primers, and probes
- Thermal cycling: 95°C for 10 minutes, followed by 40 cycles of 95°C for 15 seconds and 60°C for 1 minute
- Target bacteria: Lactobacillus crispatus, Gardnerella vaginalis, Fannyhessea vaginae [4]
- Quantification standards: Serial dilutions of quantified genomic DNA for each target to generate standard curves [2]
Diagnostic Interpretation: Results are interpreted using validated algorithms that incorporate quantitative thresholds for each bacterial target to determine BV status [4] [2]. The combination of L. crispatus depletion with increased abundance of G. vaginalis and/or F. vaginae provides optimal diagnostic accuracy [4].
Workflow Comparison: Traditional vs. Molecular Diagnostics
Research Toolkit: Essential Reagents and Platforms
Table 3: Research reagent solutions for BV diagnostic implementation
| Reagent/Platform | Specific Function | Research Application |
|---|---|---|
| Nylon-flocked ESwabs | Optimal sample collection and release | Standardized vaginal specimen collection [56] |
| Amies Transport Medium | Preserves microbial viability during transport | Maintains sample integrity for molecular analysis [56] |
| Lysozyme (20 mg/mL) | Enzymatic cell wall lysis | Initial breakdown of Gram-positive bacteria [56] |
| Proteinase K | Protein degradation | Comprehensive cellular lysis [56] |
| Silica-membrane DNA Columns | Nucleic acid purification | High-quality DNA extraction [56] |
| Species-specific Primers/Probes | Target amplification and detection | qPCR identification of key BV-associated organisms [4] [2] |
| Quantified Genomic DNA Standards | Standard curve generation | Absolute quantification of bacterial loads [2] |
| 16S rRNA Sequencing Reagents | Microbiome profiling | Comprehensive microbial community analysis [3] [56] |
Implications for Clinical Trial Design and Therapeutic Development
Enhancing Endpoint Precision and Statistical Power
The implementation of molecular diagnostics for patient stratification directly addresses two critical challenges in BV therapeutic development: high placebo response rates in clinical trials and frequent disease recurrence. By ensuring that trial populations are precisely characterized according to their microbial profiles, molecular stratification reduces heterogeneity and increases the signal-to-noise ratio for detecting true therapeutic effects [1] [4]. This precision is particularly valuable for evaluating targeted antimicrobial agents, microbiome-based therapeutics, and prevention strategies for high-risk populations. Furthermore, quantitative molecular endpoints provide sensitive measures of microbial response to intervention, potentially serving as surrogate endpoints that could accelerate therapeutic development timelines [4] [2].
Advancing Personalized Medicine Approaches in Women's Health
The enhanced resolution of molecular diagnostics enables more sophisticated trial designs that account for the considerable heterogeneity in BV presentation and treatment response. Rather than treating BV as a monolithic condition, trials can stratify participants according to specific microbial constellations (e.g., Gardnerella-dominant vs. Fannyhessea-dominant profiles) [4]. This approach aligns with evolving understanding that different microbial configurations may respond differently to targeted therapies. Additionally, molecular assessment of vaginal microbiota restoration following treatment provides crucial insights into therapeutic mechanisms beyond simple symptom resolution, potentially identifying interventions that promote sustained colonization with protective Lactobacillus species [4] [56].
The validation and implementation of molecular diagnostics for BV clinical trial stratification represents a paradigm shift in women's health therapeutic development. By replacing subjective, labor-intensive microscopy with precise, quantitative molecular assessment, researchers can achieve unprecedented accuracy in patient selection, endpoint determination, and therapeutic response evaluation. The combinatorial approach targeting L. crispatus with key pathogens such as G. vaginalis provides an optimal balance of diagnostic performance and practical implementation for large-scale trials [4]. As therapeutic strategies for BV evolve to include partner treatment approaches based on emerging evidence of sexual transmission [49] [70], precise diagnostic assessment becomes increasingly essential for evaluating comprehensive intervention strategies. The integration of these advanced diagnostic methodologies into clinical trial protocols will accelerate the development of more effective, targeted therapies for this prevalent and impactful condition.
Conclusion
The validation of molecular diagnostics against the traditional Nugent score marks a paradigm shift in BV research. While the Nugent score has served as a valuable, cost-effective tool, its limitations in subjectivity, inability to identify specific pathogens, and the problematic 'intermediate' category are clear. Molecular methods, particularly quantitative multiplex PCR, offer superior sensitivity (e.g., 90.5%-96.7%) and specificity (e.g., 85.8%-95%), providing an objective, reproducible, and comprehensive profile of the vaginal microbiome. For researchers and drug developers, this accuracy is paramount for understanding BV pathogenesis, identifying new therapeutic targets, and ensuring precise patient enrollment in clinical trials. Future directions should focus on standardizing molecular panels for global use, validating cost-effective strategies like MALDI-TOF, and exploring the integration of microbiome sequencing with machine learning to predict treatment response and recurrence, ultimately advancing personalized medicine for bacterial vaginosis.
References
- 1. From microscopy to molecular diagnostics [https://pmc.ncbi.nlm.nih.gov/articles/PMC12502618/]
- 2. Utility of Real-time PCR for Bacterial Vaginosis diagnosis [https://www.sciencedirect.com/science/article/abs/pii/S0732889323002614]
- 3. Population-level predictive variation in machine learning ... [https://www.nature.com/articles/s44294-025-00092-w]
- 4. Best among the key molecular diagnostic markers of bacterial ... [https://amb-express.springeropen.com/articles/10.1186/s13568-025-01847-z]
- 5. Molecular Testing for the Diagnosis of Bacterial Vaginosis [https://www.mdpi.com/1422-0067/25/1/449]
- 6. AI-Powered Nugent Scoring and the Future of Microbiome ... [https://carbgem.com/en/news-20250401-en/]
- 7. Applying machine learning with MobileNetV2 model for ... [https://www.nature.com/articles/s41598-025-04626-9]
- 8. Molecular Testing for the Diagnosis of Bacterial Vaginosis [https://pmc.ncbi.nlm.nih.gov/articles/PMC10779368/]
- 9. The evaluation of the Allplex™ BV molecular assay for the ... [https://www.sciencedirect.com/science/article/abs/pii/S0732889323000342]
- 10. Diagnostic performance of DNA probe-based and PCR-based ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10581173/]
- 11. Vaginal microbiota molecular profiling and diagnostic ... [https://www.frontiersin.org/journals/cellular-and-infection-microbiology/articles/10.3389/fcimb.2024.1377225/full]
- 12. Development of Method and Performance for ... [https://www.sciencedirect.com/science/article/pii/S0002937825005976]
- 13. Evaluation of the user experience for a point of care molecular ... [https://bmcinfectdis.biomedcentral.com/articles/10.1186/s12879-025-11304-8]
- 14. Healthcare utilization and costs following molecular ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11650384/]
- 15. Bacterial Vaginosis: Current Diagnostic Avenues and ... [https://www.frontiersin.org/journals/cellular-and-infection-microbiology/articles/10.3389/fcimb.2020.00354/full]
- 16. Comparison of Hay's Criteria with Nugent's Scoring System for Diagnosis of Bacterial Vaginosis - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3697286/]
- 17. Evaluation of Various Diagnostic Strategies for Bacterial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10821145/]
- 18. Drexel Researchers Develop New DNA Test For BV [https://drexel.edu/medicine/news-events/news-archive/2025/March/Drexel-Researchers-Develop-New-DNA-Test/]
- 19. New Research on BV and a Possible Link to Chlamydia [https://www.ashasexualhealth.org/new-research-on-bv-and-a-possible-link-to-chlamydia/]
- 20. Comparison of Amsel criteria, Nugent score, culture and two CE-IVD marked quantitative real-time PCRs with microbiota analysis for the diagnosis of bacterial vaginosis - PubMed [https://pubmed.ncbi.nlm.nih.gov/30903536/]
- 21. Real-world clinical burden of patients presenting with ... [https://www.sciencedirect.com/science/article/pii/S2666577825000656]
- 22. Common cervicovaginal sequencing methods result in ... [https://www.nature.com/articles/s44294-025-00089-5]
- 23. Bacterial Vaginosis - STI Treatment Guidelines [https://www.cdc.gov/std/treatment-guidelines/bv.htm]
- 24. Comparative study of Amsel's criteria and Nugent scoring ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8369704/]
- 25. BVBlue as a diagnostic instrument for the diagnosis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11869707/]
- 26. Table 4, Accuracy of Diagnostic Tests From Studies ... [https://www.ncbi.nlm.nih.gov/books/NBK555827/table/ch3.tab4/?report=objectonly]
- 27. Nugent score [https://en.wikipedia.org/wiki/Nugent_score]
- 28. Amsel Criteria - StatPearls - NCBI Bookshelf - NIH [https://www.ncbi.nlm.nih.gov/books/NBK542319/]
- 29. Real-Time PCR Test (Flora Select™) for Assessing the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12473051/]
- 30. Vaginal microbiota molecular profiling and diagnostic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11026559/]
- 31. Development of a Multiplex Real-Time Quantitative PCR ... [https://weekly.chinacdc.cn/en/article/doi/10.46234/ccdcw2024.173]
- 32. A multiplex real-time PCR assay for routine diagnosis of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4545173/]
- 33. Detection of Eight Respiratory Bacterial Pathogens Based on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7061119/]
- 34. Evaluation of Rapid Multiplex Reverse Transcription ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10455980/]
- 35. qPCR vs. ddPCR: Which Is Best for You? [https://www.bioradiations.com/qpcr-vs-ddpcr-assays-select-the-best-gene-expression-analysis-method-for-your-application1125/]
- 36. State of the Art for Diagnosis of Bacterial Vaginosis - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC10446871/]
- 37. Comparison between MICRO–CARD–FISH and 16S rRNA ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3615173/]
- 38. Molecular assessment of bacterial vaginosis by ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4841971/]
- 39. A New PNA-FISH Probe Targeting Fannyhessea vaginae [https://www.frontiersin.org/journals/cellular-and-infection-microbiology/articles/10.3389/fcimb.2021.779376/full]
- 40. NEMESISdb: A full length 16S rRNA gene dataset for the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12553003/]
- 41. Comparison of Two 16S rRNA Primers (V3–V4 and V4–V5 ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2021.637526/full]
- 42. Evaluation of the Aptima BV and CV/TV assays compared ... [https://www.sciencedirect.com/science/article/abs/pii/S0732889323000639]
- 43. Bacterial Vaginosis Diagnostics Market Size & Share ... [https://www.mordorintelligence.com/industry-reports/bacterial-vaginosis-diagnostics-market]
- 44. Real-world clinical burden of patients presenting with ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12167788/]
- 45. Nugent Score - an overview | ScienceDirect Topics [https://www.sciencedirect.com/topics/medicine-and-dentistry/nugent-score]
- 46. Evaluation of interobserver reliability of Nugent score for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6298156/]
- 47. Performance of deep learning models in predicting the nugent ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11705891/]
- 48. Revisiting Bacterial Vaginosis: Evidence for Sexual ... [https://www.cepheid.com/en-US/insights/insight-hub/community-and-global-health/2025/05/revisiting-bacterial-vaginosis-evidence-for-sexual-transmission.html]
- 49. Evolution to Revolution: Bacterial Vaginosis Testing & ... [https://blog.sekisuidiagnostics.com/dxdialogue/evolution-to-revolution-bacterial-vaginosis-testing-treatment]
- 50. Reframing Bacterial Vaginosis: New research supports STI ... [https://www.clinicallabs.com.au/about-us/doctor-media-releases/reframing-bacterial-vaginosis-wasant/]
- 51. molBV reveals immune landscape of bacterial vaginosis ... [https://www.nature.com/articles/s41467-021-27628-3]
- 52. Clinical laboratory evaluation of the Hologic Panther ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11723574/]
- 53. Statistical analysis in method comparison studies part one [https://acutecaretesting.org/en/articles/statistical-analysis-in-method-comparison-studies-part-one]
- 54. The Comparison of Methods Experiment [https://www.westgard.com/lessons/basic-method-validation/lesson23.html]
- 55. Bacterial vaginosis: a review of approaches to treatment ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10264601/]
- 56. Molecular Diagnosis of Vaginitis - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC6711928/]
- 57. Diagnostic Performance of a Molecular Test versus ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5971525/]
- 58. Diagnostic accuracy of quantitative real-time PCR assay ... [https://pubmed.ncbi.nlm.nih.gov/20814710/]
- 59. Comparison of two microscopic interpretations of vaginal ... [https://www.sciencedirect.com/science/article/pii/S0732889322000943]
- 60. Drexel Researchers Develop New DNA Test For BV [https://drexel.edu/news/archive/2025/March/Drexel-Researchers-Develop-New-DNA-Test]
- 61. ACOG Recommends Concurrent Sexual Partner Treatment ... [https://www.acog.org/news/news-releases/2025/10/acog-recommends-concurrent-sexual-partner-treatment-recurrent-bacterial-vaginosis-first-time]
- 62. identifying the complexities of clinical diagnostics for ... [https://pubmed.ncbi.nlm.nih.gov/40833126/]
- 63. The accuracy vs. coverage trade-off in patient-facing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7233065/]
- 64. Evaluation of the Vaginal Panel Realtime PCR kit... | PLOS One [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0313414]
- 65. Diagnostic performance of two molecular assays for the ... [https://link.springer.com/article/10.1007/s10096-019-03694-w]
- 66. Evaluation of a Multiplex Quantitative Real-Time PCR A Diagnostic [https://www.longdom.org/open-access/diagnostic-evaluation-of-a-multiplex-quantitative-realtime-pcr-assay-forbacterial-vaginosis-40450.html]
- 67. BVBlue as a diagnostic instrument for the diagnosis of bacterial... [https://bmcwomenshealth.biomedcentral.com/articles/10.1186/s12905-025-03615-4]
- 68. A New Real-Time PCR Test (Flora Select™) and Nugent ... [https://www.ncbi.nlm.nih.gov/sites/entrez/39458419]
- 69. Rapid molecular diagnostic method for Gardnerella ... [https://www.sciencedirect.com/science/article/abs/pii/S0009898125005042]
- 70. Revisiting Bacterial Vaginosis: Evidence for Sexual ... [https://www.cepheid.com/content/www-cepheid-com/en-NZ/insights/insight-hub/community-and-global-health/2025/05/revisiting-bacterial-vaginosis-evidence-for-sexual-transmission.html]
- 71. Performance of deep learning models in predicting the ... [https://pubmed.ncbi.nlm.nih.gov/39560430/]